Abstract
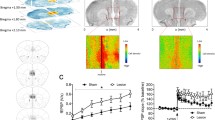
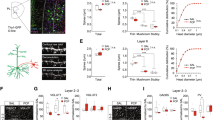
Schizophrenia is a severe psychiatric disease while its etiology and effective treatment are not completely clear. A rat model of schizophrenia was previously established by transfecting EGR3 gene into the hippocampus of rats. This study aimed to investigate the behavioral and cerebral alterations of the schizophrenic model rats and the risperidone effects. Twenty-six rats were divided into 3 groups: schizophrenia model group (E group), risperidone treatment group (T group), and healthy control group (H group). Morris water maze and open field test were used as behavioral tests, resting-state functional magnetic resonance imaging (fMRI) was performed after EGR3 gene transfection and risperidone therapy. Graph analyses were used for examining cerebral alterations of the rats. Behavioral tests demonstrated reduced spatial working memory and exploring unfamiliar space ability in schizophrenic model rats. Graph analyses revealed reduced regional architectures in the olfactory bulb, nucleus accumbens, and pineal gland in group E compared to group H (p < 0.05), while group T showed increased regional architecture in pineal gland compared to group E (p < 0.05). Besides, the regional architectures in the olfactory bulb, nucleus accumbens were lower in group T than group H, while the hippocampus showed increased regional architecture in group T compared to group H (p < 0.05). Schizophrenia induced several regional alterations in the cerebrum while risperidone can reverse part of these alterations. This study lends support for future research on the pathology of schizophrenia and provides new insights on the role of risperidone in schizophrenia.





Similar content being viewed by others
References
Achard, S., & Bullmore, E. (2007). Efficiency and cost of economical brain functional networks. PLoS Computational Biology, 3, e17. https://doi.org/10.1371/journal.pcbi.0030017.
Alexander-Bloch, A. F., Gogtay, N., Meunier, D., Birn, R., Clasen, L., Lalonde, F., et al. (2010). Disrupted modularity and local connectivity of brain functional networks in childhood-onset schizophrenia. Frontiers in Systems Neuroscience, 4, 147. https://doi.org/10.3389/fnsys.2010.00147.
Bade, A. N., Gendelman, H. E., Boska, M. D., & Liu, Y. (2017). Memri is a biomarker defining nicotine-specific neuronal responses in subregions of the rodent brain. American Journal of Translational Research, 9, 601–610.
Bassett, D. S., Bullmore, E., Verchinski, B. A., Mattay, V. S., Weinberger, D. R., & Meyer-Lindenberg, A. (2008). Hierarchical organization of human cortical networks in health and schizophrenia. The Journal of Neuroscience, 28, 9239–9248. https://doi.org/10.1523/JNEUROSCI.1929-08.2008.
Bassett, D. S., Bullmore, E. T., Meyer-Lindenberg, A., Apud, J. A., Weinberger, D. R., & Coppola, R. (2009). Cognitive fitness of costefficient brain functional networks. Proceedings of the National Academy of Sciences of the United States of America, 106, 11747–11752. https://doi.org/10.1073/pnas.0903641106.
Bustillo, J. R., Rowland, L. M., Mullins, P., Jung, R., Chen, H., Qualls, C., et al. (2010). 1 h-mrs at 4 T in minimally treated early schizophrenia. Molecular Psychiatry, 15, 629–636. https://doi.org/10.1038/mp.2009.121.
Deleon, A., Patel, N. C., & Crismon, M. L. (2004). Aripiprazole: a comprehensive review of its pharmacology, clinical efficacy, and tolerability. Clinical Therapeutics, 26, 649–666. https://doi.org/10.1016/s0149-2918(04)90066-5.
Eldredge, L. C., Gao, X. M., Quach, D. H., Li, L., Han, X., Lomasney, J., et al. (2008). Abnormal sympathetic nervous system development and physiological dysautonomia in egr3-deficient mice. Development, 135, 2949–2957. https://doi.org/10.1242/dev.023960.
Hartman, M., Steketee, M. C., Silva, S., Lanning, K., & Mccann, H. (2003). Working memory and schizophrenia: evidence for slowed encoding. Schizophrenia Research, 59, 99–113. https://doi.org/10.1016/s0920-9964(01)00366-8.
Huang, Z., Liu, T., Chattoraj, A., Ahmed, S., Wang, M. M., Deng, J., et al. (2008). Posttranslational regulation of tph1 is responsible for the nightly surge of 5-ht output in the rat pineal gland. Journal of Pineal Research, 45, 506–514. https://doi.org/10.1111/j.1600-079X.2008.00627.x.
Janoutová, J., Janácková, P., Serý, O., Zeman, T., Ambroz, P., Kovalová, M., et al. (2016). Epidemiology and risk factors of schizophrenia. Neuro Endocrinology Letters, 37, 1–8.
Kapur, S., Zipursky, R. B., & Remington, G. (1999). Clinical and theoretical implications of 5-ht2 and d2 receptor occupancy of clozapine, risperidone, and olanzapine in schizophrenia. The American Journal of Psychiatry, 156, 286–293. https://doi.org/10.1176/ajp.156.2.286.
Kelly, J. P., Wrynn, A. S., & Leonard, B. E. (1997). The olfactory bulbectomized rat as a model of depression: an update. Pharmacology & Therapeutics, 74, 299–316. https://doi.org/10.1016/s0163-7258(97)00004-1.
Kim, S. H., Song, J. Y., Joo, E. J., Lee, K. Y., & Kim, Y. S. (2010). Egr3 as a potential susceptibility gene for schizophrenia in korea. American Journal of Medical Genetics. Part B, Neuropsychiatric Genetics, 153B, 1355–1360. https://doi.org/10.1002/ajmg.b.31115.
Kiparizoska, S., & Ikuta, T. (2017). Disrupted olfactory integration in schizophrenia: functional connectivity study. The International Journal of Neuropsychopharmacology, 20, 740–746. https://doi.org/10.1093/ijnp/pyx045.
Krausz, M., Moritz, S., Lambert, M., & Naber, D. (2000). Dosage of conventional neuroleptic medication and subjective cognitive functioning in schizophrenia. International Clinical Psychopharmacology, 15, 77–81. https://doi.org/10.1097/00004850-200015020-00003.
Lin, E., Lee, N. K., Karl, T., Duffy, L., O’Brien, E., Matsumoto, I., et al. (2006). Distinct endocrine effects of chronic haloperidol or risperidone administration in male rats. Neuropharmacology, 51, 1129–1136. https://doi.org/10.1016/j.neuropharm.2006.07.006.
Liu, Y., Liang, M., Zhou, Y., He, Y., Hao, Y., Song, M., et al. (2008). Disrupted small-world networks in schizophrenia. Brain, 131, 945–961. https://doi.org/10.1093/brain/awn018.
Lynall, M. E., Bassett, D. S., Kerwin, R., Mckenna, P. J., Kitzbichler, M., Muller, U., et al. (2010). Functional connectivity and brain networks in schizophrenia. The Journal of Neuroscience, 30, 9477–9487. https://doi.org/10.1523/JNEUROSCI.0333-10.2010.
Ma, G. L., Song, T. B., Chen, M., Fu, Y., Xu, Y., Ma, E. S., et al. (2013). Hippocampal and thalamic neuronal metabolism in a putative rat model of schizophrenia. Neural Regeneration Research, 8, 2415–2423. https://doi.org/10.1016/s0920-9964(02)00288-8.
Meyer-Lindenberg, A. S., Olsen, R. K., Kohn, P. D., Brown, T., Egan, M. F., Weinberger, D. R., et al. (2005). Regionally specific disturbance of dorsolateral prefrontal-hippocampal functional connectivity in schizophrenia. Archives of General Psychiatry, 62, 379–386. https://doi.org/10.1001/archpsyc.62.4.379.
Micheloyannis, S. (2012). Graph-based network analysis in schizophrenia. World Journal Psychiatry, 2, 1–12. https://doi.org/10.5498/wjp.v2.i1.1.
Pakkenberg, B. (1990). Pronounced reduction of total neuron number in mediodorsal thalamic nucleus and nucleus accumbens in schizophrenics. Archives of General Psychiatry, 47, 1023. https://doi.org/10.1001/archpsyc.1990.01810230039007.
Paxinos, G., & Watson, C. (2005). The Rat Brain in Stereotaxic Coordinates (5th ed.). New York: Academic. https://doi.org/10.1046/j.1469-7580.1997.191203153.x.
Pegues, M. P., Rogers, L. J., Amend, D., Vinogradov, S., & Deicken, R. F. (2003). Anterior hippocampal volume reduction in male patients with schizophrenia. Schizophrenia Research, 60, 105–115.
Rajarethinam, R., Gupta, S., & Andreasen, N. C. (1995). Volume of the pineal gland in schizophrenia; an MRI study. Schizophrenia Research, 14, 253–255. https://doi.org/10.1016/0920-9964(94)00077-L.
Reinwald, J. R., Becker, R., Mallien, A. S., Falfan-Melgoza, C., Sack, M., Christian, C. V. H., et al. (2017). Neural mechanisms of early-life social stress as a developmental risk factor for severe psychiatric disorders. Biological Psychiatry, 84, 116–128. https://doi.org/10.1016/j.biopsych.2017.12.010.
Rubinov, M., & Bullmore, E. (2013). Schizophrenia and abnormal brain network hubs. Dialogues in Clinical Neuroscience, 15, 339–349.
Rubinov, M., & Sporns, O. (2010). Complex network measures of brain connectivity: Uses and interpretations. Neuroimage, 52, 1059–1069. https://doi.org/10.1016/j.neuroimage.2009.10.003.
Sandyk, R. (1992). The pineal gland and the mode of onset of schizophrenia. The International Journal of Neuroscience, 67, 9–17. https://doi.org/10.3109/00207459208994772.
Shamir, E., Rotenberg, V. S., Laudon, M., Zisapel, N., & Elizur, A. (2001). First-night effect of melatonin treatment in patients with chronic schizophrenia. Journal of Clinical Psychopharmacology, 20, 691–694. https://doi.org/10.1097/00004714-200012000-00017.
Singh, S., Kumar, M., Sharma, A., Modi, S., Kaur, P., Bhatia, T., et al. (2015). Disrupted functional connectivity in schizophrenia: a resting state fmri study. European Psychiatry, 30, 907. https://doi.org/10.1016/S0924-9338(15)30707-0.
Song, T., Nie, B., Ma, E., Che, J., Sun, S., Wang, Y., et al. (2015). Functional magnetic resonance imaging reveals abnormal brain connectivity in egr3 gene transfected rat model of schizophrenia. Biochemical and Biophysical Research Communications, 460, 678–683. https://doi.org/10.1016/j.bbrc.2015.03.089.
Stone, M., Gabrieli, J. D. E., Stebbins, G. T., & Sullivan, E. V. (1998). Working and strategic memory deficits in schizophrenia. Neuropsychology, 12, 278–288. https://doi.org/10.1037/0894-4105.12.2.278.
Turetsky, B. I. (2000). Reduced olfactory bulb volume in patients with schizophrenia. The American Journal of Psychiatry, 157, 828–830. https://doi.org/10.1176/appi.ajp.157.5.828.
Turetsky, B. I., Hahn, C. G., Borgmann-Winter, K., & Moberg, P. J. (2009). Scents and nonsense: olfactory dysfunction in schizophrenia. Schizophrenia Bulletin, 35, 1117–1131. https://doi.org/10.1093/schbul/sbp111.
Varela, F., Lachaux, J. P., Rodriguez, E., & Martinerie, J. (2001). The brainweb: phase synchronization and large-scale integration. Nature Reviews. Neurology, 2, 229–239. https://doi.org/10.1038/35067550.
Vargha-Khadem, F., Gadian, D. G., Watkins, K. E., Connelly, A., Van Paesschen, W., & Mishkin, M. (1997). Differential effects of early hippocampal pathology on episodic and semantic memory. Science, 277, 376–380. https://doi.org/10.1126/science.277.5324.376.
Wang, J., Wang, X., Xia, M., Liao, X., Evans, A., & He, Y. (2015). GRETNA: a graph theoretical network analysis toolbox for imaging connectomics. Frontiers in Human Neuroscience, 9, 386. https://doi.org/10.3389/fnhum.2015.00386.
Wang, J. H., Zuo, X. N., & He, Y. (2010). Graph-based network analysis of resting-state functional MRI. Frontiers in Systems Neuroscience, 4, 16. https://doi.org/10.3389/fnsys.2010.00016.
Wang, L., Metzak, P. D., Honer, W. G., & Woodward, T. S. (2010). Impaired efficiency of functional networks underlying episodic memory-forcontext in schizophrenia. The Journal of Neuroscience, 30, 13171–13179. https://doi.org/10.1523/JNEUROSCI.3514-10.2010.
Yamada, K., Gerber, D. J., Iwayama, Y., Ohnishi, T., Ohba, H., Toyota, T., et al. (2007). Genetic analysis of the calcineurin pathway identifies members of the egr gene family, specifically egr3, as potential susceptibility candidates in schizophrenia. Proceedings of the National Academy of Sciences of the United States of America, 104, 2815–2820. https://doi.org/10.1073/pnas.0610765104.
Zuo, X. N., Ehmke, R., Mennes, M., Imperati, D., Castellanos, F. X., Sporns, O., et al. (2012). Network Centrality in the Human Functional Connectome. Cerebral Cortex, 22, 1862–1875. https://doi.org/10.1093/cercor/bhr269.
Funding
This study was supported by the grants from National Natural Science Foundation of China (NSFC) (Nos. 81971585 and 81571641), and The National Key Research and Development Program of China (Nos. 2020YFC2003903 and 2016YFC1307001).
Author information
Authors and Affiliations
Contributions
XT, GM conceived the experiments; GM, HM, AC and FS designed the experiments; GL, XH, WG and SZ performed the experiments; GL, ZS analyzed the data; WG, YW, XL, YC discussed the data; WG wrote the manuscript; LZ revised the manuscript; All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The author has no conflicts of interest to declare.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, W., Li, G., Han, X. et al. Regional brain network and behavioral alterations in EGR3 gene transfected rat model of schizophrenia. Brain Imaging and Behavior 15, 2606–2615 (2021). https://doi.org/10.1007/s11682-021-00462-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-021-00462-3