Abstract
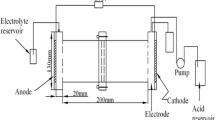
As a new technology used for the cleaning of chromium-contaminated soil, worldwide interest in eletrokinetic (EK) remediation has grown considerably in recent times. However, owing to the fact that chromium exists as both cationic and anionic species in the soil, it is not an efficient method. This paper reports upon a study in which a process using approaching anodes (AAs) was used to enhance the removal efficiency of chromium by eletrokinetics. Two bench-scale experiments to remove chromium from contaminated soil were performed, one using a fixed anode (FA) and the other using AAs. In the AAs experiment, the anode moved toward the cathode by 7 cm every three days. After remediation, soil pH, total chromium, and fractionation of chromium in the soil were determined. The average removal efficiency of total chromium was 11.32% and 18.96% in the FA and AAs experiments, respectively. After remediation, acidic soil conditions throughout the soil were generated through the use of AAs, while 80% of the soil remained neutral or alkalic when using the FA approach. The acidic soil environment and high field intensity in the AAs experiment might have favored chromium desorption, dissolution and dissociation from the soil, plus the mobility of chromium in the soil was also enhanced. The results demonstrate that AAs used in the process of EK remediation can enhance the efficiency of chromium removal from soil.
Similar content being viewed by others
References
Page M M, Page C L. Electroremediation of contaminated soils. Journal of Environmental Engineering, 2002, 128(3): 208–219
Acar Y B, Alshawabkeh A N. Principles of electrokinetic remediation. Environmental Science & Technology, 1993, 27(13): 2638–2647
Probstein R F, Hicks R E. Removal of contaminants from soils by electric fields. Science, 1993, 260(5107): 498–503
Reddy K R, Parupudi U S. Removal of chromium, nickel and cadmium from clays by in-situ electrokinetic remediation. Journal of Soil Contamination, 1997, 6(4): 391–407
Reddy K R, Chinthamreddy S. Electrokinetic remediation of heavy metal-contaminated soils under reducing environments. Waste Management (New York, N.Y.), 1999, 19(4): 269–282
Kotas J, Stasicka Z. Chromium occurrence in the environment and methods of its speciation. Environmental Pollution, 2000, 107(3): 263–283
Reddy K R, Chinthamreddy S. Effects of initial form of chromium on electrokinetic remediation in clays. Advances in Environmental Research, 2003, 7(2): 353–365
Weng C H, Yuan C. Removal of Cr(III) from clay soils by electrokinetics. Environmental Geochemistry and Health, 2001, 23 (3): 281–285
Gent D B, Bricka R M, Alshawabkeh A N, Larson S L, Fabian G, Granade S. Bench- and field-scale evaluation of chromium and cadmium extraction by electrokinetics. Journal of Hazardous Materials, 2004, 110(1–3): 53–62
Reddy K R, Chinthamreddy S. Enhanced electrokinetic remediation of heavy metals in glacial till soils using different electrolyte solutions. Journal of Environmental Engineering, 2004, 130(4): 442–455
Zhou D M, Alshawabkeh A N, Deng C F, Cang L, Si Y B. Electrokinetic removal of chromium and copper from contaminated soils by lactic acid enhancement in the catholyte. Journal of Environmental Sciences (China), 2004, 16(4): 529–532
Hansen H K, Ottosen LM, Kliem B K, Villumsen A. Electrodialytic remediation of soils polluted with Cu, Cr, Hg, Pb and Zn. Journal of Chemical Technology and Biotechnology (Oxford, Oxfordshire), 1997, 70(1): 67–73
Reddy K R, Chinthamreddy S. ASCE M, Chinthamreddy S. Sequentially enhanced electrokinetic remediation of heavy metals in low buffering clayey soils. Journal of Geotechnical and Geoenvironmental Engineering, 2003, 129(3): 263–277
Li Z M, Yu J W, Neretnieks I. Removal of Pb(II), Cd(II) and Cr(III) from sand by electromigration. Journal of Hazardous Materials, 1997, 55(1–3): 295–304
USEPA. SW-846 Method 3060A Alkaline digestion for hexavalent chromium, Revision 1, 1996
Rauret G, López-Sánchez J F, Sahuquillo A, Rubio R, Davidson C, Ure A, Quevauviller P. Improvement of the BCR three step sequential extraction procedure prior to the certification of new sediment and soil reference materials. Journal of Environmental Monitoring, 1999, 1(1): 57–61
Shen Z M, Chen X J, Jia J P, Qu L, Wang W H. Comparison of electrokinetic soil remediation methods using one fixed anode and approaching anodes. Environmental Pollution, 2007, 150(2): 193–199
Kimbrough D E, Cohen Y, Winer A M, Creelman L, Mabuni C. A critical assessment of chromium in the environment. Critical Reviews in Environmental Science and Technology, 1999, 29(1): 1–46
Yang J, Guo R, Chen S, Li L. Interaction between Cr(VI) and a Ferich soil in the presence of oxalic and tartaric acids. Environmental Geology, 2008, 53(7): 1529–1533
Reddy K R, Xu C Y, Chinthamreddy S. Assessment of electrokinetic removal of heavy metals from soils by sequential extraction analysis. Journal of Hazardous Materials, 2001, 84(2–3): 279–296
Ando Y, Tanaka T. Proposal for a new system for simultaneous production of hydrogen and hydrogen peroxide by water electrolysis. International Journal of Hydrogen Energy, 2004, 29(13): 1349–1354
Choi J H, Maruthamuthu S, Lee H G, Ha T H, Bae J H. Electrochemical studies on the performance of SS316L electrode in electrokinetics. Metals and Materials International, 2009, 15(5): 771–781
Nieto Castillo A, Soriano J, García Delgado R. Changes in chromium distribution during the electrodialytic remediation of a Cr(VI)-contaminated soil. Environmental Geochemistry and Health, 2008, 30(2): 153–157
Yuan C, Weng C H. Electrokinetic enhancement removal of heavy metals from industrial wastewater sludge. Chemosphere, 2006, 65 (1): 88–96
Yin J, Ma X, Sun H. Study on electrokinetic remediation of soil contaminated by chromium. Journal of Environmental Engineering, 2008, 2(5): 684–689 (in Chinese)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, S., Li, T., Li, G. et al. Enhanced electrokinetic remediation of chromium-contaminated soil using approaching anodes. Front. Environ. Sci. Eng. 6, 869–874 (2012). https://doi.org/10.1007/s11783-012-0437-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11783-012-0437-4