Abstract
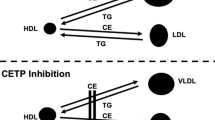
By virtue of their effects on low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, and cellular cholesterol efflux, there is considerable interest in the potential use of pharmacological inhibitors of cholesteryl ester transfer protein (CETP) as a novel approach for cardiovascular disease prevention. This is supported by observations from genetic and animal studies suggesting that less CETP activity has favorable cardiovascular effects. Despite the adverse effects of the first CETP inhibitor to move forward in clinical development, torcetrapib, there remains considerable interest in developing alternative CETP inhibitors without the off-target effects of torcetrapib. The clinical development programs leading to a number of promising CETP inhibitors will be reviewed.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Baigent C, Blackwell L, Emberson J, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376:1670–81.
Libby P. The forgotten majority: unfinished business in cardiovascular risk reduction. J Am Coll Cardiol. 2005;46:1225–8.
Barter PJ, Chapman MJ, Hennekens CH, et al. Cholesteryl ester transfer protein: a novel target for raising HDL and inhibiting atherosclerosis. Arterioscler Thromb Vasc Biol. 2003;23:160–7.
Brown ML, Inazu A, Hesler CB, et al. Molecular basis of lipid transfer protein deficiency in a family with increased high-density lipoproteins. Nature. 1989;342:448–51.
Inazu A, Brown ML, Hesler CB, et al. Increased high-density lipoprotein levels caused by a common cholesteryl-ester transfer protein gene mutation. N Engl J Med. 1990;323:1234–8.
Vasan RS, Pencina MJ, Robins SJ, et al. Association of circulating cholesteryl ester transfer protein activity with incidence of cardiovascular disease in the community. Circulation. 2009;120:2414–20.
Ritsch A, Scharnagl H, Eller P, et al. Cholesteryl ester transfer protein and mortality in patients undergoing coronary angiography: the Ludwigshafen Risk and Cardiovascular Health study. Circulation. 2010;121:366–74.
Boekholdt SM, Kuivenhoven JA, Wareham NJ, et al. Plasma levels of cholesteryl ester transfer protein and the risk of future coronary artery disease in apparently healthy men and women: the prospective EPIC (European Prospective Investigation into Cancer and nutrition)-Norfolk population study. Circulation. 2004;110:1418–23.
Marschang P, Sandhofer A, Ritsch A, et al. Plasma cholesteryl ester transfer protein concentrations predict cardiovascular events in patients with coronary artery disease treated with pravastatin. J Intern Med. 2006;260:151–9.
Khera AV, Wolfe ML, Cannon CP, et al. On-statin cholesteryl ester transfer protein mass and risk of recurrent coronary events (from the pravastatin or atorvastatin evaluation and infection therapy—thrombolysis in myocardial infarction 22 [PROVE IT-TIMI 22] study). Am J Cardiol. 2010;106:451–6.
Duwensee K, Breitling LP, Tancevski I, et al. Cholesteryl ester transfer protein in patients with coronary heart disease. Eur J Clin Investig. 2010;40:616–22.
Robins SJ, Lyass A, Brocia RW, et al. Plasma lipid transfer proteins and cardiovascular disease. the Framingham Heart Study. Atherosclerosis. 2013;228:230–6.
Kappelle PJ, Perton F, Hillege HL, et al. High plasma cholesteryl ester transfer but not CETP mass predicts incident cardiovascular disease: a nested case-control study. Atherosclerosis. 2011;217:249–52.
Zeller M, Masson D, Farnier M, et al. High serum cholesteryl ester transfer rates and small high-density lipoproteins are associated with young age in patients with acute myocardial infarction. J Am Coll Cardiol. 2007;50:1948–55.
Thompson A, Di Angelantonio E, Sarwar N, et al. Association of cholesteryl ester transfer protein genotypes with CETP mass and activity, lipid levels, and coronary risk. Jama. 2008;299:2777–88.
Johannsen TH, Frikke-Schmidt R, Schou J, et al. Genetic inhibition of CETP, ischemic vascular disease and mortality, and possible adverse effects. J Am Coll Cardiol. 2012;60:2041–8.
Voight BF, Peloso GM, Orho-Melander M, et al. Plasma HDL cholesterol and risk of myocardial infarction: a Mendelian randomisation study. Lancet. 2012;380:572–80. Mendelian randomization analysis suggesting that CETP plays a causative role in atherosclerotic cardiovascular disease.
Marotti KR, Castle CK, Boyle TP, et al. Severe atherosclerosis in transgenic mice expressing simian cholesteryl ester transfer protein. Nature. 1993;364:73–5.
Plump AS, Masucci-Magoulas L, Bruce C, et al. Increased atherosclerosis in ApoE and LDL receptor gene knock-out mice as a result of human cholesteryl ester transfer protein transgene expression. Arterioscler Thromb Vasc Biol. 1999;19:1105–10.
Westerterp M, van der Hoogt CC, de Haan W, et al. Cholesteryl ester transfer protein decreases high-density lipoprotein and severely aggravates atherosclerosis in APOE*3-Leiden mice. Arterioscler Thromb Vasc Biol. 2006;26:2552–9.
Herrera VL, Makrides SC, Xie HX, et al. Spontaneous combined hyperlipidemia, coronary heart disease and decreased survival in Dahl salt-sensitive hypertensive rats transgenic for human cholesteryl ester transfer protein. Nat Med. 1999;5:1383–9.
MacLean PS, Bower JF, Vadlamudi S, et al. Cholesteryl ester transfer protein expression prevents diet-induced atherosclerotic lesions in male db/db mice. Arterioscler Thromb Vasc Biol. 2003;23:1412–5.
Foger B, Chase M, Amar MJ, et al. Cholesteryl ester transfer protein corrects dysfunctional high density lipoproteins and reduces aortic atherosclerosis in lecithin cholesterol acyltransferase transgenic mice. J Biol Chem. 1999;274:36912–20.
Cazita PM, Berti JA, Aoki C, et al. Cholesteryl ester transfer protein expression attenuates atherosclerosis in ovariectomized mice. J Lipid Res. 2003;44:33–40.
Casquero AC, Berti JA, Salerno AG, et al. Atherosclerosis is enhanced by testosterone deficiency and attenuated by CETP expression in transgenic mice. J Lipid Res. 2006;47:1526–34.
Hayek T, Masucci-Magoulas L, Jiang X, et al. Decreased early atherosclerotic lesions in hypertriglyceridemic mice expressing cholesteryl ester transfer protein transgene. J Clin Invest. 1995;96:2071–4.
Hildebrand RB, Lammers B, Meurs I, et al. Restoration of high-density lipoprotein levels by cholesteryl ester transfer protein expression in scavenger receptor class B type I (SR-BI) knockout mice does not normalize pathologies associated with SR-BI deficiency. Arterioscler Thromb Vasc Biol. 2010;30:1439–45.
Rittershaus CW, Miller DP, Thomas LJ, et al. Vaccine-induced antibodies inhibit CETP activity in vivo and reduce aortic lesions in a rabbit model of atherosclerosis. Arterioscler Thromb Vasc Biol. 2000;20:2106–12.
Sugano M, Makino N, Sawada S, et al. Effect of antisense oligonucleotides against cholesteryl ester transfer protein on the development of atherosclerosis in cholesterol-fed rabbits. J Biol Chem. 1998;273:5033–6.
Okamoto H, Yonemori F, Wakitani K, et al. A cholesteryl ester transfer protein inhibitor attenuates atherosclerosis in rabbits. Nature. 2000;406:203–7.
Morehouse LA, Sugarman ED, Bourassa PA, et al. Inhibition of CETP activity by torcetrapib reduces susceptibility to diet-induced atherosclerosis in New Zealand White rabbits. J Lipid Res. 2007;48:1263–72.
Brousseau ME, Schaefer EJ, Wolfe ML, et al. Effects of an inhibitor of cholesteryl ester transfer protein on HDL cholesterol. N Engl J Med. 2004;350:1505–15.
Barter PJ, Caulfield M, Eriksson M, et al. Effects of torcetrapib in patients at high risk for coronary events. N Engl J Med. 2007;357:2109–22.
Nissen SE, Tardif JC, Nicholls SJ, et al. Effect of torcetrapib on the progression of coronary atherosclerosis. N Engl J Med. 2007;356:1304–16.
Bots ML, Visseren FL, Evans GW, et al. Torcetrapib and carotid intima-media thickness in mixed dyslipidaemia (RADIANCE 2 study): a randomised, double-blind trial. Lancet. 2007;370:153–60.
Kastelein JJ, van Leuven SI, Burgess L, et al. Effect of torcetrapib on carotid atherosclerosis in familial hypercholesterolemia. N Engl J Med. 2007;356:1620–30.
Nicholls SJ, Tuzcu EM, Brennan DM, et al. Cholesteryl ester transfer protein inhibition, high-density lipoprotein raising, and progression of coronary atherosclerosis: insights from ILLUSTRATE (Investigation of Lipid Level Management Using Coronary Ultrasound to Assess Reduction of Atherosclerosis by CETP Inhibition and HDL Elevation). Circulation. 2008;118:2506–14.
Barter P. Lessons learned from the Investigation of Lipid Level Management to Understand its Impact in Atherosclerotic Events (ILLUMINATE) trial. Am J Cardiol. 2009;104:10E–5E.
Tall AR, Yvan-Charvet L, Wang N. The failure of torcetrapib: was it the molecule or the mechanism? Arterioscler Thromb Vasc Biol. 2007;27:257–60.
Vergeer M, Stroes ES. The pharmacology and off-target effects of some cholesterol ester transfer protein inhibitors. Am J Cardiol. 2009;104:32E–8E.
Barter PJ, Rye KA, Tardif JC, et al. Effect of torcetrapib on glucose, insulin, and hemoglobin A1c in subjects in the Investigation of Lipid Level Management to Understand its Impact in Atherosclerotic Events (ILLUMINATE) trial. Circulation. 2011;124:555–62. Potential anti-diabetic effects of torcetrapib.
Ray KK, Ditmarsch M, Kallend D, et al. The effect of cholesteryl ester transfer protein inhibition on lipids, lipoproteins, and markers of HDL function after an acute coronary syndrome: the dal-ACUTE randomized trial. Eur Heart J. 2014;35:1792–800.
Luscher TF, Taddei S, Kaski JC, et al. Vascular effects and safety of dalcetrapib in patients with or at risk of coronary heart disease: the dal-VESSEL randomized clinical trial. Eur Heart J. 2012;33:857–65.
Fayad ZA, Mani V, Woodward M, et al. Safety and efficacy of dalcetrapib on atherosclerotic disease using novel non-invasive multimodality imaging (dal-PLAQUE): a randomised clinical trial. Lancet. 2011;378:1547–59.
Schwartz GG, Olsson AG, Abt M, et al. Effects of dalcetrapib in patients with a recent acute coronary syndrome. N Engl J Med. 2012;367:2089–99.
Besler C, Heinrich K, Rohrer L, et al. Mechanisms underlying adverse effects of HDL on eNOS-activating pathways in patients with coronary artery disease. J Clin Invest. 2011;121:2693–708.
Tardif JC, Rheaume E, Lemieux Perreault LP. Pharmacogenomic determinants of the cardiovascular effects of dalcetrapib. circulation. Cardiovasc Genet. 2015.
Cannon CP, Shah S, Dansky HM, et al. Safety of anacetrapib in patients with or at high risk for coronary heart disease. N Engl J Med. 2010;363:2406–15. Preliminary lipid and clinical outcome effects of anacetrapib in the DEFINE study.
Gotto Jr AM, Cannon CP, Li XS, et al. Evaluation of lipids, drug concentration, and safety parameters following cessation of treatment with the cholesteryl ester transfer protein inhibitor anacetrapib in patients with or at high risk for coronary heart disease. Am J Cardiol. 2014;113:76–83.
Nicholls SJ, Brewer HB, Kastelein JJ, et al. Effects of the CETP inhibitor evacetrapib administered as monotherapy or in combination with statins on HDL and LDL cholesterol: a randomized controlled trial. JAMA. 2011;306:2099–109. Primary lipid effects of torcetrapib.
Nicholls SJ, Ruotolo G, Brewer HB, et al. Cholesterol efflux capacity and pre-beta-1 HDL concentrations are increased in dyslipidemic patients treated with evacetrapib. J Am Coll Cardiol. 2015;66:2201–10.
Nicholls SJ, Lincoff AM, Barter PJ, et al. Assessment of the clinical effects of cholesteryl ester transfer protein inhibition with evacetrapib in patients at high-risk for vascular outcomes: rationale and design of the ACCELERATE trial. Am Heart J. 2015;170:1061–9.
Hovingh GK, Kastelein JJ, van Deventer SJ. Cholesterol ester transfer protein inhibition by TA-8995 in patients with mild dyslipidaemia (TULIP): a randomised, double-blind, placebo-controlled phase 2 trial. Lancet. 2015;386:452–60. Primary lipid effects of TA-8995.
Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387–97.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Stephen J. Nicholls reports receiving research support from AstraZeneca, Amgen, Eli Lilly, Novartis, Resverlogix, Sanofi-Regeneron, Anthera, and Cerenis and is a consultant for AstraZeneca, Amgen, Boehringer Ingelheim, CSL Behring, Eli Lilly, Merck, Takeda, Roche, Novartis, and Sanofi-Regeneron.
Kohei Takata reports grants from the Clinical Research Promotion Foundation.
Belinda Di Bartolo and MyNgan Duong declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Lipid Abnormalities and Cardiovascular Prevention
Rights and permissions
About this article
Cite this article
Di Bartolo, B., Takata, K., Duong, M. et al. CETP Inhibition in CVD Prevention: an Actual Appraisal. Curr Cardiol Rep 18, 43 (2016). https://doi.org/10.1007/s11886-016-0724-y
Published:
DOI: https://doi.org/10.1007/s11886-016-0724-y