Abstract
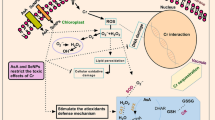
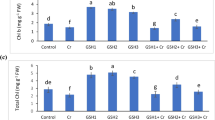
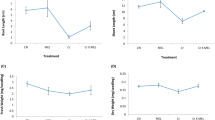
A hydroponic experiment was conducted to determine the possible effect of exogenous glutathione (GSH) in alleviating chromium (Cr) stress through examining plant growth, chlorophyll contents, antioxidant enzyme activity, and lipid peroxidation in rice seedlings exposed to Cr toxicity. The results showed that plant growth and chlorophyll content were dramatically reduced when rice plants were exposed to 100 μM Cr. Addition of GSH in the culture solution obviously alleviated the reduction of plant growth and chlorophyll content. The activities of some antioxidant enzymes, including superoxide dismutase, catalase (CAT) and glutathione reductase in leaves, and CAT and glutathione peroxidase in roots showed obvious increase under Cr stress. Addition of GSH reduced malondialdehyde accumulation and increased the activities of these antioxidant enzymes in both leaves and roots, suggesting that GSH may enhance antioxidant capacity in Cr-stressed plants. Furthermore, exogenous GSH caused significant decrease of Cr uptake and root-to-shoot transport in the Cr-stressed rice plants. It can be assumed that GSH is involved in Cr compartmentalization in root cells.



Similar content being viewed by others
References
Sinha S, Saxena R, Singh S (2005) Chromium induced lipid peroxidation in the plants of Pistia stratiotes L.: role of antioxidants and antioxidant enzymes. Chemosphere 58:595–604
Zayed AM, Terry N (2003) Chromium in the environment: factors affecting biological remediation. Plant Soil 249:139–156
Kotas J, Stasicka Z (2000) Chromium occurrence in the environment on methods of its speciation. Environ Pollut 107:263–283
Dixit V, Pandey V, Shyam R (2002) Chromium ions inactivate electron transport and enhance superoxide generation in vivo in pea (Pisum sativum L. cv. Azad) root mitochondria. Plant Cell Environ 25:687–690
Panda SK (2003) Heavy metal phytotoxicity induces oxidative stress in Taxithelium sp. Cur Sci 84:631–633
Panda SK (2007) Chromium-mediated oxidative stress and ultrastructural changes in root cells of developing rice seedlings. J Plant Physiol 164:1419–1428
Shanker AK, Cervantes C, Loza-Tavera H, Avudainayagam S (2005) Chromium toxicity in plants. Environ Inter 31:739–753
Bai TH, Li Y, Ma FW, Shu HR, Han MY (2009) Exogenous salicylic acid alleviates growth inhibition and oxidative stress induced by hypoxia stress in Malus robusta Rehd. J Plant Growth Regul 28:358–366
Panda SK, Khan MH (2003) Antioxidant efficiency in rice (Oryza saliva L.) leaves under heavy metal toxicity. Biol Plant 30:23–29
Choudhury S, Panda SK (2005) Toxic effect, oxidative stress and ultrastructural changes in moss Taxitheelium nepalense (Schwaegr.) Broth. under lead and chromium toxicity. Water Air Soil Pollut 167:73–90
Molassiotis A, Sotiropoulos T, Tanou G, Diamantidis G, Therios L (2006) Boron-induced oxidative damage and antioxidant and nucleolytic responses in shoot tips culture of the apple rootstock EM 9 (Malus domestica Borkh). Environ Exp Bot 56:54–62
Ogawa K (2005) Glutahione-associated regulation of plant growth and stress responses. Antioxid Redox Sign 7:973–981
Salt DE, Rauser WE (1995) MgATP-dependent of phytochelatins across the tonoplast of oat roots. Plant Physiol 107:1293–1301
Wu FB, Chen F, Wei K, Zhang GP (2004) Effect of cadmium on free amino acid, glutathione and ascorbic acid concentrations in two barley genotypes (Hordeum vulgare L.) differing in cadmium tolerance. Chemosphere 57:447–454
Srivalli S, Khanna-Chopra R (2008) Role of glutathione in abiotic stress tolerance. In: Khan NA, Singh S, Umar S (eds) Sulfur assimilation and abiotic stress in plants. Springer, Berlin, pp 207–225
Sharma SS, Dietz KJ (2006) The significance of amino acids and amino acid-derived molecules in plant responses and adaptation to heavy metal stress. J Exp Bot 57:711–726
Zhu YL, Pilon-Smiths EA, Jouanin L, Terry N (1999) Overexpression of glutathione synthetase in Indian mustard enhances cadmium accumulation and tolerance. Plant Physiol 109:1141–1149
Zhu YL, Pilon-Smits EA, Tarun A, Weber SU, Jouanin L, Terry N (1999) Cadmium tolerance and accumulation in Indian mustard is enhanced by overexpressing g-glutamylcysteine synthetase. Plant Physiol 121:1169–1177
Xiang C, Werner B, Christensen E, Oliver DC (2001) The biological function of glutathione revisited in Arabidopsis transgenic plants with altered glutathione levels. Plant Physiol 126:564–574
Bogs J, Bourbouloux A, Cagnac O, Wachter W, Rausch T, Delrot S (2003) Functional characterization and expression analysis of a glutathione transporter, BjGT1, from Brassica juncea: evidence for regulation by heavy metal exposure. Plant Cell Environ 26:1703–1711
Zeng FR, Mao Y, Cheng WD, Wu FB, Zhang GP (2008) Genotypic and environmental variation in chromium, cadmium and lead concentrations in rice. Environ Pollut 153:309–314
Yoshida S, Forna DA, Cock HJ, Gomez KA (1976) Laboratory manual for physiological studies of rice. International Rice Research Institute, Los Baños, pp 62–63
Miller RO (1998) Nitric-perchloric acid wet digestion in an open vessel. In: Kalra YP (ed) Handbook of reference methods for plant analysis. CRC Press, Taylor & Francis Group, Boca Raton, pp 57–62
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Meth Enzymol 148:350–382
Hodges DM, DeLong JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Putter J (1974) Peroxidases. In: Bergmeyer HU (ed) Methods of enzymatic analysis: II. Academic, New York, pp 685–690
Aebi H (1984) Catalase in vitro. Meth Enzymol 105:121–126
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Garcia-Limones C, Hervas A, Navas-Cortes JA, Jimenez-Diaz RM, Tena M (2002) Induction of an antioxidant enzyme system and other oxidative stress markers associated with compatible and incompatible interactions between chickpea (Cicer arietinum L.) and Fusarium oxysporum f. sp. Ciceris. Physiol Mol Plant Pathol 61:325–337
Nagalakshmi N, Prasad MNV (2001) Responses of glutathione cycle enzymes and glutathione metabolism to copper stress in Scenedesmus bijugatus. Plant Sci 160:291–299
Mei B, Puryear JD, Newton RJ (2002) Assessment of Cr tolerance and accumulation in selected plant species. Plant Soil 247:223–231
Shanker AK (2003) Physiological, biochemical and molecular aspects of chromium toxicity and tolerance in selected crops and tree species. PhD thesis, Tamil Nadu Agricultural University. Coimbatore, India
Sinha S, Saxena R, Singh S (2002) Comparative studies on accumulation of Cr from metal solution and tannery effluent under repeated metal exposure by aquatic plants: its toxic effects. Environ Monitor Assess 80:17–31
Panda SK, Choudhury S (2005) Changes in nitrate reductase activity and oxidative stress response in the moss Polytrichum commune subjected to chromium, copper and zinc toxicity. Braz J Plant Physiol 17:191–197
Panda SK, Choudhury S (2005) Chromium stress in plants. Braz J Plant Physiol 17:95–102
Panda SK, Chaodhury I, Khan MH (2003) Heavy metal phytotoxicity induces lipid peroxidation and affect antioxidants in wheat leaves. Biol Planta 46:289–294
Shanker AK, Sudhagar R, Pathmanabhan G (2001) Growth, phytochelatin SH and antioxidative response of sunflower as affected by chromium speciation. 2nd International Congress of Plant Physiology on sustainable plant productivity under changing environment. New Delhi, India
Fridovich I (1986) Biological effects of superoxide radical. Arch Biochem Biophys 247:1–11
Matés JM (2000) Effects of antioxidant enzymes in the molecular control of reactive oxygen species toxicology. Toxicology 153:83–104
Shanker AK, Djanaguiraman M, Sudhagar R, Chandrashekar CN, Pathmanabhan G (2004) Differential antioxidative response of ascorbate glutathione pathway enzymes and metabolites to chromium speciation stress in green gram (Vigna radiata (L) R Wilczek, cv CO 4) roots. Plant Sci 166:1035–1043
Cobbett C (2000) Phytochelatins and their role in heavy metal detoxification. Plant Physiol 123:825–832
Diwan H, Khan I, Ahmad A, Iqbal M (2010) Induction of phytochelatins and antioxidant defence system in Brassica juncea and Vigna radiata in response to chromium treatments. Plant Growth Regul 61:97–107
Lima AIG, Corticeiro SC, Figueira E (2006) Glutathione-mediated cadmium sequestration in Rhizobium leguminosarum. Enzyme Microb Technol 39:763–769
Brauer SL, Wetterhahn KE (1991) Chromium (VI) forms thiolate complex with glutathione. J Am Chem Soc 113:3001–3007
Wu FB, Zhang GP, Peter D (2003) Four barley genotypes respond differently to cadmium:lipid peroxidation and activities of antioxidant capacity. Environ Exp Bot 50:67–77
Diwan H, Ahmad A, Iqbal M (2008) Genotypic variation in the phytoremediation potential of Indian mustard for chromium. Environ Manage 41:734–741
Acknowledgments
The project was supported by the Zhejiang Bureau of Science and Technology (2009C12050).
Author information
Authors and Affiliations
Corresponding author
Additional information
Highlights
►Exogenous application of GSH alleviated the reduction of plant growth and chlorophyll contents in Cr-stressed rice plants.
►Exogenous application of GSH reduced MDA accumulation and enhanced the antioxidant capacity in Cr-stressed rice plants.
►Exogenous application of GSH decreased Cr uptake and root-to-shoot transport in Cr-stressed rice plants.
Rights and permissions
About this article
Cite this article
Zeng, F., Qiu, B., Wu, X. et al. Glutathione-Mediated Alleviation of Chromium Toxicity in Rice Plants. Biol Trace Elem Res 148, 255–263 (2012). https://doi.org/10.1007/s12011-012-9362-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-012-9362-4