Abstract
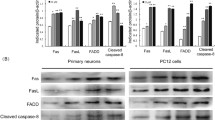
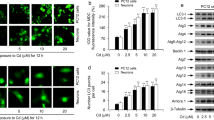
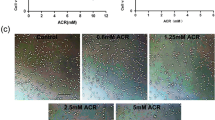
To investigate the role of mitogen-activated protein kinase (MAPK) and downstream events in cadmium (Cd)-induced neuronal apoptosis executed via the mitochondrial apoptotic pathway, this study used the PC-12 cell line as a neuronal model. The result showed that Cd significantly decreased cell viability and the Bcl-2 / Bax ratio and increased the percentage of apoptotic cells, release of cytochrome c, caspase-3, and poly(ADP-ribose) polymerase cleavage, and nuclear translocation of apoptosis-inducing factor (AIF) and endonuclease G. In addition, exposure to Cd-induced phosphorylation of extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK), and p38 MAPK. Inhibition of ERK and JNK, but not p38 MAPK, partially protected the cells from Cd-induced apoptosis. ERK and JNK inhibition also blocked alteration of the Bcl-2 / Bax ratio and cytochrome c release and suppressed caspase-3 and poly(ADP-ribose) polymerase cleavage and AIF and endonuclease G nuclear translocation. Taken together, these data suggest that the ERK- and JNK-mediated mitochondrial apoptotic pathway played an important role in Cd-induced PC12 cells apoptosis.







Similar content being viewed by others
References
Mendez-Armenta M, Rios C (2007) Cadmium neurotoxicity. Environ Toxicol Pharmacol 23:350–358. doi:10.1016/j.etap.2006.11.009
Shukla A, Shukla GS, Srimal RC (1996) Cadmium-induced alterations in blood–brain barrier permeability and its possible correlation with decreased microvessel antioxidant potential in rat. Hum Exp Toxicol 15:400–405
Lopez E, Arce C, Oset-Gasque MJ, Canadas S, Gonzalez MP (2006) Cadmium induces reactive oxygen species generation and lipid peroxidation in cortical neurons in culture. Free Radic Biol Med 40:940–951. doi:10.1016/j.freeradbiomed.2005.10.062
Chen L, Xu B, Liu L, Luo Y, Zhou H, Chen W, Shen T, Han X, Kontos CD, Huang S (2011) Cadmium induction of reactive oxygen species activates the mTOR pathway, leading to neuronal cell death. Free Radic Biol Med 50:624–632. doi:10.1016/j.freeradbiomed.2010.12.032
Bonithon-Kopp C, Huel G, Moreau T, Wendling R (1986) Prenatal exposure to lead and cadmium and psychomotor development of the child at 6 years. Neurobehav Toxicol Teratol 8:307–310
Jiang LF, Yao TM, Zhu ZL, Wang C, Ji LN (2007) Impacts of Cd(II) on the conformation and self-aggregation of Alzheimer’s tau fragment corresponding to the third repeat of microtubule-binding domain. Biochim Biophys Acta 1774:1414–1421. doi:10.1016/j.bbapap.2007.08.014
Chang L, Karin M (2001) Mammalian MAP kinase signalling cascades. Nature 410:37–40. doi:10.1038/35065000
Johnson GL, Lapadat R (2002) Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 298:1911–1912. doi:10.1126/science.1072682
Chen L, Liu L, Luo Y, Huang S (2008) MAPK and mTOR pathways are involved in cadmium-induced neuronal apoptosis. J Neurochem 105:251–261. doi:10.1111/j.1471-4159.2007.05133.x
Rigon AP, Cordova FM, Oliveira CS, Posser T, Costa AP, Silva IG, Santos DA, Rossi FM, Rocha JB, Leal RB (2008) Neurotoxicity of cadmium on immature hippocampus and a neuroprotective role for p38 MAPK. Neurotoxicology 29:727–734. doi:10.1016/j.neuro.2008.04.017
Wang S, Tang M, Pei B, Xiao X, Wang J, Hang H, Wu L (2008) Cadmium-induced germline apoptosis in Caenorhabditis elegans: the roles of HUS1, p53, and MAPK signaling pathways. Toxicol Sci 102:345–351. doi:10.1093/toxsci/kfm220
Valbonesi P, Ricci L, Franzellitti S, Biondi C, Fabbri E (2008) Effects of cadmium on MAPK signalling pathways and HSP70 expression in a human trophoblast cell line. Placenta 29:725–733. doi:10.1016/j.placenta.2008.05.004
Hirano S, Sun X, DeGuzman CA, Ransom RF, McLeish KR, Smoyer WE, Shelden EA, Welsh MJ, Benndorf R (2005) p38 MAPK/HSP25 signaling mediates cadmium-induced contraction of mesangial cells and renal glomeruli. Am J Physiol Renal Physiol 288:F1133–F1143. doi:10.1152/ajprenal.00210.2004
Kim SM, Park JG, Baek WK, Suh MH, Lee H, Yoo SK, Jung KH, Suh SI, Jang BC (2008) Cadmium specifically induces MKP-1 expression via the glutathione depletion-mediated p38 MAPK activation in C6 glioma cells. Neurosci Lett 440:289–293. doi:10.1016/j.neulet.2008.05.064
Nemmiche S, Chabane-Sari D, Kadri M, Guiraud P (2012) Cadmium-induced apoptosis in the BJAB human B cell line: involvement of PKC/ERK1/2/JNK signaling pathways in HO-1 expression. Toxicology 300:103–111. doi:10.1016/j.tox.2012.05.003
Muller L (1986) Consequences of cadmium toxicity in rat hepatocytes: mitochondrial dysfunction and lipid peroxidation. Toxicology 40:285–295
Hossain S, Liu HN, Nguyen M, Shore G, Almazan G (2009) Cadmium exposure induces mitochondria-dependent apoptosis in oligodendrocytes. Neurotoxicology 30:544–554. doi:10.1016/j.neuro.2009.06.001
Lasfer M, Vadrot N, Aoudjehane L, Conti F, Bringuier AF, Feldmann G, Reyl-Desmars F (2008) Cadmium induces mitochondria-dependent apoptosis of normal human hepatocytes. Cell Biol Toxicol 24:55–62. doi:10.1007/s10565-007-9015-0
Yuan Y, Jiang C-Y, Xu H, Sun Y, Hu F-F, Bian J-C, Liu X-Z, Gu J-H, Liu Z-P (2013) Cadmium-induced apoptosis in primary rat cerebral cortical neurons culture is mediated by a calcium signaling pathway. PLoS One 8:e64330
Wang Q, Zhu J, Zhang K, Jiang C, Wang Y, Yuan Y, Bian J, Liu X, Gu J, Liu Z (2013) Induction of cytoprotective autophagy in PC-12 cells by cadmium. Biochem Biophys Res Commun 438:186–192. doi:10.1016/j.bbrc.2013.07.050
Bossy-Wetzel E, Green DR (2000) Detection of apoptosis by annexin V labeling. Methods Enzymol 322:15–18
Jayanthi S, Deng X, Noailles PA, Ladenheim B, Cadet JL (2004) Methamphetamine induces neuronal apoptosis via cross-talks between endoplasmic reticulum and mitochondria-dependent death cascades. FASEB J 18:238–251. doi:10.1096/fj.03-0295com
Chen L, Liu L, Huang S (2008) Cadmium activates the mitogen-activated protein kinase (MAPK) pathway via induction of reactive oxygen species and inhibition of protein phosphatases 2A and 5. Free Radic Biol Med 45:1035–1044. doi:10.1016/j.freeradbiomed.2008.07.011
Shen J, Wu Y, Xu JY, Zhang J, Sinclair SH, Yanoff M, Xu G, Li W, Xu GT (2010) ERK- and Akt-dependent neuroprotection by erythropoietin (EPO) against glyoxal-AGEs via modulation of Bcl-xL, Bax, and BAD. Invest Ophthalmol Vis Sci 51:35–46. doi:10.1167/iovs.09-3544
Asakura T, Maeda K, Omi H, Matsudaira H, Ohkawa K (2008) The association of deamidation of Bcl-xL and translocation of Bax to the mitochondria through activation of JNK in the induction of apoptosis by treatment with GSH-conjugated DXR. Int J Oncol 33:389–395
Xu B, Chen S, Luo Y, Chen Z, Liu L, Zhou H, Chen W, Shen T, Han X, Chen L, Huang S (2011) Calcium signaling is involved in cadmium-induced neuronal apoptosis via induction of reactive oxygen species and activation of MAPK/mTOR network. PLoS One 6:e19052. doi:10.1371/journal.pone.0019052
Chang KC, Hsu CC, Liu SH, Su CC, Yen CC, Lee MJ, Chen KL, Ho TJ, Hung DZ, Wu CC, Lu TH, Su YC, Chen YW, Huang CF (2013) Cadmium induces apoptosis in pancreatic beta-cells through a mitochondria-dependent pathway: the role of oxidative stress-mediated c-Jun N-terminal kinase activation. PLoS One 8:e54374. doi:10.1371/journal.pone.0054374
Mertens HJ, Heineman MJ, Evers JL (2002) The expression of apoptosis-related proteins Bcl-2 and Ki67 in endometrium of ovulatory menstrual cycles. Gynecol Obstet Invest 53:224–230
Biagioli M, Watjen W, Beyersmann D, Zoncu R, Cappellini C, Ragghianti M, Cremisi F, Bucci S (2001) Cadmium-induced apoptosis in murine fibroblasts is suppressed by Bcl-2. Arch Toxicol 75:313–320
Oh SH, Lee BH, Lim SC (2004) Cadmium induces apoptotic cell death in WI 38 cells via caspase-dependent Bid cleavage and calpain-mediated mitochondrial Bax cleavage by Bcl-2-independent pathway. Biochem Pharmacol 68:1845–1855. doi:10.1016/j.bcp.2004.06.021
Orrenius S (2004) Mitochondrial regulation of apoptotic cell death. Toxicol Lett 149:19–23. doi:10.1016/j.toxlet.2003.12.017
Mohamad N, Gutiérrez A, Núñez M, Cocca C, Martín G, Cricco G, Medina V, Rivera E, Bergoc R (2005) Mitochondrial apoptotic pathways. Biocell 29:149–161
Robertson JD, Orrenius S (2002) Role of mitochondria in toxic cell death. Toxicology 181–182:491–496
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES, Wang X (1997) Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91:479–489
Kiechle T, Dedeoglu A, Kubilus J, Kowall NW, Beal MF, Friedlander RM, Hersch SM, Ferrante RJ (2002) Cytochrome C and caspase-9 expression in Huntington’s disease. Neuromol Med 1:183–195. doi:10.1385/NMM:1:3:183
Joza N, Pospisilik JA, Hangen E, Hanada T, Modjtahedi N, Penninger JM, Kroemer G (2009) AIF: not just an apoptosis-inducing factor. Ann N Y Acad Sci 1171:2–11. doi:10.1111/j.1749-6632.2009.04681.x
Li LY, Luo X, Wang X (2001) Endonuclease G is an apoptotic DNase when released from mitochondria. Nature 412:95–99
Ohsato T, Ishihara N, Muta T, Umeda S, Ikeda S, Mihara K, Hamasaki N, Kang D (2002) Mammalian mitochondrial endonuclease G. Digestion of R-loops and localization in intermembrane space. Eur J Biochem 269:5765–5770
Tsujimoto Y (2003) Cell death regulation by the Bcl-2 protein family in the mitochondria. J Cell Physiol 195:158–167. doi:10.1002/jcp.10254
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 31101866, to Yan Yuan; No. 31172373, to Zong-ping Liu; No. 31302058, to Yi Wang ) and a project Funded by Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Conflict of interest
All other authors have read the manuscript and have agreed to submit it in its current form for consideration for publication in the journal.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jiang and Yuan contributed equally to this work.
Rights and permissions
About this article
Cite this article
Jiang, C., Yuan, Y., Hu, F. et al. Cadmium Induces PC12 Cells Apoptosis via an Extracellular Signal-Regulated Kinase and c-Jun N-Terminal Kinase-Mediated Mitochondrial Apoptotic Pathway. Biol Trace Elem Res 158, 249–258 (2014). https://doi.org/10.1007/s12011-014-9918-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-014-9918-6