Abstract
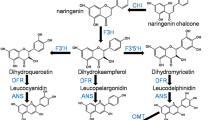
Eggplant is rich in anthocyanins, which are one of the important secondary metabolites and beneficial to human health. In this study, four anthocyanin biosynthesis genes, including chalcone synthase (SmCHS), chalcone isomerase (SmCHI), flavanone 3-hydroxylase (SmF3H) and dihydroflavonol 4-reductase (SmDFR), were isolated from eggplant. Their expression profiles were investigated along with other two structural genes (SmF3′5′H and SmANS) in different tissues, bagging, and low-temperature treatments. The highest expression levels were observed in peels except for SmF3H which was detected in stems, and SmF3H was also the exclusive gene that did not show correlation with anthocyanin content. Unlike purple peels of the control, the bagged fruits displayed white peels and had no anthocyanin accumulation, because of the expression of SmCHI, SmF3′5′H, SmDFR, and SmANS totally depended on light. As low temperature stimulates anthocyanin accumulation, all the six anthocyanin biosynthesis genes were up-regulated in cold stress experiment, and SmCHS increased most obviously. Moreover, early biosynthesis genes (SmCHS, SmCHI, and SmF3H) responded earlier than late biosynthesis genes (SmF3′5′H, SmDFR, and SmANS) under low temperature. Subcellular localization suggested that the enzymes encoded by the genes cloned in this study were all located in cytosol and nucleus. To further characterize the function, their ectopic expression in Arabidopsis was performed, and overexpression lines displayed higher anthocyanin accumulation in stems and siliques. The present study provides insight into anthocyanin biosynthesis in eggplant and may facilitate genetic engineering for improvement of the anthocyanin content in plants.







Similar content being viewed by others
References
Ahmed NU, Park JI, Jung HJ, Yang TJ, Hur Y, Nou I (2014) Characterization of dihydroflavonol 4-reductase (DFR) genes and their association with cold and freezing stress in Brassica rapa. Gene 550:46–55
Azuma A, Yakushiji H, Koshita Y, Kobayashi S (2012) Flavonoid biosynthesis-related genes in grape skin are differentially regulated by temperature and light conditions. Planta 236:1067–1080
Buer CS, Muday GK (2004) The transparent testa4 mutation prevents flavonoid synthesis and alters auxin transport and the response of Arabidopsis roots to gravity and light. Plant Cell 16:1191–1205
Cao G, Sofic E, Prior RL (1996) Antioxidant capacity of tea and common vegetables. J Agric Food Chem 44:3426–3431
Cericola F, Portis E, Lanteri S, Toppino L, Barchi L, Acciarri N, Pulcini L, Sala T, Rotino GL (2014) Linkage disequilibrium and genome-wide association analysis for anthocyanin pigmentation and fruit color in eggplant. BMC Genom 15:896
Chiu L, Li L (2012) Characterization of the regulatory network of BoMYB2 in controlling anthocyanin biosynthesis in purple cauliflower. Planta 236:1153–1164
Chu H, Jeong JC, Kim WJ, Chung DM, Jeon HK, Ahn YO, Kim SH, Lee HS, Kwak SS, Kim CY (2013) Expression of the sweetpotato R2R3-type IbMYB1a gene induces anthocyanin accumulation in Arabidopsis. Physiol Plant 148:189–199
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Dare AP, Tomes S, Jones M, McGhie TK, Stevenson DE, Johnson RA, Greenwood DR, Hellens RP (2013) Phenotypic changes associated with RNA interference silencing of chalcone synthase in apple (Malus × domestica). Plant J 74:398–410
Davies KM, Albert NW, Schwinn KE (2012) From landing lights to mimicry: the molecular regulation of flower colouration and mechanisms for pigmentation patterning. Funct Plant Biol 39:619–638
Dela G, Or E, Ovadia R, Nissim-Levi A, Weiss D, Oren-Shamir M (2003) Changes in anthocyanin concentration and composition in ‘Jaguar’ rose flowers due to transient high-temperature conditions. Plant Sci 164:333–340
Feng F, Li M, Ma F, Cheng L (2013) Phenylpropanoid metabolites and expression of key genes involved in anthocyanin biosynthesis in the shaded peel of apple fruit in response to sun exposure. Plant Physiol Biochem 69:54–61
Feng FJ, Li MJ, Ma FW, Cheng LL (2014) The effects of bagging and debagging on external fruit quality, metabolites, and the expression of anthocyanin biosynthetic genes in ‘Jonagold’ apple (Malus domestica Borkh.). Sci Hortic 165:123–131
Ferrer JL, Jez JM, Bowman ME, Dixon RA, Noel JP (1999) Structure of chalcone synthase and the molecular basis of plant polyketide biosynthesis. Nat Struct Mol Bio 6:775–784
Huang X, Ouyang XH, Yang PY, Lau OS, Li G, Li JG, Chen HD, Deng XW (2012a) Arabidopsis FHY3 and HY5 positively mediate induction of COP1 transcription in response to photomorphogenic UV-B light. Plant Cell 24:4590–4606
Huang Y, Gou JQ, Jia ZC, Yang L, Sun YM, Xiao XY, Song F, Luo K (2012b) Molecular cloning and characterization of two genes encoding dihydroflavonol-4-reductase from populus trichocarpa. PLoS One 7:e30364
Jez JM, Bowman ME, Dixon RA, Noel JP (2000) Structure and mechanism of the evolutionarily unique plant enzyme chalcone isomerase. Nat Struct Biol 7:786–791
Jiang F, Wang JY, Jia HF, Jia WS, Wang HQ, Xiao M (2013) RNAi-mediated silencing of the flavanone 3-hydroxylase gene and its effect on flavonoid biosynthesis in strawberry fruit. J Plant Growth Regul 32:182–190
Johnson ET, Ryu S, Yi H, Shin B, Cheong H, Choi G (2001) Alteration of a single amino acid changes the substrate specificity of dihydroflavonol 4-reductase. Plant J 25:325–333
Kadomura-Ishikawa Y, Miyawaki K, Takahash A, Masuda T, Noji S (2015) Light and abscisic acid independently regulated FaMYB10 in Fragaria × ananassa fruit. Planta 241:953–965
Koduri PH, Gordon GS, Barker EI, Colpitts CC, Ashton NW, Suh DY (2010) Genome-wide analysis of the chalcone synthase superfamily genes of Physcomitrella patens. Plant Mol Biol 72:247–263
Lazze MC, Pizzala R, Perucca P, Cazzalini O, Savio M, Forti L, Vannini V, Bianchi L (2006) Anthocyanidins decrease endothelin-1 production and increase endothelial nitric oxide synthase in human endothelial cells. Mol Nutr Food Res 50:44–51
Leyva A, Jarillo JA, Salinas J, Martinez-Zapater JM (1995) Low temperature induces the accumulation of phenylalanine ammonialyase and chalcone synthase mRNAs of Arabidopsis thaliana in a light-dependent manner. Plant Physiol 108:39–46
Li HH, Qiu J, Chen FD, Lv XF, Fu CX, Zhao DX, Hua XJ, Zhao Q (2012) Molecular characterization and expression analysis of dihydroflavonol 4-reductase (DFR) gene in Saussurea medusa. Mol Biol Rep 39:2991–2999
Li X, Ma H, Huang H, Li D, Yao S (2013) Natural anthocyanins from phytoresources and their chemical researches. Nat Prod Res 27:456–469
Li SJ, Bai YC, Li CL, Yao HP, Chen H, Zhao HX, Wu Q (2015) Anthocyanins accumulate in tartary buckwheat (Fagopyrum tataricum) sprout in response to cold stress. Acta Physiol Plant. doi:10.1007/s11738-015-1913-9
Liu HP, Fu DQ, Zhu BZ, Yan HX, Shen XY, Zuo JH, Zhu Y, Luo YB (2012) Virus-induced gene silencing in Eggplant (Solanum melongena). J Integr Plant Biol 54:422–429
Liu ML, Li XR, Liu YB, Cao B (2013a) Regulation of flavanone 3-hydroxylase gene involved in the flavonoid biosynthesis pathway in response to UV-B radiation and drought stress in the desert plant, Reaumuria soongorica. Plant Physiol Biochem 73:161–167
Liu XF, Yin XR, Allan AC, Wang KL, Shi YN, Huang YJ, Ferguson IB, Xu CJ, Chen KS (2013b) The role of MrbHLH1 and MrMYB1 in regulating anthocyanin biosynthesis genes in tobacco and Chinese bayberry (Myrica rubra) during anthocyanin biosynthesis. Plant Cell Tissue Org 115:285–298
Liu SH, Ju JF, Xia GG (2014) Identification of the flavonoid 3′-hydroxylase and flavonoid 3′,5′-hydroxylase genes from Antarctic moss and their regulation during abiotic stress. Gene 543:145–152
Liu T, Song S, Yuan YB, Wu DJ, Chen MJ, Sun QN, Zhang B, Xu CJ, Chen KS (2015) Improved peach peel color development by fruit bagging. Enhanced expression of anthocyanin biosynthetic and regulatory genes using white non-woven polypropylene as replacement for yellow paper. Sci Hortic 184:142–148
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Lo Piero AR, Puglisi I, Rapisarda P, Petrone G (2005) Anthocyanins accumulation and related gene expression in red orange fruit induced by low temperature storage. J Agric Food Chem 53:9083–9088
Lukacin R, Britsch L (1997) Identification of strictly conserved histidine and arginine residues as part of the active site in Petunia hybrida flavanone 3ß-hydroxylase. Eur J Biochem 249:748–757
Man YP, Wang YC, Li ZZ, Jiang ZW, Yang HL, Gong JJ, He SS, Wu SQ, Yang ZQ, Zheng J, Wang ZY (2015) High-temperature inhibition of biosynthesis and transportation of anthocyanins results in the poor red coloration in red-fleshed Actinidia chinensis. Physiol Plant 153:565–583
Martens S, Teeri T, Forkmann G (2002) Heterologous expression of dihydroflavonol 4-reductase from various plants. FEBS Lett 531:453–458
Matsumoto H, Nakamura Y, Tachibanaki S, Kawamura S, Hirayama M (2003) Stimulatory effect of cyanidin 3-glycosides on the regeneration of rhodopsin. J Agric Food Chem 51:3560–3563
Mori K, Goto-Yamamoto N, Kitayama M, Hashizume K (2007) Loss of anthocyanins in red-wine grape under high temperature. J Exp Bot 58:1935–1945
Muir S, Collins G, Robinson S, Hughes S, Bovy A, De Vos C, van Tunen A, Verhoeyen M (2001) Overexpression of petunia chalcone isomerase in tomato results in fruit containing increased levels of flavonols. Nat Biotechnol 19:470–474
Niu SS, Xu CJ, Zhang WS, Zhang B, Li X, Lin-Wang K, Ferguson IB, Allan AC, Chen KS (2010) Coordinated regulation of anthocyanin biosynthesis in Chinese bayberry (Myrica rubra) fruit by a R2R3 MYB transcription factor. Planta 231:887–899
Petroni K, Tonelli C (2011) Recent advances on the regulation of anthocyanin synthesis in reproductive organs. Plant Sci 181:219–229
Petroni K, Pilu R, Tonelli C (2014) Anthocyanins in corn: a wealth of genes for human health. Planta 240:901–911
Polster J, Dithmar H, Burgemeister R, Friedemann G, Feucht W (2006) Flavonoids in plant nuclei: detection by laser microdissection and pressure catapulting (LMPC), in vivo staining, and UV-visible spectroscopic titration. Physiol Plant 128:163–174
Ren LJ, Hu AL, Li YL, Zhang B, Zhang YJ, Tu Y, Chen GP (2014) Heterologous expression of BoPAP1 in tomato induces stamen specific anthocyanin accumulation and enhances tolerance to a long-term low temperature stress. J Plant Growth Regul 33:757–768
Romero I, Sanchez-Ballesta MT, Maldonado R, Escribano MI, Merodio C (2008) Anthocyanin, antioxidant activity and stress-induced gene expression in high CO2-treated table grapes stored at low temperature. Plant Physiol 165:522–530
Rowan DD, Cao M, Lin-Wang K, Cooney JM, Jensen DJ, Austin PT, Hunt MB, Norling CR, Hellens P, Schaffer RJ, Allan AC (2009) Environmental regulation of leaf colour in red 35S:PAP1 Arabidopsis thaliana. New Phytol 182:102–115
Sarma AD, Sharma R (1999) Anthocyanin-DNA copigmentation complex: mutual protection against oxidative damage. Phytochemistry 52:1313–1318
Saslowsky DE, Warek U, Winkel BSJ (2005) Nuclear localization of flavonoid enzymes in Arabidopsis. J Biol Chem 280:23735–23740
Shimada N, Aoki T, Sato S, Nakamura Y, Tabata S, Ayabe S (2003) A cluster of genes encodes the two types of chalcone isomerase involved in the biosynthesis of general flavonoids and legume-specific 5-deoxy(iso)flavonoids in Lotus japonicus. Plant Physiol 131:941–951
Shvarts M, Borochov A, Weiss D (1997) Low temperature enhances petunia flower pigmentation and induces chalcone synthase gene expression. Physiol Plant 99:67–72
Sparkes IA, Runions J, Kearns A, Hawes C (2006) Rapid, transient expression of fluorescent fusion proteins in tobacco plants and generation of stably transformed plants. Nat Protoc 1:2019–2025
Sun CD, Zheng YX, Tang XL, Jiang M, Zhang JK, Li X, Chen KS (2012) Purification and anti-tumor activity of cyanidin-3-O-glucoside from Chinese bayberry fruit. Food Chem 13:1287–1294
Sun W, Meng XY, Liang LJ, Jiang WS, Huang YF, He J, Hu HY, Almqvist J, Gao X, Wang L (2015) Molecular and biochemical analysis of chalcone synthase from Freesia hybrid in flavonoid biosynthesis pathway. PLoS One 10:e0119054
Tai DQ, Tian J, Zhang J, Song TT, Yao YC (2014) A Malus crabapple chalcone synthase gene, McCHS, regulates red petal color and flavonoid biosynthesis. PLoS One 9:e110570
Takos AM, Jaffe FW, Jacob SR, Bogs J, Robinson SP, Walker AR (2006) Light-induced expression of a MYB gene regulates anthocyanin biosynthesis in red apples. Plant Physiol 142:1216–1232
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Wang HL, Wang W, Zhang P, Pan QH, Zhan JC, Huang WD (2011) Gene transcript accumulation, tissue and subcellular localization of anthocyanidin synthase (ANS) in developing grape berries. Plant Sci 179:103–113
Wang HX, Fan WJ, Li H, Yang J, Huang JR, Zhang P (2013) Functional characterization of dihydroflavonol-4-reductase in anthocyanin biosynthesis of purple sweet potato underlies the direct evidence of anthocyanins function against abiotic stresses. PLoS One 8:e78484
Wei YZ, Hu FC, Hu GB, Li XJ, Huang XM, Wang HC (2011) Differential expression of anthocyanin biosynthesis genes in relation to anthocyanin accumulation in the pericarp of Litchi Chinensis Sonn. PLoS One 6:e19455
Winkel-Shirley B (2002) Biosynthesis of flavonoids and effects of stress. Curr Opin Plant Biol 5:218–223
Wu X, Beecher GR, Holden JM, Haytowitz DB, Gebhardt SE, Prior RL (2006) Concentrations of anthocyanins in common foods in the United States and estimation of normal consumption. J Agric Food Chem 54:4069–4075
Xie XB, Li S, Zhang RF, Zhao J, Chen YC, Zhao Q, Yao YX, You CX, Zhang XS, Hao YJ (2012) The bHLH transcription factor MdbHLH3 promotes anthocyanin accumulation and fruit colouration in response to low temperature in apples. Plant Cell Environ 35:1884–1897
Xu WJ, Dubos C, Lepiniec L (2015) Transcriptional control of flavonoid biosynthesis by MYB-bHLH-WDR complexes. Trends Plant Sci 20:1360–1385
Zhang Y, Zheng S, Liu Z, Wang L, Bi Y (2010) Both HY5 and HYH are necessary regulators for low temperature-induced anthocyanin accumulation in Arabidopsis seedlings. J Plant Physiol 47:934–945
Zhang B, Hu Z, Zhang Y, Li Y, Zhou S, Chen G (2012) A putative functional MYB transcription factor induced by low temperature regulates anthocyanin biosynthesis in purple kale (Brassica oleracea var. acephala f. tricolor). Plant Cell Rep 31:281–289
Zhang HR, Zhao LX, Wang J, Zheng LL, Dang ZH, Wang YC (2014) Cloning and functional analysis of two flavanone-3-hydroxylase genes from Reaumuria trigyna. Acta Physiol Plant 36:1221–1229
Zhou L, Wang Y, Ren L, Shi QQ, Zheng BQ, Miao K, Cuo X (2014) Overexpression of Ps-CHI1, a homologue of the chalcone isomerase gene from tree peony (Paeonia suffruticosa), reduces the intensity of flower pigmentation in transgenic tobacco. Plant Cell Tissue Org 116:285–295
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31471870), the National Science Foundation for Young Scholars of China (31301770), and the Shanghai Seed Industry of Agricultural Science Project (2013, No. 5).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors agreed the content of the manuscript and had no conflicting interest.
Additional information
Communicated by A. Chandra.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11738_2016_2172_MOESM1_ESM.tif
Supplementary Fig. 1 A schematic representation of the exon-intron architecture of SmCHS, SmCHI, SmF3H, and SmDFR. SmCHS consists of two exons (black box) and one intron (fold line); SmCHI consists of four exons (black box) and three introns (fold line); SmF3H consists of three exons (black box) and two introns (fold line); SmDFR consists of six exons (black box) and five introns (fold line). The scale bar indicates 100 bp. (TIFF 154 kb)
11738_2016_2172_MOESM2_ESM.tif
Supplementary Fig. 2 Phylogenetic trees based on the amino acid sequences of CHSs CHIs, F3Hs, and DFRs. These trees were constructed using the MEGA 6.0 and Neighbor-Joining method with 1000 bootstrap replicates. (TIFF 1815 kb)
11738_2016_2172_MOESM3_ESM.tif
Supplementary Fig. 3 Expression of SmCHS, SmCHI, SmF3H, and SmDFR in different transgenic Arabidopsis lines. (A) SmCHS; (B) SmCHI; (C) SmF3H; and (D) SmDFR. WT, wild-type Arabidopsis; L1-L10, transgenic Arabidopsis lines, respectively. Quantifications were normalized by the expression of Arabidopsis actin (NM_179953). Data represent mean ± SD of three biological replicates. (TIFF 664 kb)
Rights and permissions
About this article
Cite this article
Jiang, M., Liu, Y., Ren, L. et al. Molecular cloning and characterization of anthocyanin biosynthesis genes in eggplant (Solanum melongena L.). Acta Physiol Plant 38, 163 (2016). https://doi.org/10.1007/s11738-016-2172-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-016-2172-0