Abstract
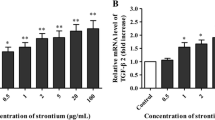
Strontium stimulates cartilage matrix formation in vitro. However, the mechanisms governing these effects have not yet been extensively reported. In this study, chondrocytes were isolated from rat articular cartilage by enzymatic digestion and cultured for 24–72 h with 1–5 mM strontium. We investigated the effects of different concentrations of strontium on collagen content, type II collagen, insulin-like growth factor (IGF-1) and matrix metalloproteinase (MMP)-13 expression in rat cultured articular chondrocytes in vitro. The collagen content of the chondrocytes, determined as hydroxyproline, was measured by a colorimetry method. Type II collagen, IGF-1, and MMP-13 mRNA abundance and protein expression levels were determined by real-time polymerase chain reaction (real-time PCR) and western blot, respectively. The results showed that collagen content from the chondrocytes extracellular matrix increased with increasing strontium concentration. Moreover, 3 and 5 mM strontium strongly stimulated protein expression and mRNA levels of type II collagen and IGF-1. Conversely, MMP-13 expression in chondrocytes decreased dose-dependently with increasing strontium concentration. These results should provide insight into the ability of strontium to promote chondrocyte extracellular matrix synthesis. Strontium could promote collagen synthesis and suppress collagen degradation via the repression of MMP-13 expression.







Similar content being viewed by others
References
Goldring MB (2000) The role of the chondrocyte in osteoarthritis. Arthritis Rheum 43:1916–1926
Aigner T, Stöve J (2003) Collagens-major component of the physiological cartilage matrix, major target of cartilage degeneration, major tool in cartilage repair. Adv Drug Deliv Rev 55:1569–1593
Adachi N, Sato K, Usas A et al (2002) Muscle derived, cell based ex vivo gene therapy for treatment of full thickness articular cartilage defects. J Rheumatol 29:1920–1930
Angele P, Schumann D, Angele M et al (2004) Cyclic, mechanical compression enhances chondrogenesis of mesenchymal progenitor cells in tissue engineering scaffolds. Biorheology 41:335–348
Poole A, Kobayashi M, Yasuda T et al (2002) Type II collagen degradation and its regulation in articular cartilage in osteoarthritis. Ann Rheum Dis 61:ii78–ii81
Gangl M, Serteyn D, Lejeune JP et al (2007) A type II-collagen derived peptide and its nitrated form as new markers of inflammation and cartilage degradation in equine osteochondral lesions. Res Vet Sci 82:68–75
Lark M, Bayne E, Flanagan J et al (1997) Aggrecan degradation in human cartilage. Evidence for both matrix metalloproteinase and aggrecanase activity in normal, osteoarthritic, and rheumatoid joints. J Clin Invest 100:93
Hadjidakis DJ, Androulakis II (2007) Bone remodeling. Ann NY Acad Sci 1092:385–396
Bonewald L, Dallas S (2004) Role of active and latent transforming growth factor β in bone formation. J Cell Biochem 55:350–357
Fukumoto T, Sperling J, Sanyal A et al (2003) Combined effects of insulin-like growth factor-1 and transforming growth factor-β1 on periosteal mesenchymal cells during chondrogenesis in vitro. Osteoarthr Cartil 11:55–64
Stickens D, Behonick DJ, Ortega N et al (2004) Altered endochondral bone development in matrix metalloproteinase 13-deficient mice. Development 131:5883–5895
Reboul P, Pelletier JP, Tardif G et al (1996) The new collagenase, collagenase-3, is expressed and synthesized by human chondrocytes but not by synoviocytes. A role in osteoarthritis. J Clin Invest 97:2011
Billinghurst RC, Dahlberg L, Ionescu M et al (1997) Enhanced cleavage of type II collagen by collagenases in osteoarthritic articular cartilage. J Clin Invest 99:1534
Wang X, Manner PA, Horner A et al (2004) Regulation of MMP-13 expression by RUNX2 and FGF2 in osteoarthritic cartilage. Osteoarthr Cartil 12:963–973
Li Z, Lam W, Yang C et al (2007) Chemical composition, crystal size and lattice structural changes after incorporation of strontium into biomimetic apatite. Biomaterials 28:1452–1460
Marie PJ, Hott M, Modrowski D et al (2005) An uncoupling agent containing strontium prevents bone loss by depressing bone resorption and maintaining bone formation in estrogen–deficient rats. J Bone Miner Res 20:1065–1074
Henrotin Y, Labasse A, Zheng S et al (2001) Strontium ranelate increases cartilage matrix formation. J Bone Miner Res 16:299–308
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29:e45–e45
Yuan X, Wang J, Zhu X et al (2011) Effect of Copper on Levels of Collagen and Alkaline Phosphatase Activity from Chondrocytes in Newborn Piglets In Vitro. Biol Trace Elem Res 1–9
Wang J, Zhu X, Li X et al (2011) Effects of copper on proliferation and autocrine secretion of insulin-like growth factor-1 (IGF-1) and igf-binding protein-3 (IGFBP-3) in chondrocytes from newborn pigs in vitro. Biol Trace Elem Res 144:588–596
Henrotin Y, Addison S, Kraus V et al (2007) Type II collagen markers in osteoarthritis: what do they indicate? Curr Opin Rheumatol 19:444
Reginster JY, Deroisy R, Jupsin I (2003) Strontium ranelate: a new paradigm in the treatment of osteoporosis. Drugs Today 39:89–101
Henrotin YE, Deberg MA, Crielaard JM et al (2006) Avocado/soybean unsaponifiables prevent the inhibitory effect of osteoarthritic subchondral osteoblasts on aggrecan and type II collagen synthesis by chondrocytes. J Rheumatol 33:1668–1678
Loeser RF, Pacione CA, Chubinskaya S (2003) The combination of insulin–like growth factor 1 and osteogenic protein 1 promotes increased survival of and matrix synthesis by normal and osteoarthritic human articular chondrocytes. Arthritis Rheum 48:2188–2196
Martin J, Buckwalter J (2000) The role of chondrocyte-matrix interactions in maintaining and repairing articular cartilage. Biorheology 37:129–140
Gulhan I, Bilgili S, Gunaydin R et al (2008) The effect of strontium ranelate on serum insulin like growth factor-1 and leptin levels in osteoporotic post-menopausal women: a prospective study. Arch Gynecol Obstet 278:437–441
Li Z, Lu WW, Chiu PKY et al (2009) Strontium–calcium coadministration stimulates bone matrix osteogenic factor expression and new bone formation in a large animal model. J Orthop Res 27:758–762
Knäuper V, López-Otin C, Smith B et al (1996) Biochemical characterization of human collagenase-3. J Biol Chem 271:1544–1550
Knäuper V, Will H, López-Otin C et al (1996) Cellular mechanisms for human procollagenase-3 (MMP-13) activation. J Biol Chem 271:17124–17131
Sandell LJ, Aigner T (2001) Articular cartilage and changes in arthritis. An introduction: cell biology of osteoarthritis. Arthritis Res 3:107–113
Wu CW, Tchetina EV, Mwale F et al (2002) Proteolysis involving matrix metalloproteinase 13 (collagenase-3) is required for chondrocyte differentiation that is associated with matrix mineralization. J Bone Miner Res 17:639–651
Mitchell PG, Magna HA, Reeves LM et al (1996) Cloning, expression, and type II collagenolytic activity of matrix metalloproteinase-13 from human osteoarthritic cartilage. J Clin Invest 97:761
Lee HP, Choi YJ, Cho KA et al (2012) Effect of Spa spring water on cytokine expression in human keratinocyte HaCaT cells and on differentiation of CD4+ T cells. Ann Dermatol 24:324–336
Acknowledgments
This work was supported by the Program for New Century Excellent Talents in University (NCET-11-0199), the Joint Funds of the NSFC-Yunnan province (U0836601), the National Key Technology R&D Program (Grant No. 2012BAD12B03), the National Natural Science Foundation of China (Grant Nos. 30600441, 30972212, 30871897 and 31072178) and the Science Fund for Distinguished Young Scholars of Jilin University (Grant No. 201100009).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, J., Zhu, X., Liu, L. et al. Effects of Strontium on Collagen Content and Expression of Related Genes in Rat Chondrocytes Cultured In Vitro . Biol Trace Elem Res 153, 212–219 (2013). https://doi.org/10.1007/s12011-013-9640-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-013-9640-9