Abstract
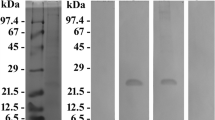
Two orders of green alga (Cladophorales and Charales) were investigated for the presence of protein tyrosine kinase activity. Proteins of 70 and 85 kDa were found to be tyrosine phosphorylated in Cladophora fracta, with an additional phosphorylated band evident at the 120-kDa region in Chara vulgaris, suggestive of the presence of putative tyrosine kinase activity in these algal species. A 70-kDa protein was immunoprecipitated from both species using a polyclonal antibody against non-receptor protein tyrosine kinase Syk. The protein was found to be phosphorylated on tyrosine, which was prevented upon pretreatment of algal cells with piceatannol. The extent of phosphorylation directly correlated with algal growth, suggesting a link between Syk kinase activity and growth signaling. These observations supported the presence of Syk-like kinase in the green algal species, which could have critical role in the algal growth and development.




Similar content being viewed by others
References
Annen F, Stockhaus J (1999) SbRLK1, a receptor-like protein kinase of Sorghum bicolor (L.) Moench that is expressed in mesophyll cells. Planta 208:420–425
Blume Y, Yemets A, Sulimenko V, Sulimenko T, Chan J, Lloyd C, Dráber P (2008) Tyrosine phosphorylation of plant tubulin. Planta 229:143–150
Brand U, Fletcher JC, Hobe M, Meyerowitz EM, Simon R (2000) Dependence of stem cell fate in Arabidopsis on a feedback loop regulated by CLV3 activity. Science 289:617–619
Braun DM, Walker JC (1996) Plant transmembrane receptors: new pieces in the signalling puzzle. Trends Biochem Sci 21:70–73
Chitcholtan K, Garrill A (2005) A beta4 integrin-like protein co-localises with a phosphotyrosine containing protein in the oomycete Achlya bisexualis: inhibition of tyrosine phosphorylation slows tip growth. Fungal Genet Biol 42:534–545
Chu SP (1942) The influence of mineral composition of the medium on the growth of planktonic algae. Part I. Methods and culture media. J Ecol 30:284–325
Collins MO, Yu L, Coba MP, Husi H, Campuzano I, Blackstock WP, Choudhary JS, Grant SGN (2005) Proteomic analysis of in vivo phosphorylated synaptic proteins. J Biol Chem 280:5972–5982
Fazi B, Cope MJ, Douangamath A, Ferracuti S, Schirwitz K, Zucconi A, Drubin DG, Wilmanns M, Cesareni G, Castagnoli L (2002) Unusual binding properties of the SH3 domain of the yeast actin-binding protein Abp1: structural and functional analysis. J Biol Chem 277:5290–5298
Heck DA (2000) Future research directions in plant signal transduction mechanisms. Indian J Biochem Biophys 37:369–376
Jeong S, Trotochaud AE, Clark SE (1999) The Arabidopsis CLAVATA2 gene encodes a receptor-like protein required for the stability of the CLAVATA1 receptor-like kinase. Plant Cell 11:1925–1934
Jinn TL, Stone JM, Walker JC (2000) HAESA, an Arabidopsis leucine-rich repeat receptor kinase, controls floral organ abscission. Genes Dev 14:108–117
Klimowicz AC, Bisson SA, Hans K, Long EM, Hansen HC, Robbins SM (2009) The phytochemical piceatannol induces the loss of CBL and CBL-associated proteins. Mol Cancer Ther 8:602–614
Kohorn BD (1999) Shuffling the deck: plant signalling plays a club. Trends Cell Biol 9:381–383
Lam BC, Blumwald E (2002) Domains as functional building blocks of plant proteins. Trends Plant Sci 7:544–549
Li YL, Sun J, Xia GX (2005) Cloning and characterization of a gene for an LRR receptor-like protein kinase associated with cotton fiber development. Mol Genet Genomics 273:217–224
Luan S (2003) Protein phosphatases in plants. Annu Rev Plant Biol 54:63–92
Oliver JM, Burg DL, Wilson BS, McLaughlin JL, Geahlen RL (1994) Inhibition of mast cell Fc epsilon R1-mediated signaling and effector function by the Syk-selective inhibitor, piceatannol. J Biol Chem 269:29697–29703
Ortega X, Velásquez JC, Pérez LM (2005) IP3 production in the hypersensitive response of lemon seedlings against Alternaria alternata involves active protein tyrosine kinases but not a G-protein. Biol Res 38:89–99
Pawson T (1995a) Protein tyrosine kinases: getting down to specifics. Nature 373:477–478
Pawson T (1995b) Protein modules and signalling network. Nature 373:573–580
Pawson T, Kofler M (2009) Kinome signaling through regulated protein–protein interactions in normal and cancer cells. Curr Opin Cell Biol 21:147–153
Rodriguez-Zapata LC, Hemadez-Sotomayor SM (1998) Evidence of protein tyrosine kinase activity in Catharanthus roseus roots transformed by Agrobacterium rhizogenes. Planta 204:70–77
Rudrabhatla P, Reddy MM, Rajasekharan R (2006) Genome-wide analysis and experimentation of plant serine/threonine/tyrosine-specific protein kinases. Plant Mol Biol 60:293–319
Schoof H, Lenhard M, Haecker A, Mayer KF, Jurgens G, Laux T (2000) The stem cell population of Arabidopsis shoot meristems in maintained by a regulatory loop between the CLAVATA and WUSCHEL genes. Cell 100:635–644
Sokolovski S, Hills A, Gay R, Garcia-Mata C, Lamattina L, Blatt MR (2005) Protein phosphorylation is a prerequisite for intracellular Ca2+ release and ion channel control by nitric oxide and abscisic acid in guard cells. Plant J 43:520–529
Sugiyama N, Nakagami H, Mochida K, Daudi A, Tomita M, Shirasu K, Ishihama Y (2008) Large-scale phosphorylation mapping reveals the extent of tyrosine phosphorylation in Arabidopsis. Mol Syst Biol 4:193
Tohyama Y, Yamamura H (2009) Protein tyrosine kinase, Syk: a key player in phagocytic cells. J Biochem 145:267–273
Trojanek J, Ek P, Scoble J, Muszynska G, Engstrom L (1996) Phosphorylation of plant proteins and the identification of protein-tyrosine kinase activity in maize seedlings. Eur J Biochem 235:338–344
Trojanek JB, Klimecka MM, Fraser A, Dobrowolska G, Muszynska G (2004) Characterization of dual specificity protein kinase from maize seedlings. Acta Biochim Pol 51:635–647
Trotochaud AE, Jeong S, Clark SE (2000) CLAVATA3, a multimeric ligand for the CLAVATA1 receptor-kinase. Science 289:613–617
Tullberg A, Hakansson G, Race HL (1998) A protein tyrosine kinase of chloroplast thylakoid membranes phosphorylates light harvesting complex II proteins. Biochem Biophys Res Commun 250:617–622
Vendel AC, Calemine-Fenaux J, Izrael-Tomasevic A, Chauhan V, Arnott D, Eaton DL (2009) B and T lymphocyte attenuator regulates B cell receptor signaling by targeting Syk and BLNK. J Immunol 182:1509–1517
Wang Q, Snell WJ (2003) Flagellar adhesion between mating type plus and mating type minus gametes activates a flagellar protein-tyrosine kinase during fertilization in Chlamydomonas. J Biol Chem 278:32936–32942
Williams JG, Zvelebil M (2004) SH2 domains in plants imply new signalling scenarios. Trends Plant Sci 9:161–163
Woodside DG, Obergfell A, Talapatra A, Calderwood DA, Shattil SJ, Ginsberg MH (2002) The N-terminal SH2 domains of Syk and ZAP-70 mediate phosphotyrosine-independent binding to integrin beta cytoplasmic domains. J Biol Chem 277:39401–39408
Yamashita T, Suzuki R, Backlund PS, Yamashita Y, Yergey AL, Rivera J (2008) Differential dephosphorylation of the FcRgamma immunoreceptor tyrosine-based activation motif tyrosines with dissimilar potential for activating Syk. J Biol Chem 283:28584–28594
Yu LP, Simon EJ, Trotochaud AE, Clark SE (2000) POLTERGEIST functions to regulate meristem development downstream of the CLAVATA loci. Development 127:1661–1670
Zou W, Kitaura H, Reeve J, Long F, Tybulewicz VL, Shattil SJ, Ginsberg MH, Ross FP, Teitelbaum SL (2007) Syk, c-Src, the alphavbeta3 integrin, and ITAM immunoreceptors, in concert, regulate osteoclastic bone resorption. J Cell Biol 176:877–888
Acknowledgments
The author thankfully acknowledges grants received from the Department of Science and Technology, Government of India, under the DST Women Scientist Scheme (WOS-A), and the University Grants Commission, under the UGC Part-time Research Associateship Scheme.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Janska.
Rights and permissions
About this article
Cite this article
Dash, A. Evidence for the presence of non-receptor protein tyrosine kinases in algal cells. Acta Physiol Plant 32, 177–182 (2010). https://doi.org/10.1007/s11738-009-0395-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-009-0395-z