Abstract
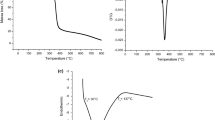
Starch is a polysaccharide that has an important role in food and other industries. Anchote, a local name for “Coccinia abyssinica” is an indigenous tuber crop to Ethiopia supposed to contain considerable amount of starch. This study aims to extract starch from this plant using sodium metabisulfite (0.025, 0.075 and 0.125% w/v), sodium chloride (0.5, 1.0 and 1.5 M) and water. Anchote starch extracted by using 0.075% (w/v) sodium metabisulfite has presented the highest extraction yield (75.56%) on a dry weight basis. The values of moisture content, bulk density and amylose content of anchote starch were recorded as 11.6%, 0.51 g/mL and 32.14%, repectively. The color of anchote starch was measured in L*, a*, b* values and obtained 89.80, 0.025, 3.63, respectively and whiteness was 89.16%. Its granular shapes exhibited polygonal, semi-oval with some round. The swelling power and the solubility of anchote starch were ranged from 2.15 to 10.6 g/g and 0.51–19.15% respectively. Syneresis of anchote starch increased when pastes were stored at low temperatures and decline in paste clarity upon storage. Anchote starch exhibited gelatinization temperature; onset, peak and conclusion (68.94, 74.99 and 84.87 °C) respectively. Pasting properties of anchote starch; peak, trough, breakdown, final and setback viscosities were recorded as 4879 cP, 2101.7 cP, 2777 cP, 5636.1 cP, 3535 cP, respectively. Anchote starch showed type B crystalline pattern with 39.15% relative crystallinity. FTIR spectra of anchote starch bands were almost similar with potato starch. The results indicate that this starch may be an alternative source for the production of industrial products that may require starch.







Similar content being viewed by others
References
F. Zhu, Q. Xie, Physical Modifications of Starch (Springer, Singapore, 2018), pp. 1–14
A.S. Babu, R. Parimalavalli, Food Technol. 38, 48–63 (2014)
A. Wondimu, F. Molla, S.C. Dinda, N. Gebre-Samuel, E.T. Tadese, J. Drug Deliv. Ther. 4, 1–6 (2014). https://doi.org/10.22270/jddt.v4i3.822
D.H. Kringel, S.L.M. El Halal, E.R. da Zavareze, A.R.G. Dias, Starch/Staerke (Wiley, New York, 2020).
H. Fekadu, J. Food Process. Technol. (2013). https://doi.org/10.4172/2157-7110.1000249
H. Fekadu, Food Sci. Qual. Manag. 26, 25–39 (2014)
A. Bekele, T. Feyissa, K. Tesfaye, Genet. Resour. Crop Evol. 61, 707–719 (2014)
N.G. Ayana, Agric. Sci. Res. J. 9, 1–11 (2019)
R. Kale, D. Shere, J. Pharmacogn. Phytochem. 6, 223–227 (2017)
C.K. Riley, A.O. Wheatley, H.N. Asemota, African J. Biotechnol. 5, 1528–1536 (2006). https://doi.org/10.4314/ajb.v5i17.43151
H.A.M. Wickramasinghe, S. Takigawa, C. Matsuura-Endo, H. Yamauchi, T. Noda, Food Chem. 112, 98–103 (2009). https://doi.org/10.1016/j.foodchem.2008.05.046
AOAC, Official Method of Analysis, 17th edn. (AOAC, Washington, DC, 2000)
C. Sudheesh, K.V. Sunooj, J. George, S. Kumar, V.A. Sajeevkumar, J. Food Meas. Charact. 13, 1020–1030 (2019). https://doi.org/10.1007/s11694-018-0016-x
H.M. Thao, A. Noomhorm, J. Food Process. Technol. (2011). https://doi.org/10.4172/2157-7110.1000105
M. Piecyk, E. Worobiej, R. Wołosiak, B. Drużyńska, E. Ostrowska-Ligęza, J. Food Meas. Charact. 13, 848–856 (2019). https://doi.org/10.1007/s11694-018-9997-8
P. Vithu, S.K. Dash, K. Rayaguru, M.K. Panda, M. Nedunchezhiyan, J. Food Meas. Charact. 14, 1520–1532 (2020). https://doi.org/10.1007/s11694-020-00401-8
O.K. Abegunde, T. Mu, J. Chen, F. Deng, Food Hydrocolloids 33, 169–177 (2013). https://doi.org/10.1016/j.foodhyd.2013.03.005
A. Gani, S.S. Haq, F.A. Masoodi, A.A. Broadway, A. Gani, Brazilian Arch. Biol. Technol. 53, 731–740 (2010). https://doi.org/10.1590/S1516-89132010000300030
D.P.R. Ascheri, C.C. Morais, E.R. Asquieri, C.W.P. Carvalho, J.L.R. Ascheri, Semin. Agrar. 35, 787–800 (2014). https://doi.org/10.5433/1679-0359.2014v35n2p787
B. Noora, C. Sudheesh, N. Sangeetha, K.V. Sunooj, J. Food Meas. Charact. 13, 2147–2156 (2019). https://doi.org/10.1007/s11694-019-00135-2
H. Huang, Q. Jiang, Y. Chen, X. Li, X. Mao, X. Chen, L. Huang, W. Gao, Food Hydrocolloids 55, 244–253 (2016). https://doi.org/10.1016/j.foodhyd.2015.11.016
H. Julianti, R. Ridwansyah, E. Yusraini, SciTePress 1, 37–41 (2018)
R.A.I. Chandra, S.A.N. Hasanah, R. Agustina, Int. J. Chem. Eng. Appl. 7, 89–95 (2016). https://doi.org/10.7763/ijcea.2016.v7.549
S.M. Chisenga, T.S. Workneh, G. Bultosa, M. Laing, AIMS Agric. Food 4, 939–966 (2019). https://doi.org/10.3934/agrfood.2019.4.939
M.M. Aly, N.M. Gumgumjee, African J. Biotechnol. 10, 12058–12063 (2011). https://doi.org/10.4314/ajb.v10i56
M.C. Iheagwara, J. Food Process. Technol. 04, 1–6 (2012). https://doi.org/10.4172/2157-7110.1000198
A. Assefa, A. Belete, T. Gebre-mariam, Ethiop. J. Sci. 39, 11–20 (2016)
A.A. Adebowale, L.O. Sanni, E.L. Fadahunsi, African J. Root Tuber Crop. 9, 6–10 (2011)
H.A. Hassan, A.I. Mustafa, A.R. Ahmed, Adv. J. Food Sci. Technol. 5, 976–981 (2013). https://doi.org/10.19026/ajfst.5.3335
M.I. Akpata, P.I. Akubor, Plant Foods Hum. Nutr. 54, 353–362 (1999). https://doi.org/10.1023/A:1008153228280
T. Krishnakumar, M.S. Sajeev, Int. J. Curr. Microbiol. Appl. Sci. 7, 3122–3135 (2018). https://doi.org/10.20546/ijcmas.2018.710.362
J. Kim, C. Ren, M. Shin, Starch/Staerke 65, 923–930 (2013). https://doi.org/10.1002/star.201200217
T. Zhu, D.S. Jackson, R.L. Wehling, B. Geera, Cereal Chem. 85, 51–58 (2008). https://doi.org/10.1094/CCHEM-85-1-0051
T. Gebre-Mariam, P.C. Schmidt, Starch/Staerke 48, 208–214 (1996). https://doi.org/10.1002/star.19960480603
O.O. Oladayo, Q.C. Umunna, O.S. Joseph, W. Oluwasegun, Songklanakarin. J. Sci. Technol. 38, 349–355 (2016). https://doi.org/10.14456/sjst-psu.2016.47
P.F. Builders, P.A. Anwunobi, C.C. Mbah, M.U. Adikwu, AAPS PharmSciTech 14, 818–827 (2013). https://doi.org/10.1208/s12249-013-9968-7
J.A. Sanchez-González, C. Echeverria, L. Lescano, G. Linares, H.L. Arteaga-Miñano, J. Soriano-Colchado, G. Barraza-Jáuregui, Sci. Agropecu. 10, 63–71 (2019). https://doi.org/10.17268/sci.agropecu.2019.01.07
M.B.K. Niazi, A.A. Broekhuis, J. Appl. Polym. Sci. 126, E143–E153 (2012). https://doi.org/10.1002/app.36551
X. Yu, J. Zhang, A. Li, Z. Wang, F. Xiong, J. Food Sci. 80, C1661–C1669 (2015). https://doi.org/10.1111/1750-3841.12969
M.F. Zuluaga, Y. Baena, C.E. Mora, L.F.P. D’León, Starch/Staerke 59, 307–317 (2007). https://doi.org/10.1002/star.200600516
R.V. Manek, P.F. Builders, W.M. Kolling, M. Emeje, O.O. Kunle, AAPS PharmSciTech 13, 379–388 (2012). https://doi.org/10.1208/s12249-012-9761-z
H. Hauwa, LaminuHH, A.S. Falmata, B.P. Bintu, G. Maryam Babakura Chamba, M. Babagana, S. Modu, EC Nutr. 3, 572–588 (2016)
L. Wang, B. Xie, J. Shi, S. Xue, Q. Deng, Y. Wei, B. Tian, Food Hydrocolloids 24, 208–216 (2010). https://doi.org/10.1016/j.foodhyd.2009.09.007
A. Ali, T.A. Wani, I.A. Wani, F.A. Masoodi, J. Saudi Soc. Agric. Sci. 15, 75–82 (2016). https://doi.org/10.1016/j.jssas.2014.04.002
J. Jane, Y.Y. Chen, L.F. Lee, A.E. McPherson, K.S. Wong, M. Radosavljevic, T. Kasemsuwan, Cereal Chem. 76, 629–637 (1999). https://doi.org/10.1094/CCHEM.1999.76.5.629
H. Atrous, N. Benbettaieb, F. Hosni, S. Danthine, C. Blecker, H. Attia, D. Ghorbel, Int. J. Biol. Macromol. 80, 64–76 (2015). https://doi.org/10.1016/j.ijbiomac.2015.06.014
N. Singh, A. Kaur, K. Shevkani, R. Ezekiel, P. Kaur, N. Isono, T. Noda, Starch/Staerke 70, 1700130 (2018). https://doi.org/10.1002/star.201700130
O.A. Adekunle, A.A. Mary, Int. J. Food Stud. 3, 175–185 (2014). https://doi.org/10.7455/ijfs/3.2.2014.a4
O.F. Ekunseitan, A.O. Obadina, O.P. Sobukola, A.M. Omemu, M.O. Adegunwa, O.E. Kajihausa, A.R.A. Adebowale, S.A. Sanni, L.O. Sanni, T. Keith, J. Food Process. Preserv. 41, e13150 (2017). https://doi.org/10.1111/jfpp.13150
W. Zhang, X. Liu, Q. Wang, H. Zhang, M. Li, B. Song, Z. Zhao, Int. J. Biol. Macromol. 117, 467–472 (2018). https://doi.org/10.1016/j.ijbiomac.2018.05.131
K.S. Sandhu, N. Singh, Food Chem. 101, 1499–1507 (2007). https://doi.org/10.1016/j.foodchem.2006.01.060
L. Zhang, Y. Zhao, W. Hu, J.Y. Qian, X.L. Ding, C.R. Guan, Y.Q. Lu, Y. Cao, Carbohydr. Polym. 200, 400–407 (2018). https://doi.org/10.1016/j.carbpol.2018.08.022
K.N. Jan, P.S. Panesar, S. Singh, J. Food Meas. Charact. 11, 1919–1927 (2017). https://doi.org/10.1007/s11694-017-9574-6
P.R. Correia, M.C. Nunes, M.L. Beirão-da-Costa, Food Hydrocolloids 27, 256–263 (2012). https://doi.org/10.1016/j.foodhyd.2011.05.010
R.L. Zapata-Luna, T. Ayora-Talavera, N. Pacheco, E. García-Márquez, H. Espinosa-Andrews, Á. Ku-González, J. Ruiz-Ruiz, J.C. Cuevas-Bernardino, J. Food Meas. Charact. (2020). https://doi.org/10.1007/s11694-020-00739-z
Acknowledgements
The authors are thankful to the Department of Food Process Engineering and Central Research Laboratory of Addis Ababa Science and Technology University for providing laboratory and instrumental facilities. The authors also acknowledge Debre Zeit Agricultural Research Center and Adama Science and Technology University.
Funding
Ministry of Science and Higher Education (MOSHE), Arba Minch University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tessema, A., Admassu, H. Extraction and characterization of starch from anchote (Coccinia abyssinica): physico-chemical, functional, morphological and crystalline properties. Food Measure 15, 3096–3110 (2021). https://doi.org/10.1007/s11694-021-00885-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-021-00885-y