Abstract
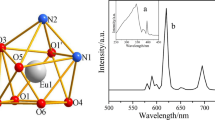
Two new complexes, [M(4-CNB)2(DENA)2(H2O)2] (where M: Co and Mn, 4-CNB = 4-cyanobenzoate and DENA = N,N’-diethylnicotinamide), were synthesized and characterized using different techniques (elemental analysis, FT-IR Spectroscopy, single-crystal X-ray diffraction, and TGA/DTA analysis). Looking at the crystal structure of the complexes, the metal atoms are coordinated by two nitrogen atoms from two DENA ligands, two carboxyl oxygen atoms from two 4-cyanobenzoate anions, and two oxygen atoms from two water molecules, and the complexes have distorted octahedral geometry around the metal atom center. Both complexes were crystallized with the P-1 space group in the triclinic system. The linear absorption and emission features of the complexes were recorded by UV–Vis and fluorescence spectrophotometers, respectively. Linear absorption and emission results of the complexes showed that [Mn(4-CNB)2(DENA)2(H2O)2] complex can be used as optical filter below 400 nm wavelength when [Co(4-CNB)2(DENA)2(H2O)2] complex has a good application potential among the fluorescent materials, because of its high emission intensity. In addition, the Hirshfeld surfaces analyses of complexes have been investigated in detail.









Similar content being viewed by others
References
Akduran N, Necefoglu H, Aydogdu O, Hokelek T (2016a) Crystal structure of catena-poly[[aquabis(4-cyanobenzoato-kappa O)copper(II)]-mu-N, N-diethyl-nicotinamide-kappa N-2(1):O]. Acta Crystallogr Sect E Crystallogr Commun 72:1183. https://doi.org/10.1107/S205698901601183X
Akduran N, Necefoglu H, Aydogdu O, Hokelek T (2016b) Crystal structure of trans-diaquabis(4-cyanobenzoato-jO) bis(N, N-diethylnicotinamide-kappa N)zinc(II). Acta Crystallogr Sect E-Crystallogr Commun 72:1374. https://doi.org/10.1107/S2056989016013815
Akduran N, Sertcelik M, Aydogdu O et al (2016c) Crystal structure of trans-diaquabis(4-cyanobenzoato-kappa O)bis(N, N-diethylnicotinamide-kappa N)cadmium. Acta Crystallogr Sect E-Crystallogr Commun 72:1827. https://doi.org/10.1107/S2056989016018247
Allendorf MD, Bauer CA, Bhakta RK, Houk RJT (2009) Luminescent metal–organic frameworks. Chem Soc Rev 38:1330–1352. https://doi.org/10.1039/B802352M
Altürk S, Tamer Ö, Avcı D, Atalay Y (2015) Synthesis, spectroscopic characterization, second and third-order nonlinear optical properties, and DFT calculations of a novel Mn(II) complex. J Organomet Chem 797:110–119. https://doi.org/10.1016/j.jorganchem.2015.08.014
Askin GS, Necefoglu H, Nayir GY et al (2015) Crystal structure of trans-diaquabis(4-cyanobenzoato-kappa O)bis(nicotinamide-kappa N-1)cobalt(II). Acta Crystallogr Sect E-Crystallogr Commun 71:561. https://doi.org/10.1107/S2056989015008270
Basabe-Desmonts L, Reinhoudt DN, Crego-Calama M (2007) Design of fluorescent materials for chemical sensing. Chem Soc Rev 36:993–1017. https://doi.org/10.1039/b609548h
Baum J, Mcclure D (1979) Ultraviolet transitions of benzoic-acid. 1. Interpretation of the singlet absorption-spectrum. J Am Chem Soc 101:2335–2339. https://doi.org/10.1021/ja00503a016
Bernstein J, Davis RE, Shimoni L, Chang N-L (1995) Patterns in hydrogen bonding: functionality and graph set analysis in crystals. Angew Chem Int Ed Engl 34:1555–1573. https://doi.org/10.1002/anie.199515551
Bushma M, Lukienko P (1985) Interaction invitro of nicotinamide and diethylnicotinamide (cordiamine) with enzymes of rat-liver microsomal hydroxylating systems. Farmakol Toksikol 48:97–100
Caron T, Guillemot M, Veignal F et al (2009) Optical sensor for the detection of explosives: example of a fluorescent material. In: Pardo M, Sberveglieri G (eds) Olfaction and electronic nose, proceedings. American Institute of Physics, Melville, p 83
Coronado E, Espallargas GM (2013) Dynamic magnetic MOFs. Chem Soc Rev 42:1525–1539. https://doi.org/10.1039/C2CS35278H
Das K, Datta A, Massera C, Sinha C (2019) Structural diversity, topology and luminescent properties of a two-dimensional Cd(II) coordination polymer incorporating 4,4′-dipyridyl and 4,4′-sulfonyldibenzoic acid. J Mol Struct 1179:618–622. https://doi.org/10.1016/j.molstruc.2018.11.051
Farrugia LJ (2012) WinGX and ORTEP for windows: an update. J Appl Crystallogr 45:849–854. https://doi.org/10.1107/S0021889812029111
Findoráková L, Győryová K, Melník M et al (2010) Preparation, thermal decomposition, and crystal structure of Zn(II) 2-chlorobenzoate complex with nicotinamide. J Coord Chem 63:3348–3355. https://doi.org/10.1080/00958972.2010.512083
Fujita M, Kwon YJ, Washizu S, Ogura K (1994) Preparation, clathration ability, and catalysis of a two-dimensional square network material composed of cadmium(II) and 4,4′-bipyridine. J Am Chem Soc 116:1151–1152. https://doi.org/10.1021/ja00082a055
Ghosh S, Ganguly A, Bhattacharyya A et al (2016) Selective chromo-fluorogenic molecular sensor for dual channel recognition of Cu2+ and F−: effect of functional group on selectivity. RSC Adv 6:67693–67700. https://doi.org/10.1039/C6RA09877K
Guo-Qing L, Yan L, Wen-Qiang Z et al (2007) One-dimensional supramolecular chain based on a dinuclear terbium(III) 4-cyanobenzoate complex. Chin J Struct Chem 26:805–810
Hathwar VR, Sist M, Jørgensen MRV et al (2015) Quantitative analysis of intermolecular interactions in orthorhombic rubrene. IUCr J 2:563–574. https://doi.org/10.1107/S2052252515012130
Hirshfeld FL (1977) Bonded-atom fragments for describing molecular charge densities. Theor Chim Acta 44:129–138. https://doi.org/10.1007/BF00549096
Jayatilaka D, Grimwood DJ, Lee A et al (2005) TONTO—a system for computational chemistry. Available at: http://hirshfeldsurface.net/
Kanda Y, Shimada R, Takenoshita Y (1963) The phosphorescence spectrum of benzoic acid, methyl benzoate and benzamide at 90°K. Spectrochim Acta 19:1249–1260. https://doi.org/10.1016/0371-1951(63)80044-2
Kose DA, Toprak E, Avci E et al (2014) Synthesis, spectral, thermal studies of Co(II), Ni(II), Cu(II) and Zn(II)-arginato complexes. crystal structure of monoaquabis(arginato-kappa O, kappa N) copper(II). [Cu(arg)(2)(H2O)]center dot NaNO3. J Chin Chem Soc 61:881–890. https://doi.org/10.1002/jccs.201300351
Köse DA, Sahin O, Büyükgüngör O (2012) Synthesis, spectral, thermal, magnetic and structural study of diaquabis(m-hydroxybenzoato-kO)bis(N,N-diethylnicotinamide-kN)cobalt(II). Eur Chem Bull 1:196–201. https://doi.org/10.17628/ecb.2012.1.196-201
Lakowicz JR (2006) Principles of fluorescence spectroscopy, 3rd edn. Springer, Cham
Li Y, Zheng F-K, Liu X et al (2006) Crystal structures and magnetic and luminescent properties of a series of homodinuclear lanthanide complexes with 4-cyanobenzoic ligand. Inorg Chem 45:6308–6316. https://doi.org/10.1021/ic0602603
Li Y, Li G-Q, Zheng F-K et al (2007) Synthesis, crystal structures and magnetic properties of three new 4-cyanobenzoate complexes. J Mol Struct 842:38–45. https://doi.org/10.1016/j.molstruc.2006.12.013
Mason JA, Veenstra M, Long JR (2013) Evaluating metal–organic frameworks for natural gas storage. Chem Sci 5:32–51. https://doi.org/10.1039/C3SC52633J
McKinnon JJ, Jayatilaka D, Spackman MA (2007) Towards quantitative analysis of intermolecular interactions with Hirshfeld surfaces. Chem Commun Camb Engl (Issue 37):3814–3816
Meunier G, Meunier B, Auclair C et al (1983) Unexpected regiospecific alkylation of the antitumor agent N2-methyl-9-hydroxyellipticinium acetate with N, O or S donors. Tetrahedron Lett 24:365–368. https://doi.org/10.1016/S0040-4039(00)81408-2
Morales-Tapia AA, Colorado-Peralta R, Duarte-Hernandez AM et al (2015) Crystal structure of catena-poly[[[triaqua(4-cyanobenzoato-kappa O)nickel(II)]-mu-4,4′-bipyridine-kappa N-2: N′] 4-cyanobenzoate]. Acta Crystallogr Sect E-Crystallogr Commun 71:M197-U138. https://doi.org/10.1107/S2056989015018344
Muhammad K, Nazir S, Hameed S, Bechtold IH (2016) Mesomorphic and fluorescence properties of methyl 4-(4-alkoxystyryl) benzoates. Liq Cryst 43:863–873. https://doi.org/10.1080/02678292.2016.1148205
Nakamoto K (2006) Infrared and Raman spectra of inorganic and coordination compounds. Wiley, Hoboken, NJ
Ozbek FE, Sertcelik M, Yuksek M et al (2017) Cu(II) and Ni(II) 4-cyanobenzoate complexes with nicotinamide: synthesis, spectral, structural and optical characterization and thermal behavior. J Mol Struct 1150:112–117. https://doi.org/10.1016/j.molstruc.2017.08.074
Roy TG, Hazari SKS, Barua KK et al (2010) Synthesis, characterization and anti-microbial studies of cadmium(II) compounds containing 3,10-C-meso-Me-8[14]ane(C). Crystal and molecular structure of cis-[CdLC(NO3)](NO3). Appl Organomet Chem 24:878–887. https://doi.org/10.1002/aoc.1703
Sheldrick GM (2008) A short history of SHELX. Acta Crystallogr A 64:112–122. https://doi.org/10.1107/S0108767307043930
Smolkova R, Smolko L, Gyoryova K et al (2018) New zinc(II) 3-aminobenzoates with bioactive ligands: synthesis, thermal, spectral and antimicrobial properties. Thermochim Acta 669:1–7. https://doi.org/10.1016/j.tca.2018.09.004
Spackman MA, McKinnon JJ, Jayatilaka D (2008) Electrostatic potentials mapped on Hirshfeld surfaces provide direct insight into intermolecular interactions in crystals. CrystEngComm 10:377–388. https://doi.org/10.1039/B715227B
Szmigiel K, Nentwig M, Oeckler O et al (2018) Crystal structure, vibrational spectroscopic characterization, thermal behavior and antifungal activity of a novel coordination polymer of indole-3-carboxylic acid with cobalt(II) and a comparison with the isostructural Zn(II) complex. Inorg Chem Commun 97:56–62. https://doi.org/10.1016/j.inoche.2018.09.009
Takeshita T, Tsuji K (1974) Electron-spin resonance study of photosensitized radical formation in N-hexane by benzoic-acid. J Phys Chem 78:105–107. https://doi.org/10.1021/j100595a004
Turner MJ, McKinnon JJ, Wolff SK et al (2017) CrystalExplorer17, Univ West Aust. http://hirshfeldsurface.net
Uzun A, Keskin S (2014) Site characteristics in metal organic frameworks for gas adsorption. Prog Surf Sci 89:56–79. https://doi.org/10.1016/j.progsurf.2013.11.001
Venkatesan P, Thamotharan S, Ilangovan A et al (2016) Crystal structure, Hirshfeld surfaces and DFT computation of NLO active (2E)-2-(ethoxycarbonyl)-3-[(1-methoxy-1-oxo-3-phenylpropan-2-yl) amino] prop-2-enoic acid. Spectrochim Acta A Mol Biomol Spectrosc 153:625–636
Yeşilel OZ, Ilker I, Şahin E (2011) One-dimensional Ni(II) and Cu(II) coordination polymers containing syn-syn thiophene-2,5-dicarboxylate and propane-1,3-diamine. J Inorg Organomet Polym Mater 21:103–109. https://doi.org/10.1007/s10904-010-9414-1
Yıldırım T, Köse DA, Avcı E et al (2019) Novel mixed ligand complexes of acesulfame/nicotinamide with some transition metals. Synthesis, crystal structural characterization, and biological properties. J Mol Struct 1176:576–582. https://doi.org/10.1016/j.molstruc.2018.08.099
Zhou Q-K, Li N-Y (2017) A three-dimensional cadmium coordination polymer based on 1,4-bis-(1,2,4-triazol-1-yl)but-2-ene and benzene-1,3,5-tri-carb-oxy-lic acid. Acta Crystallogr Sect C Struct Chem 73:749–753. https://doi.org/10.1107/S2053229617012189
Zhou SY, Li X, Li T et al (2015) A series of heterospin complexes based on lanthanides and pyridine biradicals: synthesis, structure and magnetic properties. RSC Adv 5:17131–17139. https://doi.org/10.1039/C4RA15074K
Zhou X, Liu K, Liu L et al (2019) Two 3-D metal organic frameworks based on pyridine carboxylic acid ligands with magnetism properties and photoluminescence. Sci Adv Mater 11:332–337. https://doi.org/10.1166/sam.2019.3465
Acknowledgements
This research was supported by the Science Research Fund of Kafkas University (Project no: 2016-FM-49).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sertçelik, M., Özbek, F.E., Yüksek, M. et al. Synthesis, spectral, thermal, structural, optical characterization, and Hirshfield surface analysis of N,N’-diethylnicotinamide complexes of Mn(II) and Co(II) 4-cyanobenzoates. Chem. Pap. 74, 2021–2033 (2020). https://doi.org/10.1007/s11696-019-01040-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-019-01040-0