Abstract
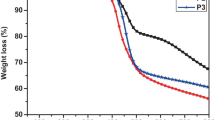
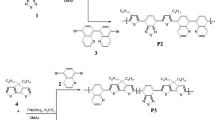
This research reports the synthesis and characterization of two new donor–acceptor type conjugated polymers carrying indolocarbazole as donor, benzothiadiazole and diketopyrrolopyrrole as acceptor via stille coupling polymerization reactions. The structures of the polymer were established by spectroscopic techniques and polymer molecular weight by gel permeation chromatography. Further the linear optical and electrochemical properties of the polymers have been investigated. Polymers showed broad absorption and good fluorescence behaviour with good quantum yields. Optical bandgaps of the polymers were estimated using UV–visible absorption edge and found to be 2.34 and 2.12 eV respectively. Further, theoretical evaluation of the polymers was carried using density functional theory to investigate their geometrical, electrochemical and optical properties using DFT/B3LYP. The obtained results are in good agreement with the experimental results.












Similar content being viewed by others
References
Akkuratov AV, Nikitenko SL, Kozlov AS et al (2020) Design of novel thiazolothiazole-containing conjugated polymers for organic solar cells and modules. Sol Energy 198:605–611. https://doi.org/10.1016/j.solener.2020.01.087
Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652. https://doi.org/10.1063/1.464913
Chen Q, Zhang L, Ebrahim S et al (2013) Synthesis and structure study of copolymers from thiadiazole fused indolocarbazole and dithienosilole. Polymer 54:223–229. https://doi.org/10.1016/j.polymer.2012.11.006
Cheng Y-J, Yang S-H, Hsu C-S (2009) Synthesis of conjugated polymers for organic solar cell applications. Chem Rev 109:5868–5923. https://doi.org/10.1021/cr900182s
Chung K, McAllister A, Bilby D et al (2015) Designing interchain and intrachain properties of conjugated polymers for latent optical information encoding. Chem Sci 6:6980–6985. https://doi.org/10.1039/C5SC02403J
Chung HY, Oh J, Park J-H et al (2020) Spectroscopic studies on intramolecular charge-transfer characteristics in small-molecule organic solar cell donors: a case study on ADA and DAD triad donors. J Phys Chem C. https://doi.org/10.1021/acs.jpcc.0c06741
de Leeuw DM, Simenon MMJ, Brown AR, Einerhand REF (1997) Stability of n-type doped conducting polymers and consequences for polymeric microelectronic devices. Synth Met 87:53–59. https://doi.org/10.1016/S0379-6779(97)80097-5
Ekbote A, Mobin SM, Misra R (2018) Structure–property relationship in multi-stimuli responsive D-A–A′ benzothiazole functionalized isomers. J Mater Chem C 6:10888–10901. https://doi.org/10.1039/C8TC04310H
Fan L-J, Zhang Y, Murphy CB et al (2009) Fluorescent conjugated polymer molecular wire chemosensors for transition metal ion recognition and signaling. Coord Chem Rev 253:410–422. https://doi.org/10.1016/j.ccr.2008.03.008
Gokce G, Icli Ozkut M (2018) An indolocarbazole based yellow-to-cyan soluble electrochromic polymer. Org Electron 52:317–322. https://doi.org/10.1016/j.orgel.2017.11.017
Grzybowski M, Gryko DT (2015) Diketopyrrolopyrroles: synthesis, reactivity, and optical properties. Adv Opt Mater 3:280–320. https://doi.org/10.1002/adom.201400559
Hehre WJ, Ditchfield R, Pople JA (1972) Self—consistent molecular orbital methods. XII. Further extensions of Gaussian—type basis sets for use in molecular orbital studies of organic molecules. J Chem Phys 56:2257–2261. https://doi.org/10.1063/1.1677527
Janosik T, Rannug A, Rannug U et al (2018) Chemistry and properties of indolocarbazoles. Chem Rev 118:9058–9128. https://doi.org/10.1021/acs.chemrev.8b00186
Jin Cheon H, Li X, Jin Jeong Y et al (2020) A novel design of donor–acceptor polymer semiconductors for printed electronics: application to transistors and gas sensors. J Mater Chem C 8:8410–8419. https://doi.org/10.1039/D0TC01341B
Khetubol A, Van Snick S, Clark ML et al (2015) Improved spectral coverage and fluorescence quenching in donor-acceptor systems involving indolo[3-2-b]carbazole and boron-dipyrromethene or diketopyrrolopyrrole. Photochem Photobiol 91:637–653. https://doi.org/10.1111/php.12437
Kim M, Ryu SU, Park SA et al (2020) Donor–acceptor-conjugated polymer for high-performance organic field-effect transistors: a progress report. Adv Funct Mater 30:1904545. https://doi.org/10.1002/adfm.201904545
Lee C, Yang W, Parr RG (1988) Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789. https://doi.org/10.1103/PhysRevB.37.785
Li J, Grimsdale AC (2010) Carbazole-based polymers for organic photovoltaic devices. Chem Soc Rev 39:2399–2410. https://doi.org/10.1039/B915995A
Li J, Liu J, Wei C-W et al (2013) Emerging applications of conjugated polymers in molecular imaging. Phys Chem Chem Phys 15:17006–17015. https://doi.org/10.1039/C3CP51763B
Li W, Guo Y, Wang Y et al (2018) A “chain–lock” strategy to construct a conjugated copolymer network for supercapacitor applications. J Mater Chem A 7:116–123. https://doi.org/10.1039/C8TA08766K
Mahesh K, Karpagam S, Pandian K (2019) How to design donor-acceptor based heterocyclic conjugated polymers for applications from organic electronics to sensors. Top Curr Chem (Z) 377:12. https://doi.org/10.1007/s41061-019-0237-4
Majidizadeh Fini A, Kalantari Fotooh F, Nateghi MR, Shahi S (2020) Combined experimental and theoretical investigation of optical and structural properties of poly aniline derivatives. Chem Pap. https://doi.org/10.1007/s11696-020-01273-4
Neese F (2012) The ORCA program system. WIREs Comput Mol Sci 2:73–78. https://doi.org/10.1002/wcms.81
Negru OI, Grigoras M (2019) Synthesis and properties of copolyarylenes containing indolo[3,2-b]carbazole moieties in the backbone. J Polym Res 26:30. https://doi.org/10.1007/s10965-018-1693-1
Park KS, Kwok JJ, Dilmurat R et al (2019) Tuning conformation, assembly, and charge transport properties of conjugated polymers by printing flow. Sci Adv 5:eaaw757. https://doi.org/10.1126/sciadv.aaw7757
Pei J, Wen S, Zhou Y et al (2011) A low band gap donor–acceptor copolymer containing fluorene and benzothiadiazole units: synthesis and photovoltaic properties. New J Chem 35:385–393. https://doi.org/10.1039/C0NJ00378F
Pop F, Humphreys J, Schwarz J et al (2019) Towards more sustainable synthesis of diketopyrrolopyrroles. New J Chem 43:5783–5790. https://doi.org/10.1039/C9NJ01074B
Sekine C, Tsubata Y, Yamada T et al (2014) Recent progress of high performance polymer OLED and OPV materials for organic printed electronics. Sci Technol Adv Mater 15:034203. https://doi.org/10.1088/1468-6996/15/3/034203
Shivashankar SM, Anantapadmanabha VK, Adhikari AV (2013) Optical limiting materials: synthesis, electrochemical and optical studies of new thiophene based conjugated polymers carrying 1,3,4-oxadiazole units. Polym Eng Sci 53:1347–1356. https://doi.org/10.1002/pen.23362
Suman SA, Keshtov ML et al (2019) New indolo carbazole-based non-fullerene n-type semiconductors for organic solar cell applications. J Mater Chem C 7:543–552. https://doi.org/10.1039/C8TC05318A
Sun H, Autschbach J (2014) Electronic energy gaps for π-conjugated oligomers and polymers calculated with density functional theory. J Chem Theory Comput 10:1035–1047. https://doi.org/10.1021/ct4009975
Tong J, Guo P, Zhang H et al (2015) Synthesis of modified benzothiadiazole-thiophene-cored acceptor and carbazole/indolocarbazole alternating conjugated polymers and their photovoltaic applications. Polym Bull. https://doi.org/10.1007/s00289-014-1292-1
Tsai J-H, Chueh C-C, Mei-Hsiu L et al (2009) Synthesis of new indolocarbazole-acceptor alternating conjugated copolymers and their applications to thin film transistors and photovoltaic cells. Macromolecules. https://doi.org/10.1021/ma802720n
Udayakumar D, Adhikari AV (2006) Synthesis and characterization of new light-emitting copolymers containing 3,4-dialkoxythiophenes. Synth Met 156:1168–1173. https://doi.org/10.1016/j.synthmet.2006.07.007
Wong MY (2017) Recent advances in polymer organic light-emitting diodes (PLED) using non-conjugated polymers as the emitting layer and contrasting them with conjugated counterparts. J Electr Mater 46:6246–6281. https://doi.org/10.1007/s11664-017-5702-7
Zeglio E, Rutz AL, Winkler TE et al (2019) Conjugated polymers for assessing and controlling biological functions. Adv Mater 31:1806712. https://doi.org/10.1002/adma.201806712
Zhan X, Zhu D (2010) Conjugated polymers for high-efficiency organic photovoltaics. Polym Chem 1:409–419. https://doi.org/10.1039/B9PY00325H
Zhang D-W, Li M, Chen C-F (2020) Recent advances in circularly polarized electroluminescence based on organic light-emitting diodes. Chem Soc Rev 49:1331–1343. https://doi.org/10.1039/C9CS00680J
Acknowledgements
Authors are grateful to RV College of Engineering for providing characterization facility to carry out this research work .
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
I certify that there is no actual or potential conflict of interest in relation to this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vishnumurthy, K.A., Girish, K.H. Synthesis, photophysical, electrochemical and computational study of indolocarbazole based donor acceptor type conjugated polymers. Chem. Pap. 75, 1969–1980 (2021). https://doi.org/10.1007/s11696-020-01445-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01445-2