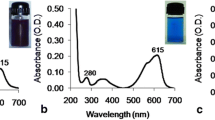
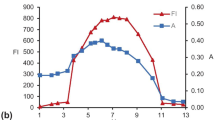
Abstract
The research in the field of food storage and preservation intensified in recent years to develop alternative preservatives and colorants to chemical ones. The algal preservatives and colorants have emerged as a viable green alternative in this context. The primary food preservatives from the algae are phycobiliproteins (PBPs) and their primary constituents: allophycocyanin (APC), phycocyanin (PC), and phycoerythrin (PE). As the foods have been subjected to the adverse environmental conditions during the production, storage, and utilization stages, it is important to research the determinants of the thermal, acidic, light, and color stability of the PBPs in these adverse environmental conditions such as light and acid exposure as well as elevated and sub-zero temperatures relevant to the food storage. Although there have been around 60 papers published in this research field, there has been no review of the research on the stability of the PBPs in these adverse environmental conditions relevant to the food storage. Hence, this paper reviews the research in this field as a first-ever population study. These studies reveal that these PBPs are stable at a wide range of acid and light exposure, and temperatures, making them perfect for the foods and beverages as a green alternative to chemical preservatives. Furthermore, a wide range of preservatives improve the stability of these PBPs through their close interaction with the PBPs and encapsulation of the PBPs is an efficient way to improve their stability, potentially using the nanotechnology. A couple of recommendations were made for the further research.
Similar content being viewed by others
References
Alizadeh-Sani, M., Mohammadian, E., Rhim, J. W., & Jafari, S. M. (2020). pH-sensitive (halochromic) smart packaging films based on natural food colorants for the monitoring of food quality. Trends in Food Science Technology, 105, 93–144. https://doi.org/10.1016/j.tifs.2020.08.014
Antelo, F. S., Costa, J. A. V., & Kalil, S. J. (2008). Thermal degradation kinetics of the phycocyanin from Spirulina platensis. Biochemical Engineering Journal, 41(1), 43–47. https://doi.org/10.1016/j.bej.2008.03.012
Bhattacharya, S., Bhayani, K., Ghosh, T., Bajaj, S., Trivedi, N., & Mishra, S. (2018). Stability of phycobiliproteins using natural preservative ε-polylysine (ε-PL). Fermentation Technology, 7, 149.
Bintsis, T., Litopoulou-Tzanetaki, E., & Robinson, R. K. (2000). Existing and potential applications of ultraviolet light in the food industry–A critical review. Journal of the Science of Food and Agriculture, 80, 637–645. https://doi.org/10.1002/(SICI)1097-0010(20000501)80:6<637::AID-JSFA603>3.0.CO;2-1 as per PUBMED
Bocker, L., Hostettler, T., Diener, M., Eder, S., Demuth, T, Adamcik, J., Reineke, K., Leeb, E., Nystrom, L., & Mathys, A. (2020). Time-temperature-resolved functional and structural changes of phycocyanin extracted from Arthrospira platensis/Spirulina. Food Chemistry, 316, 126374. https://doi.org/10.1016/j.foodchem.2020.126374
Bocker, L., Ortmann, S., Surber, J., Leeb, E., Reineke, K., & Mathys, A. (2019). Biphasic short time heat degradation of the blue microalgae protein phycocyanin from Arthrospira platensis. Innovative Food Science and Emerging Technologies, 52, 116–121. https://doi.org/10.1016/j.ifset.2018.11.007
Borch, E., Kant-Muermans, M. L., & Blixt, Y. (1996). Bacterial spoilage of meat and cured meat products. International Journal of Food Microbiology, 33(1), 103–120. https://doi.org/10.1016/0168-1605(96)01135-X
Braga, A. R. C., da Silva Figueira, F., da Silveira, J. T., de Morais, M. G., Costa, J. A. V., & Kalil, S. J. (2016). Improvement of thermal stability of C-phycocyanin by nanofiber and preservative agents. Journal of Food Processing and Preservation, 40(6), 1264–1269. https://doi.org/10.1111/jfpp.12711
Brul, S., & Coote, P. (1999). Preservative agents in foods: Mode of action and microbial resistance mechanisms. International Journal of Food Microbiology, 50, 1–17. https://doi.org/10.1016/s0168-1605(99)00072-0
Burrows, J. D. (2009). Palette of our palates: A brief history of food coloring and its regulation. Comprehensive Reviews in Food Science and Food Safety, 8(4), 394–408. https://doi.org/10.1111/j.1541-4337.2009.00089.x
Chandralekha, A., Prashanth, H. S., Tavanandi, H., & Raghavarao, K. S. M. S. (2021). A novel method for double encapsulation of C-phycocyanin using aqueous two phase systems for extension of shelf life. Journal of Food Science and Technology, 58(5), 1750–1763. https://doi.org/10.1007/s13197-020-04684-z
Chen, X., Wu, M., Yang, Q., & Wang, S. (2017). Preparation, characterization of food grade phycobiliproteins from Porphyra haitanensis and the application in liposome-meat system. LWT, 77, 468–474. https://doi.org/10.1016/j.lwt.2016.12.005
de Amarante, M. C. A., Braga, A. R. C., Sala, L., & Kalil, S. J. (2020). Colour stability and antioxidant activity of C-phycocyanin-added ice creams after in vitro digestion. Food Research International, 137, 109602. https://doi.org/10.1016/j.foodres.2020.109602
Desai, K. G. H., & Park, H. J. (2005). Recent developments in microencapsulation of food ingredients. Drying Technology, 23(7), 1361–1394. https://doi.org/10.1081/DRT-200063478
Dewi, E. N., Kurniasih, R. A., & Purnamayati, L. (2018). The application of microencapsulated phycocyanin as a blue natural colorant to the quality of jelly candy. IOP Conference Series: Earth and Environmental Science, 116, 012047. https://doi.org/10.1088/1755-1315/116/1/012047
Duangsee, R., Phoopat, N., & Ningsanond, S. (2009). Phycocyanin extraction from Spirulina platensis and extract stability under various pH and temperature. Asian Journal of Food and Agro-Industry, 2(4), 819–826.
Eghtedari, M., Porzani, S. J., & Nowruzi, B. (2021). Anticancer potential of natural peptides from terrestrial and marine environments: A review. Phytochemistry Letters, 42, 87–103. https://doi.org/10.1016/j.phytol.2021.02.008
Faieta, M., Neri, L., Di Michele, A., Di Mattia, C. D., & Pittia, P. (2021). High hydrostatic pressure treatment of Arthrospira (Spirulina) platensis extracts and the baroprotective effect of sugars on phycobiliproteins. Innovative Food Science and Emerging Technologies, 70, 102693. https://doi.org/10.1016/j.ifset.2021.102693
Faieta, M., Neri, L., Sacchetti, G., di Michele, A., & Pittia, P. (2020). Role of saccharides on thermal stability of phycocyanin in aqueous solutions. Food Research International, 132, 109093. https://doi.org/10.1016/j.foodres.2020.109093
Falkeborg, M. F., Roda-Serrat, M. C., Burnæs, K. L., & Nielsen, A. L. D. (2018). Stabilising phycocyanin by anionic micelles. Food Chemistry, 239, 771–780. https://doi.org/10.1016/j.foodchem.2017.07.007
Fan, C., Jiang, J., Yin, X., Wong, K.-H., Zheng, W., & Chen, T. (2012). Purification of selenium-containing allophycocyanin from selenium-enriched Spirulina platensis and its hepatoprotective effect against t-BOOH-induced apoptosis. Food Chemistry, 134(1), 253–261. https://doi.org/10.1016/j.foodchem.2012.02.130
Frick, D. (2003). The coloration of food. Review of Progress in Coloration and Related Topics, 33(1), 15–32. https://doi.org/10.1111/j.1478-4408.2003.tb00141.x
Galetovic, A., Seura, F., Gallardo, V., Graves, R., Cortes, J., Valdivia, C., Nunez, J., Tapia, C., Neira, I., Sanzana, S., & Gomez-Silva, S. (2020). Use of phycobiliproteins from Atacama cyanobacteria as food colorants in a dairy beverage prototype. Foods, 9(2), 244. https://doi.org/10.3390/foods9020244
Garber, L. L., Hyatt, E. M., & Starr, R. G. (2000). The effects of food color on perceived flavor. Journal of Marketing Theory and Practice, 8, 59–72. https://doi.org/10.1080/10696679.2000.11501880
Garcia, A. B., Longo, E., Murillo, M. C., & Bermejo, R. (2021). Using a B-phycoerythrin extract as a natural colorant: Application in milk-based products. Molecules, 26(2), 297. https://doi.org/10.3390/molecules26020297
Ghosh, T., & Mishra, S. (2020). Studies on extraction and stability of c-phycoerythrin from a marine cyanobacterium. Frontiers in Sustainable Food Systems, 4, 102. https://doi.org/10.3389/fsufs.2020.00102
Glazer, A. N. (1988). Phycobiliproteins. Methods in Enzymology, 167, 291–303. https://doi.org/10.1016/0076-6879(88)67034-0
Glazer, A. N., & Hixson, C. S. (1975). Characterization of R-phycocyanin. Chromophore content of R-phycocyanin and C-phycoerythrin. Journal of Biological Chemistry, 250(14), 5487–5495. https://doi.org/10.1016/S0021-9258(19)41208-8
Glazer, A. N., & Hixson, C. S. (1977). Subunit structure and chromophore composition of rhodophytan phycoerythrins. Porphyridium cruentum B-phycoerythrin and b-phycoerythrin. Journal of Biological Chemistry, 252(1), 32–42. https://doi.org/10.1016/S0021-9258(17)32794-1
Gonzalez-Ramirez, E., Andujar-Sanchez, M., Ortiz-Salmeron, E., Bacarizo, J., Cuadri, J., Mazzuca-Sobczuk, T., Ibanez, M. J., Camara-Artigas, A., & Martinez-Rodriguez, S. (2014). Thermal and pH stability of the B-phycoerythrin from the red algae Porphyridium cruentum. Food Biophysics, 9, 184–192. https://doi.org/10.1007/s11483-014-9331-x
Gram, L., Ravn, L., Rasch, M., Bruhn, J. B., Christensen, A. B., & Givskov, M. (2002). Food spoilage - Interactions between food spoilage bacteria. International Journal of Food Microbiology, 78(1–2), 79–97. https://doi.org/10.1016/S0168-1605(02)00233-7
Hadiyanto, Christwardana, M., Sutanto, H., Suzery, M., Amelia, D., & Aritonang, R. F. (2018). Kinetic study on the effects of sugar addition on the thermal degradation of phycocyanin from Spirulina. Food Bioscience, 22, 85–90. https://doi.org/10.1016/j.fbio.2018.01.007
Hadiyanto, H., Christwardana, M., Suzery, M., Sutanto, H., Nilamsari, A. M., & Yunanda, A. (2019). Effects of carrageenan and chitosan as coating materials on the thermal degradation of microencapsulated phycocyanin from Spirulina sp. International Journal of Food Engineering, 15(5–6), 20180290. https://doi.org/10.1515/ijfe-2018-0290
Hansen, T., & Coenen, L. (2015). The geography of sustainability transitions: Review, synthesis and reflections on an emergent research field. Environmental Innovation and Societal Transitions, 17, 92–109. https://doi.org/10.1016/j.eist.2014.11.001
Ilter, I., Koc, M., Demirel, Z., Dalay, M. C., & Ertekin, F. K. (2021). Improving the stability of phycocyanin by spray dried microencapsulation. Journal of Food Processing and Preservation, 45(2), e15646. https://doi.org/10.1111/jfpp.15646
Isailovic, D., Li, H. W., & Yeung, E. S. (2004). Isolation and characterization of R-phycoerythrin subunits and enzymatic digests. Journal of Chromatography A, 1051(1–2), 119–130. https://doi.org/10.1016/j.chroma.2004.07.038
Jackson, L. S., & Lee, K. (1991). Microencapsulation and the Food Industry. LWT, 24, 289–297.
Jaeschke, D. P., Teixeira, I. R., Marczak, L. D. F., & Mercali, G. D. (2021). Phycocyanin from Spirulina: A review of extraction methods and stability. Food Research International, 143, 110314. https://doi.org/10.1016/j.foodres.2021.110314
Jespersen, L., Stromdahl, L. D., Olsen, K., & Skibsted, L. H. (2005). Heat and light stability of three natural blue colorants for use in confectionery and beverages. European Food Research and Technology, 220, 261–266. https://doi.org/10.1007/s00217-004-1062-7
Kannaujiya, V. K., & Sinha, R. P. (2016). Thermokinetic stability of phycocyanin and phycoerythrin in food-grade preservatives. Journal of Applied Phycology, 28, 1063–1070. https://doi.org/10.1007/s10811-015-0638-x
Kaur, S., Khattar, J. I. S., Singh, Y., Singh, D. P., & Ahluwalia, A. S. (2019). Extraction, purification and characterisation of phycocyanin from Anabaena fertilissima PUPCCC 410.5 as a natural and food grade stable pigment. Journal of Applied Phycology, 31, 1685–1696. https://doi.org/10.1007/s10811-018-1722-9
Khandual, S., Sanchez, E. O. L., Andrews, H. E., de la Rosa, J. D. P. 2021. Phycocyanin content and nutritional profile of Arthrospira platensis from Mexico: Efficient extraction process and stability evaluation of phycocyanin. BMC Chemistry 15, 24.
Khorshidian, N., Yousefi, M., Khanniri, E., & Mortazavian, A. M. (2018). Potential application of essential oils as antimicrobial preservatives in cheese. Innovative Food Science and Emerging Technologies, 45, 62–72. https://doi.org/10.1016/j.ifset.2017.09.020
Kohata, S., Matsunaga, N., Hamabe, Y., Yumihara, K., & Sumi, T. (2010). Photo-stability of mixture of violet pigments phycoerythrin and phycocyanin extracted without separation from discolored nori seaweed. Food Science and Technology Research, 16, 617–620. https://doi.org/10.3136/fstr.16.617
Konur, O. (2020a). The scientometric analysis of the research on the algal science, technology, and medicine. In O. Konur (Ed.), Handbook of algal science, technology and medicine (pp. 3–18). Academic Press.
Konur, O. (2020b). 100 citation classics in the algal science, technology, and medicine: A scientometric analysis. In O. Konur (Ed.), Handbook of algal science, technology and medicine (pp. 19–38). Academic Press.
Konur, O. (2020c). The scientometric analysis of the research on the algal biomedicine. In O. Konur (Ed.), Handbook of algal science, technology and medicine (pp. 405–427). Academic Press.
Konur, O. (2020d). The pioneering research on the wound care by alginates. In O. Konur (Ed.), Handbook of algal science, technology and medicine (pp. 467–481). Academic Press.
Konur, O. (2020e). The scientometric analysis of the research on the algal foods. In O. Konur (Ed.), Handbook of algal science, technology and medicine (pp. 485–506). Academic Press.
Leu, J. Y., Lin, T. H., Selvamani, M. J. P., Chen, H.-C., Liang, C.-Z., & Pan, K. M. (2013). Characterization of a novel thermophilic cyanobacterial strain from Taian hot springs in Taiwan for high CO2 mitigation and C-phycocyanin extraction. Process Biochemistry, 48, 41–48. https://doi.org/10.1016/j.procbio.2012.09.019
Li, Y., Zhang, Z., & Abbaspourrad, A. (2021). Improved thermal stability of phycocyanin under acidic conditions by forming soluble complexes with polysaccharides. Food Hydrocolloids, 119, 106852. https://doi.org/10.1016/j.foodhyd.2021.106852
Liang, Y., Kaczmarek, M. B., Kasprzak, A. K., Tang, J., Shah, M. M. R., Jin, P., Smolka, A. K., Cheng, J. J., Ledakowicz, S., & Daroch, M. (2018). Thermosynechococcaceae as a source of thermostable C-phycocyanins: Properties and molecular insights. Algal Research, 35, 223–235. https://doi.org/10.1016/j.algal.2018.08.037
Martelli, G., Folli, C., Visai, L., Daglia, M., & Ferrari, D. (2014). Thermal stability improvement of blue colorant C-Phycocyanin from Spirulina platensis for food industry applications. Process Biochemistry, 49(1), 154–159. https://doi.org/10.1016/j.procbio.2013.10.008
Mazza, G., & Brouillard, R. (1987). Recent developments in the stabilization of anthocyanins in food products. Food Chemistry, 25(3), 207–225. https://doi.org/10.1016/0308-8146(87)90147-6
Mishra, S. K., Shrivastav, A., & Mishra, S. (2008). Effect of preservatives for food grade C-PC from Spirulina platensis. Process Biochemistry, 43(4), 339–345. https://doi.org/10.1016/j.procbio.2007.12.012
Mishra, S. K., Shrivastav, A., Pancha, I., Jain, D., & Mishra, S. (2010). Effect of preservatives for food grade C-phycoerythrin, isolated from marine cyanobacteria Pseudanabaena sp. International Journal of Biological Macromolecules, 47(5), 597–602. https://doi.org/10.1016/j.ijbiomac.2010.08.005
Mohammadi-Gouraji, E., Soleimanian-Zad, S., & Ghiaci, M. (2019). Phycocyanin-enriched yogurt and its antibacterial and physicochemical properties during 21 days of storage. LWT, 102, 230–236. https://doi.org/10.1016/j.lwt.2018.09.057
Moreira, I. D. O., Passos, T. S., Chiapinni, C., Silveira, G. K., Souza, J. C. M., Coca-Vellarde, L. G., Deliza, R., & de Lima Araujo, K. G. (2012). Colour evaluation of a phycobiliprotein-rich extract obtained from Nostoc PCC9205 in acidic solutions and yogurt. Journal of the Science of Food and Agriculture, 92(3), 598–605. https://doi.org/10.1002/jsfa.4614
Munawaroh, H. S. H., Darojatun, K., Gumilar, G. G., Aisyah, S., & Wulandari, A. P. (2018). Characterization of phycocyanin from Spirulina fusiformis and its thermal stability. Journal of Physics: Conference Series, 1013, 012205. https://doi.org/10.1088/1742-6596/1013/1/012205
Munier, M., Jubeau, S., Wijaya, A., Morancais, M., Dumay, J., Marchal, L., Jaouen, P., & Fleurence, J. (2014). Physicochemical factors affecting the stability of two pigments: R-phycoerythrin of Grateloupia turuturu and B-phycoerythrin of Porphyridium cruentum. Food Chemistry, 150, 400–407. https://doi.org/10.1016/j.foodchem.2013.10.113
Nouri, E., Abbasi, H., & Rahimi, E. (2018). Effects of processing on stability of water- and fat-soluble vitamins, pigments (C-phycocyanin, carotenoids, chlorophylls) and colour characteristics of Spirulina platensis. Quality Assurance and Safety of Crops and Foods, 10(4), 335–349. https://doi.org/10.3920/QAS2018.1304
Nowruzi, B., & Blanco, S. (2019). In silico identification and evolutionary analysis of candidate genes involved in the biosynthesis methylproline genes in cyanobacteria strains of Iran. Phytochemistry Letters, 29, 199–211. https://doi.org/10.1016/j.phytol.2018.12.011
Nowruzi, B., & Porzani, S. J. (2021). Toxic compounds produced by cyanobacteria belonging to several species of the order Nostocales: A review. Journal of Applied Toxicology, 41(4), 510–548. https://doi.org/10.1002/jat.4088
Orta-Ramirez, A., Merrill, J. E., & Smith, D. M. (2000). Affects the thermal inactivation parameters of R-phycoerythrin from Porphyra yezoensis. Journal of Food Science, 65(6), 1046–1050. https://doi.org/10.1111/j.1365-2621.2000.tb09415.x
Orta-Ramirez, A., Merrill, J. E., & Smith, D. M. (2001). Sucrose, sodium dodecyl sulfate, urea, and 2-mercaptoethanol affect the thermal inactivation of R-phycoerythrin. Journal of Food Protection, 64(11), 1806–1811. https://doi.org/10.4315/0362-028x-64.11.1806
Pan-Utai, W., & Iamtham, S. (2020). Enhanced microencapsulation of C-phycocyanin from Arthrospira by freeze-drying with different wall materials. Food Technology and Biotechnology, 58(4), 423–432. https://doi.org/10.17113/ftb.58.04.20.6622
Patel, A. S., Lakshmibalasubramaniam, S., & Nayak, B. (2020). Steric stabilization of phycobiliprotein loaded liposome through polyethylene glycol adsorbed cellulose nanocrystals and their impact on the gastrointestinal tract. Food Hydrocolloids, 98, 10525. https://doi.org/10.1016/j.foodhyd.2019.105252
Patel, A., Mishra, S., Pawar, R., & Ghosh, P. K. (2005). Purification and characterization of C-phycocyanin from cyanobacterial species of marine and freshwater habitat. Protein Expression and Purification, 40(2), 248–255. https://doi.org/10.1016/j.pep.2004.10.028
Patel, A., Pawar, R., Mishra, S., Sonawane, S., & Ghosh, P. K. (2004). Kinetic studies on thermal denaturation of C-phycocyanin. Indian Journal of Biochemistry & Biophysics, 41, 254–257.
Pereira, T., Barroso, S., Mendes, S., & Gil, M. M. (2020). Stability, kinetics, and application study of phycobiliprotein pigments extracted from red algae Gracilaria gracilis. Journal of Food Science, 85(10), 3400–3405. https://doi.org/10.1111/1750-3841.15422
Porzani, S. J., Konur, O., & Nowruzi, B. (2021). Cyanobacterial natural products as sources for antiviral drug discovery against COVID-19. Journal of Biomolecular Structure and Dynamics. https://doi.org/10.1080/07391102.2021.1899050
Pradeep, H. N., & Nayak, C. A. (2019). Enhanced stability of C-phycocyanin colorant by extrusion encapsulation. Journal of Food Science and Technology, 56, 4526–4534. https://doi.org/10.1007/s13197-019-03955-8
Pumas, C., Peerapornpisal, Y., Vacharapiyasophon, P., Leelapornpisid, P., Boonchum, W., Ishii, M., & Khanongnuch, C. (2012). Purification and characterization of a thermostable phycoerythrin from hot spring cyanobacterium Leptolyngbya sp. KC45. International Journal of Agriculture and Biology, 14(1), 121–125.
Rahman, D. Y., Sarian, F. D., van Wijk, A., Martinez-Garcia, M., & van der Maarel, M. J. E. C. (2017). Thermostable phycocyanin from the red microalga Cyanidioschyzon merolae, a new natural blue food colorant. Journal of Applied Phycology, 29, 1233–1239. https://doi.org/10.1007/s10811-016-1007-0
Rajabpour, N., Nowruzi, B., & Ghobeh, M. (2019). Investigation of the toxicity, antioxidant and antimicrobial activities of some cyanobacterial strains isolated from different habitats. Acta Biologica Slovenica, 62(2), 3–14.
Rastogi, R. P., Sonani, R. R., & Madamwar, D. (2015). Physico-chemical factors affecting the in vitro stability of phycobiliproteins from Phormidium rubidum A09DM. Bioresource Technology, 190, 219–226. https://doi.org/10.1016/j.biortech.2015.04.090
Sasaki, Y. F., Kawaguchi, S., Kamaya, A., Ohshita, M., Kabasawa, K., Iwama, K., Taniguchi, K., & Tsuda, J. (2002). The comet assay with 8 mouse organs: Results with 39 currently used food additives. Mutation Research - Genetic Toxicology and Environmental Mutagenesis, 519(1–2), 103–119. https://doi.org/10.1016/s1383-5718(02)00128-6
Schmatz, D. A., da Silveira Mastrantonio, D. J., Costa, J. A. V., & de Morais, M. G. (2020). Encapsulation of phycocyanin by electrospraying: A promising approach for the protection of sensitive compounds. Food and Bioproducts Processing, 119, 206–215. https://doi.org/10.1016/j.fbp.2019.07.008
Selig, M. J., Malchione, N. M., Gamaleldin, S., Padilla-Zakour, O. I., & Abbaspourrad, A. (2018). Protection of blue color in a spirulina derived phycocyanin extract from proteolytic and thermal degradation via complexation with beet-pectin. Food Hydrocolloids, 74, 46–52. https://doi.org/10.1016/j.foodhyd.2017.07.027
Smith-Palmer, A., Stewart, J., & Fyfe, L. (2001). The potential application of plant essential oils as natural food preservatives in soft cheese. Food Microbiology, 18(4), 463–470. https://doi.org/10.1006/fmic.2001.0415
Tripathi, S. N., Kapoor, S., & Shrivastava, A. (2007). Extraction and purification of an unusual phycoerythrin in a terrestrial desiccation tolerant cyanobacterium Lyngbya arboricola. Journal of Applied Phycology, 19, 441–447. https://doi.org/10.1007/s10811-006-9151-6
Wu, H.-L., Wang, G. H., Xiang, W. Z., Li, T., & He, H. (2016). Stability and antioxidant activity of food-grade phycocyanin isolated from Spirulina platensis. International Journal of Food Properties, 19(10), 2349–2362. https://doi.org/10.1080/10942912.2015.1038564
Yagoubi, A. S., Shahidi, F., Mohebbi, M., Varidi, M., & Golmohammadzadeh, S. (2018). Preparation, characterization and evaluation of physicochemical properties of phycocyanin-loaded solid lipid nanoparticles and nanostructured lipid carriers. Journal of Food Measurement and Characterization, 12, 378–385. https://doi.org/10.1007/s11694-017-9650-y
Yan, M., Liu, B., Jiao, X., & Qin, S. (2014). Preparation of phycocyanin microcapsules and its properties. Food and Bioproducts Processing, 92(1), 89–97. https://doi.org/10.1016/j.fbp.2013.07.008
Yang, R., Ma, T., Shi, L., Wang, Q., Zhang, L., Zhang, F., Wang, Z., & Zhou, Z. (2022). The formation of phycocyanin-EGCG complex for improving the color protection stability exposing to light. Food Chemistry, 370, 130985. https://doi.org/10.1016/j.foodchem.2021.130985
Zhang, S., Zhang, Z., Dadmohammadi, Y., Li, Y., Jaiswal, A., & Abbaspourrad, A. (2021a). Whey protein improves the stability of C-phycocyanin in acidified conditions during light storage. Food Chemistry, 344, 128642. https://doi.org/10.1016/j.foodchem.2020.128642
Zhang, Z., Cho, S., Dadmohammadi, Y., Li, Y., & Abbaspourrad, A. (2021b). Improvement of the storage stability of C-phycocyanin in beverages by high-pressure processing. Food Hydrocolloids, 110, 106055. https://doi.org/10.1016/j.foodhyd.2020.106055
Zhang, Z., Li, Y., & Abbaspourrad, A. (2020). Improvement of the colloidal stability of phycocyanin in acidified conditions using whey protein-phycocyanin interactions. Food Hydrocolloids, 105, 105747. https://doi.org/10.1016/j.foodhyd.2020.105747
Acknowledgements
We are grateful for the comments provided by four anonymous reviewers and the guidance provided by the editor.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix: The Keyword set for the PBPs
Appendix: The Keyword set for the PBPs
( TITLE ( phycobiliprotein* OR “c-allophycocyanin*” OR *phycocyanin OR *phycoerythrin OR phycoerythrobilin* OR phycocyanobilin* OR phycobiliviolin* OR phycourobilin* OR phycocyanorubin* OR apophycocyanin* OR allophycocyanin* OR “c-phycoerythrin*” OR biliprotein* OR phycobilisome* OR “c-phycoyanin” OR phycobilin* OR phycoerythrocyanin* OR phycoviolobilin*) OR SRCTITLE ( phycobiliprotein* OR “c-allophycocyanin*” OR phycocyanin* OR phycoerythrin* OR phycoerythrobilin* OR phycocyanobilin* OR phycobiliviolin* OR phycourobilin* OR phycocyanorubin* OR apophycocyanin* OR allophycocyanin* OR “c-phycoerythrin*” OR biliprotein* OR phycobilisome* OR “c-phycoyanin” OR phycobilin* OR phycoerythrocyanin* OR phycoviolobilin*)) AND ( TITLE ( food* OR nutrit* OR nutraceut*) OR SRCTITLE ( food* OR nutrit* OR nutraceut*)) AND ( LIMIT-TO ( DOCTYPE, “ar”) OR LIMIT-TO ( DOCTYPE, “re”) OR LIMIT-TO ( DOCTYPE, “le”) OR LIMIT-TO ( DOCTYPE, “ch”) OR LIMIT-TO ( DOCTYPE, “no”) OR LIMIT-TO ( DOCTYPE, “sh”) OR LIMIT-TO ( DOCTYPE, “bk”)) AND ( LIMIT-TO ( LANGUAGE, “English”)) AND ( LIMIT-TO ( SRCTYPE, “j”) OR LIMIT-TO ( SRCTYPE, “k”) OR LIMIT-TO ( SRCTYPE, “b”)).
Rights and permissions
About this article
Cite this article
Nowruzi, B., Konur, O. & Anvar, S.A.A. The Stability of the Phycobiliproteins in the Adverse Environmental Conditions Relevant to the Food Storage. Food Bioprocess Technol 15, 2646–2663 (2022). https://doi.org/10.1007/s11947-022-02855-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-022-02855-8