Abstract
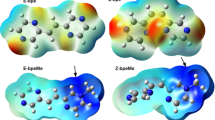
The 4-((9-phenyl-9H-xanthene-9-yl) oxy) phthalonitrile compound was synthesized and characterized as starting material. Zinc phthalocyanine was obtained by reaction of 4-((9-phenyl-9H-xanthen-9-yl) oxy) phthalonitrile with Zn(CH3COO)2. Novel compounds were characterized using mass spectra, UV–Vis spectroscopy, 1H-NMR, 13C NMR, and infrared spectroscopy. The fluorescence, emission, excitation, and absorption spectra of the zinc phthalocyanine compound were studied in tetrahydrofuran (THF). The determination of these properties is very useful for photodynamic therapy applications. Also, zinc phthalocyanine (Zn-Pc) was optimized with the basic set of 6-311G and LanL2DZ of the Density functional theory (DFT). The energy band-gap HOMO–LUMO of the molecule was determined. Chemical indices were calculated using HOMO–LUMO energies. The electrophilic region and the nucleophilic region were defined from the molecular electrostatic potential (MESP) maps. Dipole moment components have been calculated and can be considered as potential candidates for the design of non-linear optical materials.









Similar content being viewed by others
References
Abkari A, Chaabane I, Guidara K (2016) DFT (B3LYP/LanL2DZ and B3LYP/6311G + (d, p)) comparative vibrational spectroscopic analysis of organic–inorganic compound bis(4-acetylanilinium) tetrachlorocuprate(II). Physica E Low Dimens Syst Nanostruct 81:136–144. https://doi.org/10.1016/j.physe.2016.03.010
Agırtas MS (2007) Synthesis and characterization of novel symmetrical phthalocyanines substituted with four benzo [d] [1, 3] dioxol-5-ylmethoxy groups. Inorg Chim Acta 360:2499–2502. https://doi.org/10.1016/j.ica.2006.12.029
Ağırtaş MS, Yıldıko U (2008) Synthesis of new phthalocyanines substituted with four benzo[d]thiazol-2-ylthio groups. J Coord Chem 61:2781–2786. https://doi.org/10.1080/00958970801975083
Ağırtas MS, Cabir B, Özdemir S (2013) Novel metal (II) phthalocyanines with 3,4,5-trimethoxybenzyloxy-substituents: synthesis, characterization, aggregation behaviour and antioxidant activity. Dyes Pigments 96:152–157. https://doi.org/10.1016/j.dyepig.2012.07.023
Arslantaş A, Ağirtaş MS (2018) The interaction between a zinc (II) phthalocyanine compound bearing octakis phenoxyacetamide substituents and calf thymus DNA. Turk. J. Chem. 42:1310–1320. https://doi.org/10.3906/kim-1805-18
Basiuk EV, Huerta L, Basiuk VA (2019) Noncovalent bonding of 3d metal (II) phthalocyanines with single-walled carbon nanotubes: a combined DFT and XPS study. Appl Surf Sci 470:622–630. https://doi.org/10.1016/j.apsusc.2018.11.159
Chen D, Wang H (2019) HOMO-LUMO energy splitting in polycyclic aromatic hydrocarbons and their derivatives. Proceedings of the Combustion Institute. 37(1):953–959. https://doi.org/10.1016/j.proci.2018.06.120
Diab N, Morales DM, Andronescu C, Masoud M, Schuhmann W (2019) A sensitive and selective graphene/cobalt tetrasulfonated phthalocyanine sensor for detection of dopamine. Sens Actuators B 285:17–23. https://doi.org/10.1016/j.snb.2019.01.022
Farahmand S, Ghiaci M, Razavizadeh JS (2019) Copper phthalocyanine as an efficient and reusable heterogeneous catalyst for direct hydroxylation of benzene to phenol under mild conditions. Inorg. Chim Acta 484:174–179. https://doi.org/10.1016/j.ica.2018.08.051
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam MJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Revision D.01. Gaussian, Inc., Wallingford CT
Gerasymchuk Y, Guzik M, Lisiecki R, Sobczyk M, Jański J, Koll A, Boulon G, Legendziewicz J (2018) Photophysical properties and ab initio HF and DFT calculations of the structure and spectroscopy of axially chloro substituted Yb(III) monophthalocyanines in different systems. J. Luminescence 193:84–89. https://doi.org/10.1016/j.jlumin.2017.09.002
Güngördü Solğun D, Horoz S, Agırtas MS (2019) Synthesis of novel tetra (4-tritylphenoxy) substituted metallophthalocyanines and investigation of their aggregation, photovoltaic, solar cell properties. Inorg. Nano-Metal Chem. 48(10):508–514. https://doi.org/10.1080/24701556.2019.1572624
Hakiri R, Ameur I, Abid S, Derbel N (2018) Synthesis, X-ray structural, Hirshfeld surface analysis, FTIR, MEP and NBO analysis using DFT study of a 4-chlorobenzylammonium nitrate (C7ClH9N) + (NO3). J. Mol. Structure 1164:486–492. https://doi.org/10.1016/j.molstruc.2018.03.068
Halim SA, Khalil AK (2017) TD-DFT calculations, NBO analysis and electronic absorption spectra of some thiazolo[3,2-a]pyridine derivatives. J. Mol. Structure. 1147:651–667. https://doi.org/10.1016/j.molstruc.2017.06.098
Hodgkinson N, Kruger CA, Mokwena M, Abrahamse H (2017) Cervical cancer cells (HeLa) response to photodynamic therapy using a zinc phthalocyanine photosensitizer. J. Photochem Photobiol B 177:32–38. https://doi.org/10.1016/j.jphotobiol.2017
Kluson P, Drobek M, Kalaji A, Zarubova S, Krysa J, Rakusan J (2008) Singlet oxygen photogeneration efficiencies of a series of phthalocyanines in well-defined spectral regions. J. Photochemistry Photobiology A 199:267–273. https://doi.org/10.1016/j.jphotochem.2008.06.003
Krishnan MM, Baskaran S, Arumugham MN (2017) Photochemical and DFT/TD-DFT study of trifluoroethoxy substituted asymmetric metal-free and copper (II) phthalocyanines. J. Fluorine Chem. 202:1–8. https://doi.org/10.1016/j.jfluchem.2017.08.011
Laajimi M, Chaabene M, Kahouech MS, Chaabane RB, Jamoussi B, Gallot-Lavallee O (2019) Experimental and theoretical investigation on the effect of a spacer addition in zinc phthalocyanine-based anthracene properties. Physica B: Condensed Matter 565:48–60. https://doi.org/10.1016/j.physsb.2019.04.023
Li X, Zheng B-D, Peng X-H, Li S-Z, Ying J-W, Zhao Y, Huang J-D, Yoon J (2019) Phthalocyanines as medicinal photosensitizers: Developments in the last five years. Coord. Chem. Reviews. 379:147–160. https://doi.org/10.1016/j.ccr.2017.08.003
Lin H, Chen J, Zhang Y, Ulla A, Liu J, Lin F, Jiang L, Huang M (2018) Enhanced anti-microbial effect through cationization of a mono-triazatricyclodecane substituted asymmetric phthalocyanine. J. Inorg. Biochem. 189:192–198. https://doi.org/10.1016/j.jinorgbio.2018
Lv F, He X, Wu L, Liu T (2013) Lactose substituted zinc phthalocyanine: A near infrared fluorescence imaging probe for liver cancer targeting. Bioorg. Medicinal Chem. Lett. 23:1878–1882. https://doi.org/10.1016/j.bmcl.2012.12.103
Makhseed S, Bumajdad A, Ghanem B, Msayib K, McKeown NB (2004) Macrodiscotic liquid crystals derived from planar phthalocyanine oligomers. Tetrahedron Lett. 45:4865–4868. https://doi.org/10.1016/j.tetlet.2004.04.151
Nečedová MM, Martinická A, Magdolen P, Novakova V, Zahradník P (2017) Phthalocyanine-triphenylamine dyads: Synthesis, electrochemical, spectral and DFT study. Dyes Pigments 141:448–456. https://doi.org/10.1016/j.dyepig.2017.02.025
Nesterov DS, Nesterova OV, Kopylovich MN, Pombeiro AJL (2018) Pronounced retention of stereoconfiguration upon sp3 C-H bonds hydroxylation of dimethylcyclohexanes and decahydronaphthalenes with m-CPBA oxidant and a Co-phthalocyanine catalyst. Molecular Catalysis 459:8–15. https://doi.org/10.1016/j.mcat.2018.08.009
Prabhaharan M, Prabakaran AR, Gunasekaran S, Srinivasan S (2015) DFT studies on vibrational spectra, HOMO–LUMO, NBO and thermodynamic function analysis of cyanuric flüoride. Spectrochim. Acta A 136:494–503. https://doi.org/10.1016/j.saa.2014.09.062
Ravaei I, Haghighat M, Azami SM (2019) A DFT, AIM and NBO study of isoniazid drug delivery by MgO nanocage. Applied Surface Science. 469:103–112. https://doi.org/10.1016/j.apsusc.2018.11.005
Saka ET, Durmus M, Kantekin H (2011) Solvent and central metal effects on the photophysical and photochemical properties of 4-benzyloxybenzoxy substituted phthalocyanines. J. Organomet. Chem. 696:913–924. https://doi.org/10.1016/j.jorganchem.2010.10.024
Savelyev MS, Gerasimenko IY, Podgaetskii VM, Tereshchenko SA, Selishchev SV, Tolbin AY (2019) Conjugates of thermally stable phthalocyanine J-type dimers with single-walled carbon nanotubes for enhanced optical limiting applications. Opt. Laser Technol 117:272–279. https://doi.org/10.1016/j.optlastec.2019.04.036
Sayin K, Karakaş D (2015) Structural, spectral, NLO and MEP analysis of the [MgO2Ti2(OPri)6], [MgO2Ti2(OPri)2(acac)4] and [MgO2Ti2(OPri)2(bzac)4] by DFT method. Spectrochim. Acta A 144:176–182. https://doi.org/10.1016/j.saa.2015.02.086
Thimiopoulos A, Vogiatzi A, Simandiras ED, Mousdis GA, Psaroudakis N (2019) Synthesis, characterization and DFT analysis of new phthalocyanine complexes containing sulfur rich substituents. Inorg. Chim. Acta. 488:170–181. https://doi.org/10.1016/j.ica.2019.01.010
Weijer R, Broekgaarden M, Kos M, Vught R, Rauws EAJ, Breukink E, Gulik TM, Stormd G, Heger M (2015) Enhancing photodynamic therapy of refractory solid cancers: Combining second-generation photosensitizers with multi-targeted liposomal delivery. J. Photochemistry Photobiology C 23:103–131. https://doi.org/10.1016/j.jphotochemrev.2015.05.002
Wong RCH, Lo P-C, Ng DKP (2019) Stimuli responsive phthalocyanine-based fluorescent probes and photosensitizers. Coord. Chem. Reviews 379:30–46. https://doi.org/10.1016/j.ccr.2017
Yankova R, Genieva S, Dimitrova G (2017) Molecular structure, vibrational, HOMO-LUMO, MEP and NBO analysis of hafnium selenite. J. Mol. Structure. 1141:668–677. https://doi.org/10.1016/j.molstruc.2017.04.004
Zhao H, Yang Y, Shu X, Wang Y, Wu S, Ran Q, Liu J (2018) The binding of calcium ion with different groups of superplasticizers studied by three DFT methods, B3LYP, M06-2X and M06. Computational Materials Science. 152:43–50. https://doi.org/10.1016/j.commatsci.2018.05.034
Acknowledgements
This study was supported by the Scientific Research Projects Unit of Van Yuzuncu Yıl University (FDK-2019-8105).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Solğun, D.G., Keskin, M.S., yıldıko, Ü. et al. DFT analysis and electronic properties, and synthesis of tetra (9-phenyl-9H-xanthen-9-yl) oxy peripheral-substituted zinc phthalocyanine. Chem. Pap. 74, 2389–2401 (2020). https://doi.org/10.1007/s11696-020-01087-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01087-4