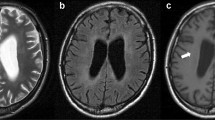
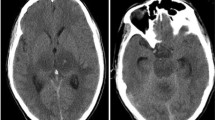
Opinion statement
The first critical step in the appropriate treatment of neurological infectious disease accompanying immunosuppressive states or immunomodulatory medication is to properly identify the offending organism. Broadly immunosuppressive conditions will predispose to both common and uncommon infectious diseases. There are substantial differences between neurological infectious disorders complicating disturbances of the innate immunity (neutrophils, monocytes and macrophages) and those due to abnormal adaptive immunity (humoral and cellular immunity). Similarly, there are differences in the types of infections with impaired humoral immunity compared to disturbed cellular immunity and between T- and B-cell disorders. HIV/AIDS has been a model of acquired immunosuppression and the nature of opportunistic infections with which it has been associated has been well characterized and generally correlates well with the degree of CD4 lymphopenia. Increasingly, immunotherapies target specific components of the immune system, such as an adhesion molecule or its ligand or surface receptors on a special class of cells. These targeted perturbations of the immune system increase the risk of particular infectious diseases. For instance, natalizumab, an α4β1 integrin inhibitor that is highly effective in multiple sclerosis, increases the risk of progressive multifocal leukoencephalopathy for reasons that still remain unclear. It is likely that other therapies that result in a disruption of a specific component of the immune system will be associated with other unique opportunistic infections. The risk of multiple simultaneous neurological infections in the immunosuppressed host must always be considered, particularly with a failure to respond to a therapeutic regimen. With respect to appropriate and effective therapy, diagnostic accuracy assumes primacy, but occasionally broad spectrum therapy is necessitated. For a number of opportunistic infectious disorders, particularly some viral and fungal diseases, antimicrobial therapy remains inadequate.
Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance
Berger JR. Progressive multifocal leukoencephalopathy and newer biological agents. Drug Saf. 2010;33(11):969–83. This article addresses the risk of PML with the newer biological agents and describe potential mechanisms by which these therapies may predispose to the development of this disorder.
Fox R. Advances in the management of PML: focus on natalizumab. Cleve Clin J Med. 2011;78 Suppl 2:S33–7.
Khatri BO, Man S, Giovannoni G, Koo AP, Lee JC, Tucky B, et al. Effect of plasma exchange in accelerating natalizumab clearance and restoring leukocyte function. Neurology. 2009;72(5):402–9.
BiogenIdec (2011) Tysabri Update. https://medinfo.biogenidec.com. Accessed January 7, 2012.
Clifford DB, De Luca A, Simpson DM, Arendt G, Giovannoni G, Nath A. Natalizumab-associated progressive multifocal leukoencephalopathy in patients with multiple sclerosis: lessons from 28 cases. Lancet Neurol. 2010;9(4):438–46. This is an excellent description of the first cases of natalizumab-associated PML focusing on the clinical and radiographic manifestations and demonstrating a high rate of concomitant IRIS
Tan IL, McArthur JC, Clifford DB, Major EO, Nath A. Immune reconstitution inflammatory syndrome in natalizumab-associated PML. Neurology. 2011;77(11):1061–7.
Hartung HP, Berger JR, Wiendl H, Meier U, Stangel M, Aktas O, et al. Therapie der Multiplen Sklerose mit monokonalen Antikorpen: aktualisierte Empfelungen im Rahmen eines Expertmeetings. Actuelle Neurologie. 2011;38:2–11.
Hou J, Major EO. The efficacy of nucleoside analogs against JC virus multiplication in a persistently infected human fetal brain cell line. J Neurovirol. 1998;4(4):451–6.
Kerr DA, Chang CF, Gordon J, Bjornsti MA, Khalili K. Inhibition of human neurotropic virus (JCV) DNA replication in glial cells by camptothecin. Virology. 1993;196(2):612–8.
Brickelmaier M, Lugovskoy A, Kartikeyan R, Reviriego-Mendoza MM, Allaire N, Simon K, et al. Identification and characterization of mefloquine efficacy against JC virus in vitro. Antimicrob Agents Chemother. 2009;53(5):1840–9.
Andrei G, Snoeck R, Vandeputte M, De Clercq E. Activities of various compounds against murine and primate polyomaviruses. Antimicrob Agents Chemother. 1997;41(3):587–93.
Hall CD, Dafni U, Simpson D, Clifford D, Wetherill PE, Cohen B, et al. Failure of cytarabine in progressive multifocal leukoencephalopathy associated with human immunodeficiency virus infection. AIDS clinical trials group 243 team. N Engl J Med. 1998;338(19):1345–51.
Gasnault J, Kousignian P, Kahraman M, Rahoiljaon J, Matheron S, Delfraissy JF, et al. Cidofovir in AIDS-associated progressive multifocal leukoencephalopathy: a monocenter observational study with clinical and JC virus load monitoring. J Neurovirol. 2001;7(4):375–81.
Marra CM, Rajicic N, Barker DE, Cohen BA, Clifford D, Donovan Post MJ, et al. A pilot study of cidofovir for progressive multifocal leukoencephalopathy in AIDS. AIDS. 2002;16(13):1791–7.
Mori Y, Miyamoto T, Nagafuji K, Kamezaki K, Yamamoto A, Saito N, et al. High incidence of human herpes virus 6-associated encephalitis/myelitis following a second unrelated cord blood transplantation. Biol Blood Marrow Transplant. 2010;16(11):1596–602.
Razonable RR. Infections due to human herpesvirus 6 in solid organ transplant recipients. Curr Opin Organ Transplant. epub before print.
Baldwin K. Ganciclovir-resistant human herpesvirus-6 encephalitis in a liver transplant patient: a case report. J Neurovirol. 2011;17(2):193–5.
Takahashi Y, Yamada M, Nakamura J, Tsukazaki T, Padilla J, Kitamura T, et al. Transmission of human herpesvirus 7 through multigenerational families in the same household. Pediatr Infect Dis J. 1997;16(10):975–8.
Erice A, Tierney C, Hirsch M, Caliendo AM, Weinberg A, Kendall MA, et al. Cytomegalovirus (CMV) and human immunodeficiency virus (HIV) burden, CMV end-organ disease, and survival in subjects with advanced HIV infection (AIDS clinical trials group protocol 360). Clin Infect Dis. 2003;37(4):567–78.
Clarke LM, Duerr A, Feldman J, Sierra MF, Daidone BJ, Landesman SH. Factors associated with cytomegalovirus infection among human immunodeficiency virus type 1-seronegative and -seropositive women from an urban minority community. J Infect Dis. 1996;173(1):77–82.
Silva CA, Oliveira AC, Vilas-Boas L, Fink MC, Pannuti CS, Vidal JE. Neurologic cytomegalovirus complications in patients with AIDS: retrospective review of 13 cases and review of the literature. Rev Inst Med Trop Sao Paulo. 2011;52(6):305–10. This is an excellent review of CMV infection in the setting of HIV/AIDS.
Reddy SM, Winston DJ, Territo MC, Schiller GJ. CMV central nervous system disease in stem-cell transplant recipients: an increasing complication of drug-resistant CMV infection and protracted immunodeficiency. Bone Marrow Transplant. 2010;45(6):979–84.
Akpek G, Mikulski M, Kleinberg M, Badros A, Yanovich S, Rapoport AP. Cellular therapy with sequential unmanipulated donor lymphocyte infusions in drug-resistant cytomegalovirus (CMV) encephalitis. Blood. 2011;117(21):5772–4.
Feuchtinger T, Opherk K, Bethge WA, Topp MS, Schuster FR, Weissinger EM, et al. Adoptive transfer of pp 65-specific T cells for the treatment of chemorefractory cytomegalovirus disease or reactivation after haploidentical and matched unrelated stem cell transplantation. Blood. 2010;116(20):4360–7.
Grover D, Newsholme W, Brink N, Manji H, Miller R. Herpes simplex virus infection of the central nervous system in human immunodeficiency virus-type 1-infected patients. Int J STD AIDS. 2004;15(9):597–600.
Solomon T, Michael BD, Smith PE, Sanderson F, Davies NW, Hart IJ, et al. National guideline for the management of suspected viral encephalitis in adults. J Infect. epub before print.
Dennett C, Klapper PE, Cleator GM. Polymerase chain reaction in the investigation of “relapse” following herpes simplex encephalitis. J Med Virol. 1996;48(2):129–32.
NIAID Collaborative Antiviral Study Group. Long term treatment of herpes simplex encephalitis (HSE) with Valtrex [cited 2012 January 12].
Kamei S, Sekizawa T, Shiota H, Mizutani T, Itoyama Y, Takasu T, et al. Evaluation of combination therapy using aciclovir and corticosteroid in adult patients with herpes simplex virus encephalitis. J Neurol Neurosurg Psychiatry. 2005;76(11):1544–9.
Martinez-Torres F, Menon S, Pritsch M, Victor N, Jenetzky E, Jensen K, et al. Protocol for German trial of acyclovir and corticosteroids in herpes-simplex-virus-encephalitis (GACHE): a multicenter, multinational, randomized, double-blind, placebo-controlled German, Austrian and Dutch trial [ISRCTN45122933]. BMC Neurol. 2008;8:40.
De Broucker T, Mailles A, Chabrier S, Morand P and Stahl JP. Acute varicella zoster encephalitis without evidence of primary vasculopathy in a case-series of 20 patients. Clin Microbiol Infect 2011. doi:10.111/j.1469.2011.03705.x.
Schweikert A, Kremer M, Ringel F, Liebig T, Duyster J, Stuve O, et al. Primary central nervous system lymphoma in a patient treated with natalizumab. Ann Neurol. 2009;66(3):403–6.
Finelli PF, Naik K, DiGiuseppe JA, Prasad A. Primary lymphoma of CNS, mycophenolate mofetil and lupus. Lupus. 2006;15(12):886–8.
Svobodova B, Hruskova Z, Rysava R and Tesar V. Brain diffuse large B-cell lymphoma in a systemic lupus erythematosus patient treated with immunosuppressive agents including mycophenolate mofetil. Lupus. 2011;20(13):1452–4.
Gonzalez-Aguilar A, Soto-Hernandez JL. The management of primary central nervous system lymphoma related to AIDS in the HAART era. Curr Opin Oncol. 2011;23(6):648–53.
Carrabba MG, Reni M, Foppoli M, Chiara A, Franzin A, Politi LS, et al. Treatment approaches for primary CNS lymphomas. Expert Opin Pharmacother. 2010;11(8):1263–76.
Batchelor TT, Grossman SA, Mikkelsen T, Ye X, Desideri S, Lesser GJ. Rituximab monotherapy for patients with recurrent primary CNS lymphoma. Neurology. 2011;76(10):929–30.
Lai GG, Koo YX, Tao M, Tan TT, Lim ST. Use of rituximab in combination with high-dose methotrexate in the treatment of primary central nervous system lymphoma in a mycophenolate mofetil treated patient with lupus nephritis. Acta Oncol. 2010;50(1):144–5.
Algazi AP, Kadoch C, Rubenstein JL. Biology and treatment of primary central nervous system lymphoma. Neurotherapeutics. 2009;6(3):587–97.
Chaudhuri A, Martinez-Martin P, Kennedy PG, Andrew Seaton R, Portegies P, Bojar M, et al. EFNS guideline on the management of community-acquired bacterial meningitis: report of an EFNS task force on acute bacterial meningitis in older children and adults. Eur J Neurol. 2008;15(7):649–59.
Mitja O, Pigrau C, Ruiz I, Vidal X, Almirante B, Planes AM, et al. Predictors of mortality and impact of aminoglycosides on outcome in listeriosis in a retrospective cohort study. J Antimicrob Chemother. 2009;64(2):416–23.
Matano S, Satoh S, Harada Y, Nagata H and Sugimoto T. Antibiotic treatment for bacterial meningitis caused by Listeria monocytogenes in a patient with multiple myeloma. J Infect Chemother. 2011;16(2):123–5.
Singh JA, Wells GA, Christensen R, Tanjong E, Maxwell GL, Macdonald JK, et al. Adverse effects of biologics: a network meta-analysis and Cochrane overview. Cochrane Database Syst Rev. 2011;2:CD008794. This article is a valuable review of the complications observed with biological agents.
Garg RK. Tuberculous meningitis. Acta Neurol Scand. 2010;122(2):75–90.
Girgis NI, Farid Z, Kilpatrick ME, Sultan Y, Mikhail IA. Dexamethasone adjunctive treatment for tuberculous meningitis. Pediatr Infect Dis J. 1991;10(3):179–83.
Schoeman JF, Van Zyl LE, Laubscher JA, Donald PR. Effect of corticosteroids on intracranial pressure, computed tomographic findings, and clinical outcome in young children with tuberculous meningitis. Pediatrics. 1997;99(2):226–31.
Berger JR. Immunodeficiency states. In: Noseworthy J, editor. Neurological therapeutics principles and practice. Oxon: Informa Healthcare; 2006. p. 1540–57.
Disclosure
A. Nath: none. J.R Berger is a consultant for Millennium (Data Safety Monitoring Board), Biogen, Novartis, Bayer, Genzyme, Eisai, Genentech, and Amgen (Data Safety Monitoring Board); has received a grant from the PML Consortium; an honorarium from PRIME; and speaking fees from Novartis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nath, A., Berger, J.R. Complications of Immunosuppressive/Immunomodulatory Therapy in Neurological Diseases. Curr Treat Options Neurol 14, 241–255 (2012). https://doi.org/10.1007/s11940-012-0172-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11940-012-0172-y