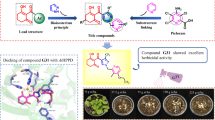
Abstract
Twenty six novel pyrimidin-4-amine derivatives containing the 1,2,4-oxadiazole motif were synthesized. Their chemical structures were confirmed by 1H nuclear magnetic resonance (NMR), 13C NMR, and highresolution mass spectrography. The insecticidal activity results indicated that some of them possessed excellent insecticidal activity (100%) against Mythimna separate, especially for compounds 6d, 6f, 6o, 6w, 6y and 6z. These compounds exhibited no activity against the insects Aphis medicagini and Tetranychus cinnabarinus. The structure-insecticidal activity relationships are discussed. Density functional theory analysis can potentially be used to design more active compounds. These results provide useful insecticide design information for further optimization.

Similar content being viewed by others
References
Yang J L, Guan A Y, Li Z N, Zhang P F, Liu C L. Design, synthesis, and structure-activity relationship of novel spiropyrimidinamines as fungicides against pseudoperonospora cubensis. Journal of Agricultural and Food Chemistry, 2020, 68(24): 6485–6492
Guo W K, Xing Y J, Zhang Q S, Xie J Q, Huang D X, Gu H J, He P, Zhou M R, Xu S F, Pang X F, et al. Synthesis and biological evaluation of B-cell lymphoma 6 inhibitors of N-phenyl-4-pyrimidinamine derivatives bearing potent activities against tumor growth. Journal of Medicinal Chemistry, 2020, 63(2): 676–695
Mohana K N, Mallesha L. Synthesis and in vitro biological activity of N-(5-amino-2-methylphenyl)-4-(3-pyridyl)-2-pyrimidinamine derivatives. Bulgarian Chemical Communications, 2011, 43: 395–400
Soliman N N, Salam M A, Fadda A A, Abdel-Motaal M. Synthesis, characterization, and biochemical impacts of some new bioactive sulfonamide thiazole derivatives as potential insecticidal agents against the cotton leafworm, spodoptera littoralis. Journal of Agricultural and Food Chemistry, 2020, 68(21): 5790–5805
Zhang N, Huang M Z, Liu A P, Liu M H, Li L Z, Zhou C G, Ren Y G, Ou X M, Long C Y, Sun J, Dang M M, Lan Z L. Design, synthesis, and insecticidal/acaricidal evaluation of novel pyrimidinamine derivatives containing phenyloxazole moiety. Chemical Papers, 2020, 74(3): 963–970
Wishka D G, Graber D R, Kopta L A, Olmsted R A, Friis J M, Hosley J D, Adams W J, Seest E P, Castle T M, Dolak L A, et al. (-)-6-Chloro-2-[(1-furo[2,3-c]pyridin-5-yl-ethyl)thio]-4-pyrimidina-mine, PNU-142721, a new broad spectrum HIV-1 non-nucleoside reverse transcriptase inhibitor. Journal of Medicinal Chemistry, 1998, 41(9): 1357–1360
Marcic D. Acaricides in modern management of plant-feeding mites. Journal of Pest Science, 2012, 85(4): 395–408
Zhang Y, Shang J F, Li H, Liu H, Song H B, Wang B L, Li Z M. Synthesis of novel N-pyridylpyrazole derivatives containing 1,2,4-oxadiazole moiety via 1,3-dipolar cycloaddition and their structures and biological activities. Chinese Chemical Letters, 2020, 31(5): 1276–1280
Zhang J, Li J C, Song J L, Cheng Z Q, Sun J Z, Jiang C S. Synthesis and evaluation of coumarin/1,2,4-oxadiazole hybrids as selective BChE inhibitors with neuroprotective activity. Asian Journal of Nature Product Research, 2019, 21(11): 1090–1103
Cao X D, Yao Z Y, Dou F, Zhang Y F, Qiu Y L, Zhao S, Xu X Q, Liu X, Liu B F, Chen Y, Zhang G. Synthesis and biological evaluation of sigma-1 (sigma(1)) receptor ligands based on phenyl-1,2,4-oxadiazole derivatives. Chemistry & Biodiversity, 2019, 16(3): e1800599
Vijaya Bhargavi M, Shashikala P, Sumakanth M, Krishna C. Synthesis, molecular docking, analgesic, and anti-inflammatory activities of new 1,2,4-oxadiazolo-sulfonamides. Russian Journal of General Chemistry, 2018, 88(4): 804–811
El Mansouri A E, Oubella A, Maatallah M, AitItto M Y, Zahouily M, Morjani H, Lazrek H B. Design, synthesis, biological evaluation and molecular docking of new uracil analogs-1,2,4-oxadiazole hybrids as potential anticancer agents. Bioorganic & Medicinal Chemistry Letters, 2020, 30(19): 127438
Ravinaik B, Ramachandran D, Rao M V B. Design, synthesis and anticancer evaluation of 1,2,4-oxadiazole bearing isoxazole-pyrazole derivatives. Letters in Organic Chemistry, 2020, 17(5): 352–359
Mohammadi-Khanaposhtani M, Fahimi K, Karimpour-Razkenari E, Safavi M, Mahdavi M, Saeedi M, Akbarzadeh T. Design, synthesis and cytotoxicity of novel coumarin-1,2,3-triazole-1,2,4-oxadiazole hybrids as potent anti-breast cancer agents. Letters in Drug Design & Discovery, 2019, 16(7): 818–824
Benmansour F, Eydoux C, Querat G, de Lamballerie X, Canard B, Alvarez K, Guillemot J C, Barral K. Novel 2-phenyl-5-[(E)-2-(thiophen-2-yl)ethenyl]-1,3,4-oxadiazole and 3-phenyl-5-[(E)-2-(thiophen-2-yl)ethenyl]-1,2,4-oxadiazole derivatives as dengue virus inhibitors targeting NS5 polymerase. European Journal of Medicinal Chemistry, 2016, 109: 146–156
Avanzo R E, Padron J M, D’Accorso N B, Fascio M L. Synthesis and in vitro antiproliferative activities of (5-ary1–1,2,4-oxadiazole-3-y1) methyl D-ribofuranosides. Bioorganic & Medicinal Chemistry Letters, 2017, 27(16): 3674–3677
Cunha F S, Nogueira J M R, de Aguiar A P. Synthesis and antibacterial evaluation of 3,5-diaryl-1,2,4-oxadiazole derivatives. Journal of the Brazilian Chemical Society, 2018, 29: 2405–2416
Santos J M, Silva D M A D E, Macedo T S, Teixeira H M P, Moreira D R M, Challal S, Wolfender J L, Queiroz E F, Soares M B P. Conjugation of N-acylhydrazone and 1,2,4-oxadiazole leads to the identification of active antimalarial agents. Bioorganic & Medicinal Chemistry, 2016, 24(22): 5693–5701
Shen Z H, Sun Z H, Becnel J J, Estep A, Wedge D E, Tan C X, Weng J Q, Han L, Liu X H. Synthesis and mosquiticidal activity of novel hydrazone containing pyrimidine derivatives against Aedes aegypti. Letters in Drug Design & Discovery, 2018, 15(9): 951–956
Liu X H, Wen Y H, Cheng L, Xu T M, Wu N J. Design, synthesis, pesticidal activities of pyrimidin-4-amine derivatives bearing a 5-(trifluoromethyl)-1,2,4-oxadiazole moiety. Journal of Agricultural and Food Chemistry, 2021, 69(25): 6968–6980
Chen S L, Zhang Y, Liu Y X, Wang Q M. Highly efficient synthesis and acaricidal and insecticidal activities of novel oxazolines with N-heterocyclic substituents. Journal of Agricultural and Food Chemistry, 2021, 69(12): 3601–3606
Fu Q, Cai P P, Cheng L, Zhong L K, Tan C X, Shen Z H, Han L, Liu X H. Synthesis and herbicidal activity of novel pyrazole aromatic ketone analogs as HPPD inhibitor. Pest Management Science, 2020, 76(3): 868–879
Liu X H, Yu W, Min L J, Wedge D E, Tan C X, Weng J Q, Wu H K, Cantrell C L, Bajsa-Hischel J, Hua X W, et al. Synthesis and pesticidal activities of new quinoxalines. Journal of Agricultural and Food Chemistry, 2020, 68(28): 7324–7332
Hoffman T J, Stierli D, Pouliot M, Beaudegnies R. Microbiocidal oxadiazole derivatives. WO Patent, 2017072247A1, 2017-05-04
Zhang Y, Shang J F, Li H, Liu H, Song H B, Wang B L, Li Z M. Synthesis of novel N-pyridylpyrazole derivatives containing 1,2,4-oxadiazole moiety via 1,3-dipolar cycloaddition and their structures and biological activities. Chinese Chemical Letters, 2020, 31(5): 1276–1280
Yu C S, Wang Q, Bajsa-Hirschel J, Cantrell C, Duke S O, Liu X H. Synthesis, crystal structure, herbicidal activity and SAR study of novel N-(arylmethoxy)-2-chloronicotinamides derived from nicotinic acid. Journal of Agricultural and Food Chemistry, 2021, 69(23): 6423–6430
Hao G F, Dong Q, Yang G F. Comparative study on the constitutive properties of marketed pesticides. Molecular Informatics, 2011, 30(6–7): 614–622
Rao H, Huang F C, Wang Y, Wang X, Tang T, Zeng X, Li Z, Chen Y. Physicochemical profiles of the marketed agrochemicals and clues for agrochemical lead discovery and screening library development. Molecular Informatics, 2015, 34(5): 331–338
Acknowledgements
This work was funded by the Zhejiang Provincial Natural Science Foundation of China (Grant No. LY19C140002), the Company of Three Win (Grant No. KYY-HX-20200746), Research Fund of Department of Education of Zhejiang Province (Grant No. Y202043162). We thank Dong-Song Hu for evaluating insecticidal activity.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wen, YH., Cheng, L., Xu, TM. et al. Synthesis, insecticidal activities and DFT study of pyrimidin-4-amine derivatives containing the 1,2,4-oxadiazole motif. Front. Chem. Sci. Eng. 16, 1090–1100 (2022). https://doi.org/10.1007/s11705-021-2091-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11705-021-2091-5