Abstract
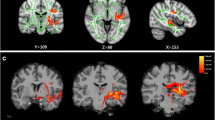
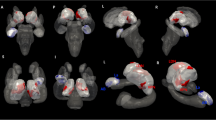
Idiopathic tinnitus can cause significant auditory-related brain structural and functional changes in patients. However, changes in patterns of the lateralization effects in idiopathic tinnitus have yet to be established, especially on white matter (WM) reorganization. In this study, we studied 19 left-sided and 19 right-sided idiopathic tinnitus (LSIT, RSIT) patients and 19 healthy controls (HCs). We combined applied voxel-based morphometry (VBM) and tract-based spatial statistics (TBSS) analyses to investigate altered features of the auditory-related brain WM. We also conducted correlation analyses between the clinical variables and WM changes in the patients. Compared with the HCs, both sided tinnitus patients showed significant auditory-related brain WM alterations. More interestingly, the LSIT patients demonstrated a greater decrease in white matter volume (WMV) in the right medial superior frontal gyrus (SFG) than the RSIT; meanwhile, we also found that compared with the RSIT group, the LSIT group showed significantly increased fractional anisotropy (FA) in the body of the corpus callosum (CC), left cingulum, and right superior longitudinal fasciculus (SLF) and decreased mean diffusivity (MD) in the body of CC. Moreover, relative to the RSIT group, the LSIT group also exhibited increases in WM axial diffusivity (AD) in the left SLF, left cingulum, right middle cerebellar peduncle (MCP), left thalamus, and bilateral forceps major (FM) and decreases in radial diffusivity (RD) in the genu of CC. Additionally, the FA value of the right SLF was closely associated with tinnitus severity in the LSIT. Our study suggests that lateralization has a significant effect on WM reorganization in patients with idiopathic tinnitus; in particular, LSIT patients may experience more severe and widespread alterations in WMV and WM microstructure than the RSIT group, and all these changes are indirectly auditory related. These findings provide new useful information that can lead to a better understanding of the tinnitus mechanisms.




Similar content being viewed by others
Data availability
The datasets generated for this study are available on request to the corresponding author.
Abbreviations
- LSIT:
-
left-sided idiopathic tinnitus
- RSIT:
-
right-sided idiopathic tinnitus
- HCs:
-
healthy controls
- VBM:
-
voxel-based morphometry
- DTI:
-
diffusion tensor imaging
- TBSS:
-
tract-based spatial statistics
- WMV:
-
white matter volume
- CSF:
-
cerebrospinal fluid
- THI:
-
tinnitus handicap inventory
- MNI:
-
Montreal Neurological Institute
- WMV:
-
white matter volume
- WM:
-
white matter
- FA:
-
fractional anisotropy
- MD:
-
mean diffusivity
- AD:
-
axial diffusivity
- RD:
-
radial diffusivity
- SFG:
-
superior frontal gyrus
- CC:
-
corpus callosum
- MCP:
-
middle cerebellar peduncle
- SLF:
-
superior longitudinal fasciculus
References
Ashburner, J. (2007). A fast diffeomorphic image registration algorithm. Neuroimage, 38, 95–113.
Baguley, D., McFerran, D., & Hall, D. (2013). Tinnitus. Lancet, 382, 1600–1607.
Benson, R. R., Gattu, R., & Cacace, A. T. (2014). Left hemisphere fractional anisotropy increase in noise-induced tinnitus: A diffusion tensor imaging (DTI) study of white matter tracts in the brain. Hearing Research, 309, 8–16.
Bledsoe, I. O., Stebbins, G. T., Merkitch, D., & Goldman, J. G. (2018). White matter abnormalities in the corpus callosum with cognitive impairment in Parkinson disease. Neurology, 91, e2244–e2255.
Boyen, K., Langers, D. R. M., de Kleine, E., & van Dijk, P. (2013). Gray matter in the brain: Differences associated with tinnitus and hearing loss. Hearing Research, 295, 67–78.
Catani, M., Howard, R. J., Pajevic, S., & Jones, D. K. (2002). Virtual in vivo interactive dissection of white matter fasciculi in the human brain. Neuroimage, 17, 77–94.
Çavdar, S., Özgür, M., Kuvvet, Y., Bay, H., & Aydogmus, E. (2018). Cortical, subcortical and brain stem connections of the cerebellum via the superior and middle cerebellar peduncle in the rat. The Journal of Integrative Neuroscience, 17, 609–618.
Chen, Q., Wang, Z., Lv, H., Zhao, P., Yang, Z., Gong, S., & Wang, Z. (2020). Reorganization of brain white matter in persistent idiopathic tinnitus patients without hearing loss: evidence from baseline data. Frontiers in Neuroscience-Switz, 4, 591.
Chen, Y., Xia, W., Luo, B., Muthaiah, V. P. K., Xiong, Z., Zhang, J., Wang, J., Salvi, R., & Teng, G. (2015). Frequency-specific alternations in the amplitude of low-frequency fluctuations in chronic tinnitus. Frontiers in Neural Circuits, 9, 67.
Crippa, A., Lanting, C., Dijk, P. V., & Roerdink, J. B. T. M. (2010). A diffusion tensor imaging study on the auditory system and tinnitus. The Open Neuroimaging Journal, 4, 16–25.
Diesch, E. (2012). Structural changes of the corpus callosum in tinnitus. Frontiers in Systems Neuroscience, 6, 17.
Dong, C., Zhao, P., Yang, J., Liu, Z., & Wang, Z. (2015). Incidence of vascular anomalies and variants associated with unilateral venous pulsatile tinnitus in 242 patients based on dual-phase contrast-enhanced computed tomography. Chinese Medical Journal-Peking, 128, 581–585.
Eggermont, J. J., & Roberts, L. E. (2004). The neuroscience of tinnitus. Trends in Neurosciences, 27, 676–682.
Elgoyhen, A. B., Langguth, B., De Ridder, D., & Vanneste, S. (2015). Tinnitus: perspectives from human neuroimaging. Nature Reviews Neuroscience, 16, 632–642.
Fabri, M., Pierpaoli, C., Barbaresi, P., & Polonara, G. (2014). Functional topography of the corpus callosum investigated by DTI and fMRI. World Journal of Radiology, 6, 895–906.
Han, L., Na, Z., Chunli, L., Yuchen, C., Pengfei, Z., Hao, W., Xu, C., Peng, Z., Zheng, W., Zhenghan, Y., Shusheng, G., & Zhenchang, W. (2019). Baseline functional connectivity features of neural network nodes can predict improvement after sound therapy through adjusted narrow band noise in tinnitus patients. Frontiers in Neuroscience, 13, 614.
Han, L., Yawen, L., Hao, W., Chunli, L., Pengfei, Z., Zhengyu, Z., Zhaodi, W., Zhenghan, Y., Shusheng, G., & Zhenchang, W. (2019). Effects of sound therapy on resting-state functional brain networks in patients with tinnitus: A graph-theoretical-based study. Journal of Magnetic Resonance Imaging, 50, 1731–1741.
Husain, F. T., Medina, R. E., Davis, C. W., Szymko-Bennett, Y., Simonyan, K., Pajor, N. M., & Horwitz, B. (2011). Neuroanatomical changes due to hearing loss and chronic tinnitus: A combined VBM and DTI study. Brain Research, 1369, 74–88.
Husain, F. T., Zimmerman, B., Tai, Y., Finnegan, M. K., Kay, E., Khan, F., Menard, C., & Gobin, R. L. (2019). Assessing mindfulness-based cognitive therapy intervention for tinnitus using behavioural measures and structural MRI: a pilot study. INT J AUDIOL, 58, 889–901.
Kan, Y., Wang, W., Zhang, S., Ma, H., Wang, Z., & Yang, J. (2019). Neural metabolic activity in idiopathic tinnitus patients after repetitive transcranial magnetic stimulation. The World Journal of Clinical Cases, 7, 1582–1590.
Krick, C. M., Grapp, M., Daneshvar-Talebi, J., Reith, W., Plinkert, P. K., & Bolay, H. V. (2015). Cortical reorganization in recent-onset tinnitus patients by the Heidelberg Model of Music Therapy. Frontiers in Neuroscience-Switz, 9, 49.
Lansley, J. A., Tucker, W., Eriksen, M. R., Riordan-Eva, P., & Connor, S. E. J. (2017). Sigmoid sinus diverticulum, dehiscence, and venous sinus stenosis: potential causes of pulsatile tinnitus in patients with idiopathic intracranial hypertension? American Journal of Neuroradiology, 38, 1783–1788.
Lanting, C. P., De Kleine, E., Bartels, H., & Van Dijk, P. (2008). Functional imaging of unilateral tinnitus using fMRI. Acta Oto-Laryngologica, 128, 415–421.
Lanting, C. P., de Kleine, E., Langers, D. R. M., & van Dijk, P. (2014). Unilateral tinnitus: changes in connectivity and response lateralization measured with fMRI. PloS One, 9, e110704.
Leaver, A. M., Renier, L., Chevillet, M. A., Morgan, S., Kim, H. J., & Rauschecker, J. P. (2011). Dysregulation of limbic and auditory networks in tinnitus. Neuron, 69, 33–43.
Lee, S., Song, S., Park, S. J., Park, H. G., Choi, B. Y., Koo, J., & Song, J. (2019a). Jugular bulb resurfacing with bone cement for patients with high dehiscent jugular bulb and ipsilateral pulsatile tinnitus. Otology & Neurotology, 40, 192–199.
Lee, S., Rhee, J., Shim, Y. J., Kim, Y., Koo, J., De Ridder, D., Vanneste, S., & Song, J. (2019b). Changes in the resting-state cortical oscillatory activity 6 months after modified tinnitus retraining therapy. Frontiers in Neuroscienc-Switz, 13, 1123.
Liu, Y., Lv, H., Zhao, P., Liu, Z., Chen, W., Gong, S., Wang, Z., & Zhu, J. (2018). Neuroanatomical alterations in patients with early stage of unilateral pulsatile tinnitus: a voxel-based morphometry study. Neural Plasticity, 2018, 1–7.
Lv, H., Liu, C., Wang, Z., Zhao, P., Cheng, X., Yang, Z., Gong, S., & Wang, Z. (2020). Altered functional connectivity of the thalamus in tinnitus patients is correlated with symptom alleviation after sound therapy. Brain Imaging and Behavior, 14(6), 2668-2678
Lv, H., Zhao, P., Liu, Z., Liu, X., Ding, H., Liu, L., Wang, G., Xie, J., Zeng, R., Chen, Y., Yang, Z., Gong, S., & Wang, Z. (2018). Lateralization effects on functional connectivity of the auditory network in patients with unilateral pulsatile tinnitus as detected by functional MRI. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 81, 228–235.
Makris, N., Kennedy, D. N., McInerney, S., Sorensen, A. G., Wang, R., Caviness, V. S., & Pandya, D. N. (2005). Segmentation of subcomponents within the superior longitudinal fascicle in humans: a quantitative, in vivo, DT-MRI study. Cerebral Cortex, 15, 854–869.
Matsuda, Y., Inagawa, T., & Amano, T. (1993). A case of tinnitus and hearing loss after cerebellar hemorrhage. Stroke, 24, 906–908.
Melcher, J. R., Levine, R. A., Bergevin, C., & Norris, B. (2009). The auditory midbrain of people with tinnitus: Abnormal sound-evoked activity revisited. Hearing Research, 257, 63–74.
Mirz, F., Gjedde, A., Sdkilde-Jrgensen, H., & Pedersen, C. B. (2000). Functional brain imaging of tinnitus-like perception induced by aversive auditory stimuli. Neuroreport, 11, 633–637.
Mtui, E., Gruener, G., & Dockery, P. (2016). Fitzgerald’s clinical neuroanatomy and neuroscience. [Electronic Resource] (7th ed.). Elsevier.
Mundada, P., Singh, A., & Lingam, R. K. (2015). CT arteriography and venography in the evaluation of Pulsatile tinnitus with normal otoscopic examination. The Laryngoscope, 125, 979–984.
Newman, C. W., Jacobson, G. P., & Spitzer, J. B. (1996). Development of the tinnitus handicap inventory. Archives of Otolaryngology–Head & Neck Surgery, 122, 143.
Raya, J. G., Horng, A., Dietrich, O., Krasnokutsky, S., Beltran, L. S., Storey, P., Reiser, M. F., Recht, M. P., Sodickson, D. K., & Glaser, C. (2012). Articular cartilage: in vivo diffusion-tensor imaging. Radiology, 262, 550–559.
Ryu, C., Park, M. S., Byun, J. Y., Jahng, G., & Park, S. (2016). White matter integrity associated with clinical symptoms in tinnitus patients: A tract-based spatial statistics study. European Radiology, 26, 2223–2232.
Schmidt, S. A., Carpenter-Thompson, J., & Husain, F. T. (2017). Connectivity of precuneus to the default mode and dorsal attention networks: A possible invariant marker of long-term tinnitus. NeuroImage: Clinical, 16, 196–204.
Schmidt, S. A., Zimmerman, B., Bido Medina, R. O., Carpenter-Thompson, J. R., & Husain, F.T. (2018). Changes in gray and white matter in subgroups within the tinnitus population. BRAIN RES, 1679, 64–74.
Schoeff, S., Nicholas, B., Mukherjee, S., & Kesser, B. W. (2014). Imaging prevalence of sigmoid sinus dehiscence among patients with and without pulsatile tinnitus. Otolaryngology–Head and Neck Surgery, 150, 841–846.
Shelton, A. L., Cornish, K. M., Godler, D., Bui, Q. M., Kolbe, S., & Fielding, J. (2017). White matter microstructure, cognition, and molecular markers in fragile X premutation females. Neurology, 88, 2080–2088.
Shim, Y. J., Bae, Y. J., An, G. S., Lee, K., Kim, Y., Lee, S., Choi, B. Y., Choi, B. S., Kim, J. H., Koo, J., & Song, J. (2019). Involvement of the internal auditory canal in subjects with cochlear otosclerosis. Otology & Neurotology, 40, e186–e190.
Smith, S. M., Jenkinson, M., Johansen-Berg, H., Rueckert, D., Nichols, T. E., Mackay, C. E., Watkins, K. E., Ciccarelli, O., Cader, M. Z., Matthews, P. M., & Behrens, T. E. J. (2006). Tract-based spatial statistics: Voxelwise analysis of multi-subject diffusion data. Neuroimage, 31, 1487–1505.
Vanneste, S., Joos, K., & De Ridder, D. (2012). Prefrontal cortex based sex differences in tinnitus perception: same tinnitus intensity, same tinnitus distress, different mood. PLoS One, 7, e31182.
Xie, X., Liu, Y., Han, X., Liu, P., Qiu, H., Li, J., & Yu, H. (2019). Differences in intrinsic brain abnormalities between patients with left- and right-sided long-term hearing impairment. Frontiers in Neuroscience-Switz, 13, 206
Xue, C., Shi, L., Hui, S. C. N., Wang, D., Lam, T. P., Ip, C. B., Ng, B. K. W., Cheng, J. C. Y., & Chu, W. C. W. (2018). Altered white matter microstructure in the corpus callosum and its cerebral interhemispheric tracts in adolescent idiopathic scoliosis: diffusion tensor imaging analysis. American Journal of Neuroradiology, 39, 1177–1184.
Acknowledgements
The authors thank the patients and healthy volunteers who participated in this study and gave generously of their time.
Funding
This work was supported by Grant No.61801311, 61931013 from the National Natural Science Foundation of China, No. [2015] 160 from Beijing Scholars Program, Grant No. 7182044 from Beijing Natural Science Foundation, No. PX2018001 from Beijing Hospitals Authority, QML20180103 from Beijing Hospitals Authority Youth Programme, No. YYZZ2017B01 from Beijing Friendship Hospital, Capital Medical University, No. 2019M660717 from China Postdoctoral Science Foundation and No. 2020-Z2-023 from Beijing Postdoctoral Research Foundation.
Author information
Authors and Affiliations
Contributions
QC and HL* conducted the research project and conceived the study. QC wrote the manuscript. ZDW/XW/PFZ/ZHY/SSG provided technical and clinical support. HL* and ZCW* reviewed the manuscript and agree to be accountable for all aspects of the work.
Corresponding authors
Ethics declarations
Ethical approval
This study was approved by the Institutional Review Board (IRB) of Beijing Friendship Hospital, Capital Medical University (Beijing, China) (IRB number: 2016-P2-012), and written informed consent was obtained from each subject before the study in accordance with the Declaration of Helsinki.
Consent to participate
All the authors consent to the participation in this study.
Consent to publish
All the authors agreed with the publication of this article.
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, Q., Lv, H., Wang, Z. et al. Lateralization effects in brain white matter reorganization in patients with unilateral idiopathic tinnitus: a preliminary study. Brain Imaging and Behavior 16, 11–21 (2022). https://doi.org/10.1007/s11682-021-00472-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-021-00472-1