Abstract
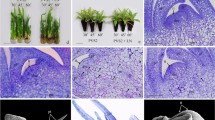
This study was carried out to optimize a modified droplet-vitrification procedure for the cryopreservation of shoot tips from different carnation genotypes. The best procedure was developed by applying orthogonal tests to the experimental data and by further investigation of the effects on the regrowth percentage. It consisted in preculturing shoot tips in liquid Murashige and Skoog (MS) medium with 0.3 M sucrose for 2 days, pretreating them in liquid MS medium with 5 % Dimethyl sulfoxide +5 % glycerol + 0.3 M sucrose for 10 min, osmoprotecting in Loading solution for 20 min at 25 °C, cryoprotecting with Plant vitrification solution No.2 (PVS2) for 60 min at 0 °C, transferring in drops of fresh PVS2 over aluminum strips and finally storing them in Liquid nitrogen. With the application of the optimized protocol, four carnation cultivars (‘Master’, ‘Calibra’, ‘Lamour’ and ‘Ofcar’) achieved regrowth percentage after cryopreservation ranging from 41 to 73 %. Ultrastructural observations investigated by using transmission electron microscopy showed that the cells encountered the stress during cryopreservation and the main damages occurred during the dehydration step. For surviving cells, the most of the damaged cells could be repaired after recovery growth. This modified protocol will aid in the long-term conservation of carnation germplasm and the ultrastructural studies will benefit for understanding the damage and recovery of the cells during cryopreservation.




Similar content being viewed by others
Abbreviations
- AC:
-
Activated carbon
- BA:
-
Benzylaminopurine
- DS:
-
Dilution solution
- DMSO:
-
Dimethyl sulfoxide
- GA3 :
-
Gibberellin A3
- IAA:
-
Indole acetic acid
- LN:
-
Liquid nitrogen
- LS:
-
Loading solution
- MS:
-
Murashige and Skoog medium
- PVS2:
-
Plant vitrification solution No. 2
- TEM:
-
Transmission electron microscopy
References
Benson EE (2008) Cryopreservation of phytodiversity: a critical appraisal of theory and practice. Crit Rev Plant Sci 27:141–219
Burchi G, Mercuri A, De Benedetti L, Giovannini A (1996) Transformation methods applicable to ornamental plants. Plant Tiss Cult Biotech 2:94–104
Cai ZG, Xu XB, Zhao YP, Zhou HP (2006) Ultrastructure of in vitro stem apex of kivi in cryopreservation. Acta Bot Boreal Occident Sin 26:1600–1604 (in Chinese)
Dereuddre J, Tannoury M (1995) Cryopreservation of germplasm of carnation (Dianthus caryophyllus L.). In: Bajaj YPS (ed) cryopreservation of plant germplasm I. Springer, Berlin Heidelberg, pp 458–477
Dereuddre J, Galerne M, Gazeau C (1987) The effects of sucrose on survival of freezing in liquid nitrogen (−196°C) of carnation (Dianthus caryophyllus L.) apical meristems cultivated in vitro. C R Acad Sci Paris Ser3 304:485–487
Engelmann F (2011) Use of biotechnologies for the conservation of plant biodiversity. In Vitro Cell Dev Plant 47:5–16
Fukai S (1989) Plant regeneration from shoot tips of Dianthus hybrid cryopreserved in liquid nitrogen up to 2 years. Plant Tissue Cult Lett 6:177–178
Gnanapragasam S, Vasil IK (1992) Ultrastructural changes in suspension culture cells of Panicum maximum during cryopreservation. Plant Cell Rep 11:l69–l174
Halmagyi A, Deliu C (2007) Cryopreservation of carnation (Dianthus caryophyllus L.) shoot tips by encapsulation-vitrification. Sci Hortic Amst 113:300–306
Halmagyi A, Lambardi M (2006) Cryopreservation of carnation (Dianthus caryophyllus L.). In: Teixeira da Silva JA (ed) Floriculture, ornamental and plant biotechnology, Volume II, Global Science Books, UK
Halmagyi A, Valimareanu S, Coste A, Deliu C, Isac V (2010) Cryopreservation of Malus shoot tips and subsequent plant regeneration. Rom Biotech Lett 15:79–85
Helliot B, Panis B, Poumay Y, Swennen R, Lepoivre P, Frison E (2002) Cryopreservation for the elimination of cucumber mosaic and banana streak viruses from banana (Musa spp.). Plant Cell Rep 20:1117–1122
Helliot B, Swennen R, Poumay Y, Frison E, Lepoivre P, Panis B (2003) Ultrastructural changes associated with cryopreservation of banana (Musa spp.) highly proliferating meristems. Plant Cell Rep 21:690–698
Heringer AS, Steinmacher DA, Schmidt EC, Bouzon ZL, Guerra MP (2013) Survival and ultrastructural features of peach palm (Bactris gasipaes, Kunth) somatic embryos submitted to cryopreservation through vitrification. Protoplasma 250:1185–1193
Kaczmarczyk A, Rutten T, Melzer M, Keller ERJ (2008) Ultrastructural changes associated with cryopreservation of potato (Solanum tuberosum L.) shoot tips. Cryo Lett 29:145–156
Kalita V, Choudhury H, Kumaria S, Tandon P (2012) Vitrification-based cryopreservation of shoot-tips of Pinus kesiya Royle ex. Gord Cryo Lett 33:58–68
Kim HH, Lee SC (2012) Personalisation’ of droplet-vitrification protocols for plant cells: a systematic approach to optimising chemical and osmotic effects. Cryo Lett 33:271–279
Leunufna S, Keller ERJ (2005) Cryopreservation of yams using vitrification modified by including droplet method: effects of cold acclimation and sucrose. Cryo Lett 26:93–102
Liu F, Wang JH, Huang CN, Yan QS, Zhang XQ (1998) Ultrastructural changes in rice embryogenic suspension cells cryopreserved by vitrification. Chin Rice Sci 12:17–20 (in Chinese)
Lyu SR, Wu WT, Hou CC, Hsieh WH (2010) Study of cryopreservation of articular chondrocytes using the Taguchi method. Cryobiology 60:165–176
Matsumoto T, Sakai A, Yamada K (1994) Cryopreservation of in vitro-grown apical meristems of wasabi (Wasabia japonica) by vitrification and subsequent high plant regeneration. Plant Cell Rep 13:442–446
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Niu YL, Luo ZR, Zhang YF, Zhang QL (2012) Cryopreservation of in vitro grown shoot tips of Diospyros kaki Thunb. using different methods. Cryo Lett 33:69–74
Panis B (1995) Cryopreservation of banana (Musa sp.) germplasm. Dissertationes de Agricultura no. 272, Katholieke Universiteit Leuven, Belgium
Panis B, Lambardi M (2006) Status of cryopreservation technologies in plants (crops and forest trees). In: Ruane J, Sonnino A (eds) The role of biotechnology in exploring and protecting agricultural genetic resources. FAO, Rome, pp 61–78
Pinker I, Halmagyi A, Olbricht K (2009) Effects of sucrose preculture on cryopreservation by droplet-vitrification of strawberry cultivars and morphological stability of cryopreserved plants. Cryo Lett 30:202–211
Reed BM (2008) Plant Cryopreservation: A Practical Guide. Springer, New York
Sakai A, Engelmann F (2007) Vitrification, encapsulation-vitrification and droplet-vitrification: a review. Cryo Lett 28:151–172
Sakai A, Kobayashi S, Oiyama I (1990) Cryopreservation of nucellar cells of navel orange (Citrus sinensis Osb. var. brasiliensis Tanaka) by vitrification. Plant Cell Rep 9:30–33
Seibert M (1976) Shoot initiation from carnation shoot apices frozen to −196°C. Science 191:1178–1179
Seibert M, Wetherbee PJ (1977) Increased survival and differentiation of frozen herbaceous plant organ cultures through cold treatment. Plant Physiol 59:1043–1046
Sekizawa K, Yamamoto SI, Rafique T, Fukui K, Niino T (2011) Cryopreservation of in vitro-grown shoot tips of carnation (Dianthus caryophyllus L.) by vitrification method using aluminium cryo-plates. Plant Biotech 28:401–405
Sershen Berjak P, Pammenter NW, Wesley-Smith J (2012) The effects of various parameters during processing for cryopreservation on the ultrastructure and viability of recalcitrant zygotic embryos of Amaryllis belladonna. Protoplasma 249:155–169
Singh J (1979) Ultrastructural alterations in cells of hardened and non-hardened winter rye during hyperosmotic and extracellular freezing stresses. Protoplasma 98:329–341
Tannoury M, Vintejoux C (1997) Cytological studies of Dianthus caryophyllus apex after cryopreservation. Acta Bot Gallica 144:107–118
Tannoury M, Ralambosoa J, Kaminski M, Dereuddre J (1991) Cryopreservation by vitrification of alginate-coated carnation (Dianthus caryophyllus L.) shoot-tips of in vitro plantlets. C R Acad Sci Paris Ser 13:633–638
Uemura M, Sakai A (1980) Survival of carnation (Dianthus caryophyllus L.) shoot apices frozen to the temperature of liquid nitrogen. Plant Cell Physiol 21:85–94
Wang QC, Valkonen JPT (2008) Efficient elimination of sweetpotato little leaf phytoplasma from sweetpotato by cryotherapy of shoot tips. Plant Pathol 57:338–347
Wang JH, Ge JG, Liu F, Huang CN (1998) Ultrastructural changes during cryopreservation of rice (Oryza sativa L.) embryogenic suspension cells by vitrification. Cryo Lett 19:49–54
Wu LM, Zeng JW, Peng SA, Yi GJ, Niu WC (2006) Ultrastructural observation of banana shoot-tip cell during cryopreservation by vitrification. Mol Cell Biol 39:563–567 (in Chinese)
Yap LV, Noor NM, Clyde MM, Chin HF (2011) Cryopreservation of Garcinia cowa shoot tips by vitrification: the effects of sucrose preculture and loading treatment on ultrastructural changes in meristematic cells. Cryo Lett 32:188–196
Zhang SM, Li JG, Chen HB, Xu CX, Han LN (2006) Ultrastructural changes associated with droplet vitrification cryopreservation of banana shoot tip. Acta Horticult Sin 33:709–713
Acknowledgments
This study was supported by Core Research Budget of the Non-profit Governmental Research Institution (ICS, CAAS, 2011006), the Crop Germplasm Resources Protection and Utilization Special Grant from the Ministry of Agriculture, People’s Republic of China (NB2012-2130135-25-11) and National Key Technology R&D Program in accordance with the 12th five-year plan of People’s Republic of China (2013BAD01B01). The authors would like to acknowledge Professor Ge Hong of Institute of Vegetables and Flowers (CAAS) for supplying materials of Dianthus species. The authors also would like to acknowledge Barbara Reed (National Center for Genetic Resources Preservation, United State Department of Agriculture, Corvallis, Oregon, U.S.A.) for assistance in revision of the paper.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by M. Lambardi.
Rights and permissions
About this article
Cite this article
Zhang, JM., Zhang, XN., Lu, XX. et al. Optimization of droplet-vitrification protocol for carnation genotypes and ultrastructural studies on shoot tips during cryopreservation. Acta Physiol Plant 36, 3189–3198 (2014). https://doi.org/10.1007/s11738-014-1685-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-014-1685-7