Abstract
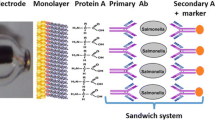
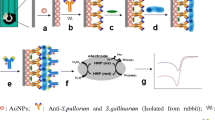
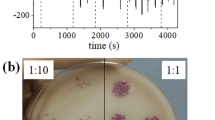
This paper reports the application of an amperometric biosensor for rapid and specific Salmonella Typhimurium detection in milk. This device was developed from self-assembled monolayer technique on a gold screen-printed electrode, using cysteamine thiol. Polyclonal antibodies were oriented by protein A immobilization. The biosensor structure was characterized by cyclic voltammetry, Fourier transform infrared spectroscopy, and scanning electron microscopy. The analytical response was obtained by a chronoamperometry technique, using a direct-sandwich peroxidase-labeled system. The biosensor device showed a qualitative behavior with a very low limit of detection of 10 CFU mL−1 and a detection time of 125 min. The biosensor specificity was demonstrated in pure and mixed samples with strains of Escherichia coli and Citrobacter freundii. The performance of the biosensor was found satisfactory, and the device was tested in skimmed and whole milk samples, being able to detect S. Typhimurium quickly, without an enrichment step. This structure of immunosensor assembly can be expended in future studies for other food matrices and bacterial species, making it a useful tool to ensure food safety.






Similar content being viewed by others
References
Afonso, A., Perez-Lopez, B., Faria, R. C., Mattoso, L., Hernandez-Herrero, M., Roig-Sagues, A., Maltez-Da Costa, M., & Merkoci, A. (2013). Electrochemical detection of Salmonella using gold nanoparticles. Biosensors and Bioelectronics, 40(1), 121–126. https://doi.org/10.1016/j.bios.2012.06.054.
Alexandre, D. L., Melo, A. M. A., Furtado, R. F., Borges, M. F., Figueiredo, E. A. T., Biswas, A., Cheng, H. N., & Alves, C. R. (2016). Amperometric biosensor for Salmonella Typhimurium: detection in milk. XXV Congresso Brasileiro de Ciência e Tecnologia de Alimentos. X CIGR Section VI International Technical Symposium. Gramado, RS, Brazil October, 2016. http://www.ufgrs.br/sbctars-eventos/xxvcbcta/anais. Accessed 25 Sep 2017.
Amaro, M., Oaew, S., & Surareungchaia, W. (2012). Scano-magneto immunoassay based on carbon nanotubes/gold nanoparticles nanocomposite for Salmonella enterica serovar Typhimurium detection. Biosensors and Bioelectronics, 38(1), 157–162. https://doi.org/10.1016/j.bios.2012.05.018.
Anandan, V., Gangadharan, R., & Zhang, G. (2009). Role of SAM chain length in enhancing the sensitivity of nanopillar modified electrodes for glucose detection. Sensors, 9(1), 1295–1305. https://doi.org/10.3390/s90301295.
Andrews, W. H., Wang, H., Jacobson, A., & Hammack, T. (2016). Food and Drug Administration. Bacteriological Analytical Manual (BAM). Chapter 5 Salmonella. http://www.fda.gov/Food/FoodScienceResearch/LaboratoryMethods/ucm070149htm. Accessed 16 Sep 2017.
Arora, P., Sindhu, A., Kaur, H., Dilbaghi, N., & Chaudhury, A. (2013). An overview of transducers as platform for the rapid detection of foodborne pathogens. Applied Microbiology and Biotechnology, 97(1), 1829–1840. https://doi.org/10.1007/s00253-013-4692-5.
Avrameas, S. (1969). Coupling of enzymes to proteins with glutaraldehyde. Use of the conjugates for the detection of antigens and antibodies. Immunochemistry, 6(1), 43–49. https://doi.org/10.1016/0019-2791(69)90177-3.
Barth, A. (2007). Infrared spectroscopy of proteins. Biochimica et Biophysica Acta, 1767(9), 1073–1101. https://doi.org/10.1016/j.bbabio.2007.06.004.
Björk, I., Petersson, B. A., & Sjöquist, J. (1972). Some physicochemical properties of protein A from Staphylococcus aureus. European Journal of Biochemistry, 29(3), 579–584. https://doi.org/10.1111/j.1432-1033.1972.tb02024.x.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72(1-2), 248–254. https://doi.org/10.1016/0003-2697(76)90527-3.
Branco, R. J. F., Dias, A. M. G. C., & Roque, A. C. A. (2012). Understanding the molecular recognition between antibody fragments and protein A biomimetic ligand. Journal of Chromatography A, 1244, 106–115. https://doi.org/10.1016/j.chroma.2012.04.071.
Brandão, D., Liébana, S., Alegret, S., Pividori, P., Campoy, S., & Cortés, M. I. (2013). Electrochemical magneto-immunosensing of Salmonella based on nano and micro-sized magnetic particles. Journal of Physics: Conference Series, 421, 012020. https://doi.org/10.1088/1742-6596/421/1/012020.
Brazil. (2014). Ministério da Saúde/Secretaria de Vigilância em Saúde. Dados Epidemiológicos: DTA período de 2000 a 2014. http://portal.saude.gov.br/ portal/ arquivos/ pdf/dados_dta_periodo_2000_2014_site. pdf. Accessed 16 September 2017.
Cao, Y., Sun, X., Guo, Y., Zhao, W., & Wang, X. (2015). An electrochemical immunosensor based on interdigitated array microelectrode for the detection of chlorpyrifos. Bioprocess and Biosystems Engineering, 38(2), 307–313. https://doi.org/10.1007/s00449-014-1269-3.
Centers for Disease Control and Prevention. (2012). Pathogens causing US foodborne illnesses, hospitalizations, and deaths, 2000–2008 http://www.cdc.gov/foodborneburden/PDFs/pathogens-complete-list-01-12.pdf. Accessed 10 Dec 2016.
Delibato, E., Volpe, G., Stangalini, D., DeMedici, D., Moscone, D., & Palleschi, G. (2006). Development of SYBR-green real-time PCR and a multichannel electrochemical immunosensor for specific detection of Salmonella enterica. Analytical Letters, 39(8), 1611–1625. https://doi.org/10.1080/00032710600713354.
European Food Safety Agency. (2014). EFSA explains zoonotic diseases—Salmonella. http://www.efsa.europa.eu/en/search/doc/factsheetsalmonella.pdf. Accessed 20 Dec 2016.
Food and Drug Administration. (2012a). Bad bug book, foodborne pathogenic microorganisms and natural toxins. Second edition. [Salmonella species, pp. 9–13].
Food and Drug Administration. (2012b). The danger of raw milk. http://www.fda.gov/food/resourcesforyou/consumers/ucm079516.htm. Accessed 20 Sep 2017.
Freitas, M., Viswanathan, S., Nouws, H., Oliveira, M., & Delerue-Matos, C. (2014). Iron oxide/ gold core/shell nanomagnetic probes and CdS biolabels for amplified electrochemical immunosensing of Salmonella typhimurium. Biosensors and Bioelectronics, 51, 195–200. https://doi.org/10.1016/j.bios.2013.07.048.
Gil, E. S., & Mello, G. R. (2010). Electrochemical biosensors in pharmaceutical analysis. Brazilian Journal of Pharmaceutical Sciences, 46(3), 375–391. https://doi.org/10.1590/S1984-82502010000300002.
Gooding, J. J., & Darwish, N. (2012). The rise of self- assembled monolayers for fabricating electrochemical biosensors—an interfacial perspective. Chemical Record, 12(1), 92–105. https://doi.org/10.1002/tcr.201100013.
Green, A. A., & Hughs, W. L. (1995). Methods in enzymology, v. 1. New York: Academic Press.
Hu, C. M., Dou, W., & Zhao, G. (2014). Enzyme immunosensor based on gold nanoparticles electroposition and Streptavidin-biotin system for detection of S. pullorum & S. gallinarum. Electrochimica Acta, 117, 239–245. https://doi.org/10.1016/j.electacta.2013.11.132.
Knirel, Y. A., Kocharova, N. A., Bystrova, O. V., Katzenellenbogen, E., & Gamian, A. (2002). Structures and serology of the O-specific polysaccharides of bacteria of the genus Citrobacter. Archivum Immunologiae et Therapiae Experimentalis, 50(6), 379–391.
Kokkinos, C., Economou, A., & Prodromidis, M. (2016). Electrochemical immunosensors: critical survey of different architectures and transduction strategies. Trends in Analytical Chemistry, 79, 88–105. https://doi.org/10.1016/j.trac.2015.11.020.
Kong, J., & Yu, S. (2007). Fourier transform infrared spectroscopic analysis of protein secondary structures. Acta Biochimica et Biophysica Sinica, 39(8), 549–559. https://doi.org/10.1111/j.1745-7270.2007.00320.x.
Lee, K. M., Runyon, M., Herrman, T. J., Hsieh, J., & Phillips, R. (2015). Review of Salmonella detection and identification methods: aspects of rapid emergency response and food safety. Food Control, 47, 264–276. https://doi.org/10.1016/j.foodcont.2014.07.011.
Lermo, A., Fabiano, S., Hernández, S., Galve, R., Marco, M. P., Alegret, S., & Pividori, M. I. (2009). Immunoassay for folic acid detection in vitamin-fortified milk based on electrochemical magneto sensors. Biosensors and Bioelectronics, 24(7), 2057–2063. https://doi.org/10.1016/j.bios.2008.10.020.
Liébana, S., Lermo, A., Campoy, S., Cortés, M. P., Alegret, S., & Pividori, M. I. (2009). Rapid detection of Salmonella in milk by electrochemical magneto-immunosensing. Biosensors and Bioelectronics, 25(2), 510–513. https://doi.org/10.1016/j.bios.2009.07.022.
Melo, A., Alexandre, D., Furtado, R., Borges, M., Figueiredo, E. A., Biswas, A., Cheng, H. N., & Alves, C. (2016). Electrochemical immunosensors for Salmonella detection in food. Applied Microbiology and Biotechnology, 100(12), 5301–5312. https://doi.org/10.1007/s00253-016-7548-y.
Mendes, R. K., Carvalhal, R. F., & Kubota, L. T. (2008). Effects of different self-assembled monolayers on enzyme immobilization procedures in peroxidase-based biosensor development. Journal of Electroanalytical Chemistry, 612(2), 164–172. https://doi.org/10.1016/j.jelechem.2007.09.033.
Mortari, A., & Lorenzelli, L. (2014). Recent sensing technologies for pathogen detection in milk: a review. Biosensors & Bioelectronics, 60, 8–21. https://doi.org/10.1016/j.bios.2014.03.063.
Painter, J. A., Hoekstra, R. M., Ayers, T., Tauxe, R. V., Braden, C. R., & Angulo, F. J. (2013). Attribution of foodborne illnesses, hospitalizations, and deaths to food commodities by using outbreak data, United States, 1998–2008. Emerging Infection Diseases, 19(3), 407–415. https://doi.org/10.3201/eid1903.111866.
Péterfi, Z., Kustos, I., Kilár, F., & Kocsis, B. (2007). Microfluidic chip analysis of outer membrane proteins responsible for serological cross-reaction between three Gram-negative bacteria: Proteus morganii O34, Escherichia coli O111 and Salmonella Adelaide O35. Journal of Chromatography A, 1155(1), 214–217. https://doi.org/10.1016/j.chroma.2007.02.093.
Pimenta-Martins, M. G. R., Furtado, R. F., Heneine, L. G. D., Dias, R. S., Borges, M. D., & Alves, C. R. (2012). Development of an amperometric immunosensor for detection of staphylococcal enterotoxin type A in cheese. Journal of Microbiological Methods, 91(1), 138–143. https://doi.org/10.1016/j.mimet.2012.05.016.
Ricci, F., Adornetto, G., & Palleschi, G. (2012). A review of experimental aspects of electrochemical immunosensors. Electrochimica Acta, 84, 74–83. https://doi.org/10.1016/j.electacta.2012.06.033.
Salam, F., & Tothill, I. E. (2009). Detection of Salmonella typhimurium using an electrochemical immunosensor. Biosensensors & Bioelectronics, 24(8), 2630–2636. https://doi.org/10.1016/j.bios.2009.01.025.
Salam, F., Uludag, Y., & Tothill, I. E. (2013). Real-time and sensitive detection of Salmonella Typhimurium using an automated quartz crystal microbalance (QCM) instrument with nanoparticles amplification. Talanta, 115, 761–767. https://doi.org/10.1016/j.talanta.2013.06.034.
Skaládal, P. (2016). Piezoelectric biosensors. Trends in Analytical Chemistry, 79, 127–133. https://doi.org/10.1016/j.trac.2015.12.009.
Sun, J., Xia, S., Bian, C., & Qu, L. (2009). Micro amperometric immunosensor for the detection of Salmonella Typhimurium. Proceedings of SPIE - The International Society for Optical Engineering, 7159(1), 71590A-1-71590A-6.
Tlili, A., Abdelghani, A., Hleli, S., & Maaref, M. A. (2004). Electrical characterization of a thiol SAM on gold as a first step for the fabrication of immunosensors based on a quartz crystal microbalance. Sensors, 4(6–7), 105–114. https://doi.org/10.3390/s40670105.
Trakhtenberg, S., Hangun-Balkir, Y., Warner, J. C., Bruno, F. F., Kumar, J., Nagarajan, R., & Samuelson, L. A. (2005). Photo-cross-linked immobilization of polyelectrolytes for enzymatic construction of conductive nanocomposites. Journal of the American Chemical Society, 127(25), 9100–9104. https://doi.org/10.1021/ja042438v.
United States Department of Agriculture. (2016). Foodborne Pathogen Test Kits Validated by Independent Organizations, https://www.fsis.usda.gov/wps/wcm/connect/f97532f4-9c28-4ecc-9aee-0e1e6cde1a89/Validated-Test-Kit-Spreadsheet.pdf?MOD=AJPERES. Accessed 10 Dec 2016.
Velusamy, V., Arshak, K., Korostynska, O., Oliwa, K., & Adley, C. (2010). An overview of foodborne pathogen detection: in the perspective of biosensors. Biotechnology Advances, 28(2), 232–254. https://doi.org/10.1016/j.biotechadv.2009.12.004.
Wang, Y., & Duncan, T. V. (2017). Nanoscale sensors for assuring the safety of food products. Current Opinion in Biotechnology, 44(1), 74–86. https://doi.org/10.1016/j.copbio.2016.10.005.
Xu, S. (2012). Electromechanical biosensors for pathogen detection. Microchimica Acta, 178(1), 245–260. https://doi.org/10.1007/s00604-012-0831-4.
Zhu, D., Yan, Y., Lei, P., Shen, B., Cheng, W., Ju, H., & Ding, S. J. (2014). A novel electrochemical sensing strategy for rapid and ultrasensitive detection of Salmonella by rolling circle amplification and DNA-AuNPs probe. Analytica Chimica Acta, 846, 44–50. https://doi.org/10.1016/j.aca.2014.07.024.
Acknowledgements
The authors thank the Analytical Center of the Federal University of Ceará for the SEM analysis. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Funding
The authors are grateful to CNPq (Award numbers: 475174/2012-7 and 405506/2013-9), CAPES, and Embrapa agencies in Brazil for their financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of Interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Alexandre, D.L., Melo, A.M.A., Furtado, R.F. et al. A Rapid and Specific Biosensor for Salmonella Typhimurium Detection in Milk. Food Bioprocess Technol 11, 748–756 (2018). https://doi.org/10.1007/s11947-017-2051-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-017-2051-8