Abstract
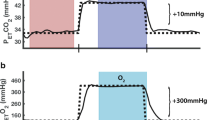
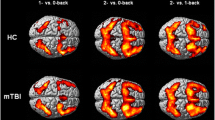
The purpose of this study was to quantify differences in blood oxygen level dependent (BOLD) activation on a working memory task, baseline cerebral blood flow (CBF0), and cerebrovascular reactivity (CVR) between participants with and without a history of concussion. A dual-echo pseudo-continuous arterial spin labelling (pCASL) sequence was performed on a group of 10 subjects with a previous concussion (126 ± 15 days prior) and on a control group (n = 10) during a visual working memory protocol. A separate dual-echo pCASL sequence was used to derive CVR and CBF0 measurements from a boxcar hypercapnic breathing protocol. Brain areas with significant activation differences on the working memory task between groups were identified and combined as an aggregate region of interest for CBF and CVR analyses. Areas of reduced BOLD activation during the working memory task in the concussed group included the ventral anterior cingulate cortex (ACC), the medial temporal gyrus (MTG), and the lateral occipital cortex in two loci. A single area of increased activation was located in the parietal operculum. Further analyses of CBF0 and CVR in these regions revealed reduced CVR in the concussed group in the MTG and ACC, while CBF0 did not differ. The differences in CVR between the two groups in these regions suggest that concussive injury may result in microvascular dysfunction. In turn, the decreased BOLD response during the task could be due to altered neurovascular coupling, rather than an impairment in neural activation alone. However, in other regions associated with working memory, unchanged CBF0 and CVR suggests that neural injury also persists after concussion. In the future, BOLD results should be normalized to CVR in order achieve a clearer understanding of the neural and vascular contributions to the differences in the signal.






Similar content being viewed by others
References
Amyot, F., Kenney, K., Moore, C., Haber, M., Turtzo, L. C., Shenouda, C., et al.(2018) Imaging of Cerebrovascular Function in Chronic Traumatic Brain Injury. Journal of Neurotrauma 35(10):1116-1123
Andersson, J. L. R., Skare, S., & Ashburner, J. (2003). How to correct susceptibility distortions in spin-echo echo-planar images: Application to diffusion tensor imaging. NeuroImage, 20(2), 870–888. https://doi.org/10.1016/S1053-8119(03)00336-7.
Aslan, S., Xu, F., Wang, P. L., Uh, J., Yezhuvath, U. S., van Osch, M., & Lu, H. (2010). Estimation of labeling efficiency in pseudocontinuous arterial spin labeling. Magnetic Resonance in Medicine, 63(3), 765–771. https://doi.org/10.1002/mrm.22245.
Attwell, D., & Iadecola, C. (2002). The neural basis of functional brain imaging signals. Trends in Neurosciences, 25(12), 621–625.
Attwell, D., Buchan, A. M., Charpak, S., Lauritzen, M., MacVicar, B. A., & Newman, E. A. (2010). Glial and neuronal control of brain blood flow. Nature, 468(7321), 232–243. https://doi.org/10.1038/nature09613.
Baddeley, A., & Hitch, G. (1974). Working memory. In The psychology of learning and motivation: Advances in research and theory (pp. 47–89). New York: Academic Press.
Bandettini, P. A., & Wong, E. C. (1997). A hypercapnia-based normalization method for improved spatial localization of human brain activation with fMRI. NMR in Biomedicine, 10(4–5), 197–203. https://doi.org/10.1002/(SICI)1099-1492(199706/08)10:4/5<197::AID-NBM466>3.0.CO;2-S.
Barlow, K. M., Marcil, L. D., Dewey, D., Carlson, H. L., MacMaster, F. P., Brooks, B. L., & Lebel, R. M. (2017). Cerebral perfusion changes in post-concussion syndrome: A prospective controlled cohort study. Journal of Neurotrauma, 34(5), 996–1004. https://doi.org/10.1089/neu.2016.4634.
Blockley, N. P., Driver, I. D., Francis, S. T., Fisher, J. A., & Gowland, P. A. (2011). An improved method for acquiring cerebrovascular reactivity maps. Magnetic Resonance in Medicine, 65(5), 1278–1286. https://doi.org/10.1002/mrm.22719.
Bright, M. G., & Murphy, K. (2013). Reliable quantification of BOLD fMRI cerebrovascular reactivity despite poor breath-hold performance. NeuroImage, 83, 559–568. https://doi.org/10.1016/j.neuroimage.2013.07.007.
Buxton, R. B., Uludağ, K., Dubowitz, D. J., & Liu, T. T. (2004). Modeling the hemodynamic response to brain activation. NeuroImage, 23(Suppl 1), S220–S233. https://doi.org/10.1016/j.neuroimage.2004.07.013.
Champagne, A. A., Bhogal, A. A., Coverdale, N. S., Mark, C. I., & Cook, D. J. (2019). A novel perspective to calibrate temporal delays in cerebrovascular reactivity using hypercapnic and hyperoxic respiratory challenges. NeuroImage, 187, 154–165. https://doi.org/10.1016/j.neuroimage.2017.11.044.
Chen, J. J., & Pike, G. (2010). Global cerebral oxidative metabolism during hypercapnia and hypocapnia in humans: Implications for BOLD fMRI. Journal of Cerebral Blood Flow and Metabolism: Official Journal of the International Society of Cerebral Blood Flow and Metabolism, 30(6), 1094–1099. https://doi.org/10.1038/jcbfm.2010.42.
Chen, J.-K., Johnston, K. M., Frey, S., Petrides, M., Worsley, K., & Ptito, A. (2004). Functional abnormalities in symptomatic concussed athletes: An fMRI study. NeuroImage, 22(1), 68–82. https://doi.org/10.1016/j.neuroimage.2003.12.032.
Chu, P. P. W., Golestani, A. M., Kwinta, J. B., Khatamian, Y. B., & Chen, J. J. (2018). Characterizing the modulation of resting-state fMRI metrics by baseline physiology. NeuroImage, 173, 72–87. https://doi.org/10.1016/j.neuroimage.2018.02.004.
Chukwudelunzu, F. E., Brown, R. D., Wijdicks, E. F. M., & Steckelberg, J. M. (2002). Subarachnoid haemorrhage associated with infectious endocarditis: Case report and literature review. European Journal of Neurology, 9(4), 423–427.
Cox, R. W. (1996). AFNI: Software for analysis and visualization of functional magnetic resonance neuroimages. Computers and Biomedical Research, an International Journal, 29(3), 162–173.
da Costa, L., van Niftrik, C. B., Crane, D., Fierstra, J., & Bethune, A. (2016). Temporal profile of cerebrovascular reactivity impairment, gray matter volumes, and persistent symptoms after mild traumatic head injury. Frontiers in Neurology, 7, 70. https://doi.org/10.3389/fneur.2016.00070.
Duffin, J., Sobczyk, O., Crawley, A. P., Poublanc, J., Mikulis, D. J., & Fisher, J. A. (2015). The dynamics of cerebrovascular reactivity shown with transfer function analysis. NeuroImage, 114, 207–216. https://doi.org/10.1016/j.neuroimage.2015.04.029.
Fierstra, J., Sobczyk, O., Battisti-Charbonney, A., Mandell, D. M., Poublanc, J., Crawley, A. P., Mikulis, D. J., Duffin, J., & Fisher, J. A. (2013). Measuring cerebrovascular reactivity: What stimulus to use? The Journal of Physiology, 591(23), 5809–5821. https://doi.org/10.1113/jphysiol.2013.259150.
Germuska, M., Merola, A., Murphy, K., Babic, A., Richmond, L., Khot, S., Hall, J. E., & Wise, R. G. (2016). A forward modelling approach for the estimation of oxygen extraction fraction by calibrated fMRI. NeuroImage, 139, 313–323. https://doi.org/10.1016/j.neuroimage.2016.06.004.
Halani, S., Kwinta, J. B., Golestani, A. M., Khatamian, Y. B., & Chen, J. J. (2015). Comparing cerebrovascular reactivity measured using BOLD and cerebral blood flow MRI: The effect of basal vascular tension on vasodilatory and vasoconstrictive reactivity. NeuroImage, 110, 110–123. https://doi.org/10.1016/j.neuroimage.2015.01.050.
Halstead, M. E., Walter, K. D., & Council on Sports Medicine and Fitness. (2010). American Academy of Pediatrics. Clinical report--sport-related concussion in children and adolescents. Pediatrics, 126(3), 597–615. https://doi.org/10.1542/peds.2010-2005.
Handwerker, D. A., Gazzaley, A., Inglis, B. A., & D’Esposito, M. (2007). Reducing vascular variability of fMRI data across aging populations using a breathholding task. Human Brain Mapping, 28(9), 846–859. https://doi.org/10.1002/hbm.20307.
Herscovitch, P., & Raichle, M. E. (1985). What is the correct value for the brain--blood partition coefficient for water? Journal of Cerebral Blood Flow and Metabolism: Official Journal of the International Society of Cerebral Blood Flow and Metabolism, 5(1), 65–69. https://doi.org/10.1038/jcbfm.1985.9.
Jenkinson, M., Bannister, P., Brady, M., & Smith, S. (2002). Improved optimization for the robust and accurate linear registration and motion correction of brain images. NeuroImage, 17(2), 825–841.
Jenkinson, M., Beckmann, C. F., Behrens, T. E. J., Woolrich, M. W., & Smith, S. M. (2012). FSL. NeuroImage, 62(2), 782–790. https://doi.org/10.1016/j.neuroimage.2011.09.015.
Jonides, J., Smith, E. E., Koeppe, R. A., Awh, E., Minoshima, S., & Mintun, M. A. (1993). Spatial working memory in humans as revealed by PET. Nature, 363(6430), 623–625. https://doi.org/10.1038/363623a0.
Keightley, M. L., Singh Saluja, R., Chen, J.-K., Gagnon, I., Leonard, G., Petrides, M., & Ptito, A. (2014). A functional magnetic resonance imaging study of working memory in youth after sports-related concussion: Is it still working? Journal of Neurotrauma, 31(5), 437–451. https://doi.org/10.1089/neu.2013.3052.
Kontos, H. A., Raper, A. J., & Patterson, J. L. (1977). Analysis of vasoactivity of local pH, PCO2 and bicarbonate on pial vessels. Stroke, 8(3), 358–360. https://doi.org/10.1161/01.str.8.3.358.
Liu, P., Li, Y., Pinho, M., Park, D. C., Welch, B. G., & Lu, H. (2017). Cerebrovascular reactivity mapping without gas challenges. NeuroImage, 146, 320–326. https://doi.org/10.1016/j.neuroimage.2016.11.054.
Liu, P., De Vis, J. B., & Lu, H. (2019). Cerebrovascular reactivity (CVR) MRI with CO2 challenge: A technical review. NeuroImage, 187, 104–115. https://doi.org/10.1016/j.neuroimage.2018.03.047.
Lyons, D. N., Vekaria, H., Macheda, T., Bakshi, V., Powell, D. K., Gold, B. T., Lin, A. L., Sullivan, P. G., & Bachstetter, A. D. (2018). A mild traumatic nrain injury in mice produces lasting deficits in brain metabolism. Journal of Neurotrauma, 35(20), 2435–2447. https://doi.org/10.1089/neu.2018.5663.
Manoach, D. S., Greve, D. N., Lindgren, K. A., & Dale, A. M. (2003). Identifying regional activity associated with temporally separated components of working memory using event-related functional MRI. NeuroImage, 20(3), 1670–1684.
McCrea, M., Guskiewicz, K. M., Marshall, S. W., Barr, W., Randolph, C., Cantu, R. C., Onate, J. A., Yang, J., & Kelly, J. P. (2003). Acute effects and recovery time following concussion in collegiate football players: The NCAA concussion study. JAMA, 290(19), 2556–2563. https://doi.org/10.1001/jama.290.19.2556.
McCrea, M., Barr, W. B., Guskiewicz, K., Randolph, C., Marshall, S. W., Cantu, R., et al. (2005). Standard regression-based methods for measuring recovery after sport-related concussion. Journal of the International Neuropsychological Society: JINS, 11(1), 58–69. https://doi.org/10.1017/S1355617705050083.
McCrea, M., Guskiewicz, K., Randolph, C., Barr, W. B., Hammeke, T. A., Marshall, S. W., Powell, M. R., Woo Ahn, K., Wang, Y., & Kelly, J. P. (2013). Incidence, clinical course, and predictors of prolonged recovery time following sport-related concussion in high school and college athletes. Journal of the International Neuropsychological Society: JINS, 19(1), 22–33. https://doi.org/10.1017/S1355617712000872.
McCrory, P., Meeuwisse, W., Aubry, M., Cantu, B., Dvořák, J., Echemendia, R., Engebretsen, L., Johnston, K., Kutcher, J., Raftery, M., Sills, A., Benson, B., Davis, G., Ellenbogen, R., Guskiewicz, K., Herring, S. A., Iverson, G., Jordan, B., Kissick, J., McCrea, M., McIntosh, A., Maddocks, D., Makdissi, M., Purcell, L., Putukian, M., Schneider, K., Tator, C., & Turner, M. (2013). Consensus statement on concussion in sport - the 4th international conference on concussion in sport held in Zurich, November 2012. Physical Therapy in Sport: Official Journal of the Association of Chartered Physiotherapists in Sports Medicine, 14(2), e1–e13. https://doi.org/10.1016/j.ptsp.2013.03.002.
Meier, T. B., Bellgowan, P. S. F., Singh, R., Kuplicki, R., Polanski, D. W., & Mayer, A. R. (2015). Recovery of cerebral blood flow following sports-related concussion. JAMA Neurology, 72(5), 530–538. https://doi.org/10.1001/jamaneurol.2014.4778.
Militana, A. R., Donahue, M. J., Sills, A. K., Solomon, G. S., Gregory, A. J., Strother, M. K., & Morgan, V. L. (2016). Alterations in default-mode network connectivity may be influenced by cerebrovascular changes within 1 week of sports related concussion in college varsity athletes: A pilot study. Brain Imaging and Behavior, 10(2), 559–568. https://doi.org/10.1007/s11682-015-9407-3.
Mutch, W. A. C., Ellis, M. J., Graham, M. R., Wourms, V., Raban, R., Fisher, J. A., Mikulis, D., Leiter, J., & Ryner, L. (2014). Brain MRI CO2 stress testing: A pilot study in patients with concussion. PLoS One, 9(7), e102181. https://doi.org/10.1371/journal.pone.0102181.
Mutch, W. A. C., Ellis, M. J., Ryner, L. N., Graham, M. R., Dufault, B., Gregson, B., et al. (2016). Brain magnetic resonance imaging CO2 stress testing in adolescent postconcussion syndrome. Journal of Neurosurgery, 125(3), 648–660. https://doi.org/10.3171/2015.6.JNS15972.
Mutch, W. A. C., Ellis, M. J., Ryner, L. N., McDonald, P. J., Morissette, M. P., Pries, P., Essig, M., Mikulis, D. J., Duffin, J., & Fisher, J. A. (2018). Patient-specific alterations in CO2 cerebrovascular responsiveness in acute and sub-acute sports-related concussion. Frontiers in Neurology, 9, 23. https://doi.org/10.3389/fneur.2018.00023.
Nee, D. E., & Jonides, J. (2013). Neural evidence for a 3-state model of visual short-term memory. NeuroImage, 74, 1–11. https://doi.org/10.1016/j.neuroimage.2013.02.019.
Ogawa, S., Lee, T. M., Kay, A. R., & Tank, D. W. (1990). Brain magnetic resonance imaging with contrast dependent on blood oxygenation. Proceedings of the National Academy of Sciences of the United States of America, 87(24), 9868–9872.
Ogawa, S., Menon, R. S., Tank, D. W., Kim, S. G., Merkle, H., Ellermann, J. M., & Ugurbil, K. (1993). Functional brain mapping by blood oxygenation level-dependent contrast magnetic resonance imaging. A comparison of signal characteristics with a biophysical model. Biophysical Journal, 64(3), 803–812. https://doi.org/10.1016/S0006-3495(93)81441-3.
Ozen, L. J., Itier, R. J., Preston, F. F., & Fernandes, M. A. (2013). Long-term working memory deficits after concussion: Electrophysiological evidence. Brain Injury, 27(11), 1244–1255. https://doi.org/10.3109/02699052.2013.804207.
Para, A. E., Sam, K., Poublanc, J., Fisher, J. A., Crawley, A. P., & Mikulis, D. J. (2017). Invalidation of fMRI experiments secondary to neurovascular uncoupling in patients with cerebrovascular disease. Journal of magnetic resonance imaging: JMRI, 46(5), 1448–1455. https://doi.org/10.1002/jmri.25639.
Pardini, J. E., Pardini, D. A., Becker, J. T., Dunfee, K. L., Eddy, W. F., Lovell, M. R., & Welling, J. S. (2010). Postconcussive symptoms are associated with compensatory cortical recruitment during a working memory task. Neurosurgery, 67(4), 1020–1027; discussion 1027-1028. https://doi.org/10.1227/NEU.0b013e3181ee33e2.
Portegies, M. L. P., de Bruijn, R. F. A. G., Hofman, A., Koudstaal, P. J., & Ikram, M. A. (2014). Cerebral vasomotor reactivity and risk of mortality: The Rotterdam study. Stroke, 45(1), 42–47. https://doi.org/10.1161/STROKEAHA.113.002348.
Reinhard, M., Schwarzer, G., Briel, M., Altamura, C., Palazzo, P., King, A., Bornstein, N. M., Petersen, N., Motschall, E., Hetzel, A., Marshall, R. S., Klijn, C. J. M., Silvestrini, M., Markus, H. S., & Vernieri, F. (2014). Cerebrovascular reactivity predicts stroke in high-grade carotid artery disease. Neurology, 83(16), 1424–1431. https://doi.org/10.1212/WNL.0000000000000888.
Shah-Basak, P. P., Urbain, C., Wong, S., da Costa, L., Pang, E. W., Dunkley, B. T., & Taylor, M. J. (2018). Concussion alters the functional brain processes of visual attention and working memory. Journal of Neurotrauma, 35(2), 267–277. https://doi.org/10.1089/neu.2017.5117.
Silvestrini, M., Vernieri, F., Pasqualetti, P., Matteis, M., Passarelli, F., Troisi, E., & Caltagirone, C. (2000). Impaired cerebral vasoreactivity and risk of stroke in patients with asymptomatic carotid artery stenosis. JAMA, 283(16), 2122–2127.
Smith, E. E., & Jonides, J. (1998). Neuroimaging analyses of human working memory. Proceedings of the National Academy of Sciences of the United States of America, 95(20), 12061–12068.
Smith, S. M., Jenkinson, M., Woolrich, M. W., Beckmann, C. F., Behrens, T. E. J., Johansen-Berg, H., Bannister, P. R., de Luca, M., Drobnjak, I., Flitney, D. E., Niazy, R. K., Saunders, J., Vickers, J., Zhang, Y., de Stefano, N., Brady, J. M., & Matthews, P. M. (2004). Advances in functional and structural MR image analysis and implementation as FSL. NeuroImage, 23(Suppl 1), S208–S219. https://doi.org/10.1016/j.neuroimage.2004.07.051.
Smits, M., Dippel, D. W. J., Houston, G. C., Wielopolski, P. A., Koudstaal, P. J., Hunink, M. G. M., & van der Lugt, A. (2009). Postconcussion syndrome after minor head injury: Brain activation of working memory and attention. Human Brain Mapping, 30(9), 2789–2803. https://doi.org/10.1002/hbm.20709.
Stobart, J. L. L., Lu, L., Anderson, H. D. I., Mori, H., & Anderson, C. M. (2013). Astrocyte-induced cortical vasodilation is mediated by D-serine and endothelial nitric oxide synthase. Proceedings of the National Academy of Sciences of the United States of America, 110(8), 3149–3154. https://doi.org/10.1073/pnas.1215929110.
Suri, S., Mackay, C. E., Kelly, M. E., Germuska, M., Tunbridge, E. M., Frisoni, G. B., et al. (2015). Reduced cerebrovascular reactivity in young adults carrying the APOE ε4 allele. Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, 11(6), 648-657.e1. https://doi.org/10.1016/j.jalz.2014.05.1755.
Svaldi, D. O., Joshi, C., McCuen, E. C., Music, J. P., Hannemann, R., Leverenz, L. J., Nauman, E. A., & Talavage, T. M. (2018). Accumulation of high magnitude acceleration events predicts cerebrovascular reactivity changes in female high school soccer athletes. Brain Imaging and Behavior. https://doi.org/10.1007/s11682-018-9983-0.
Thériault, M., De Beaumont, L., Tremblay, S., Lassonde, M., & Jolicoeur, P. (2011). Cumulative effects of concussions in athletes revealed by electrophysiological abnormalities on visual working memory. Journal of Clinical and Experimental Neuropsychology, 33(1), 30–41. https://doi.org/10.1080/13803391003772873.
Thomason, M. E., Foland, L. C., & Glover, G. H. (2007). Calibration of BOLD fMRI using breath holding reduces group variance during a cognitive task. Human Brain Mapping, 28(1), 59–68. https://doi.org/10.1002/hbm.20241.
Tukey, John W. (1977). Exploratory data analysis. Addison-Wesley.
van Niftrik, C. H. B., Piccirelli, M., Bozinov, O., Pangalu, A., Valavanis, A., Regli, L., & Fierstra, J. (2016). Fine tuning breath-hold-based cerebrovascular reactivity analysis models. Brain and Behavior: A Cognitive Neuroscience Perspective, 6(2), e00426. https://doi.org/10.1002/brb3.426.
van Niftrik, C. H. B., Piccirelli, M., Bozinov, O., Pangalu, A., Fisher, J. A., Valavanis, A., Luft, A. R., Weller, M., Regli, L., & Fierstra, J. (2017). Iterative analysis of cerebrovascular reactivity dynamic response by temporal decomposition. Brain and Behavior: A Cognitive Neuroscience Perspective, 7(9), e00705. https://doi.org/10.1002/brb3.705.
van Niftrik, C. H. B., Piccirelli, M., Muscas, G., Sebök, M., Fisher, J. A., Bozinov, O., Stippich, C., Valavanis, A., Regli, L., & Fierstra, J. (2019). The voxel-wise analysis of false negative fMRI activation in regions of provoked impaired cerebrovascular reactivity. PLoS One, 14(5), e0215294. https://doi.org/10.1371/journal.pone.0215294.
Wang, J., Alsop, D. C., Song, H. K., Maldjian, J. A., Tang, K., Salvucci, A. E., & Detre, J. A. (2003). Arterial transit time imaging with flow encoding arterial spin tagging (FEAST). Magnetic Resonance in Medicine, 50(3), 599–607. https://doi.org/10.1002/mrm.10559.
Westfall, D. R., West, J. D., Bailey, J. N., Arnold, T. W., Kersey, P. A., Saykin, A. J., & McDonald, B. C. (2015). Increased brain activation during working memory processing after pediatric mild traumatic brain injury (mTBI). Journal of Pediatric Rehabilitation Medicine, 8(4), 297–308. https://doi.org/10.3233/PRM-150348.
Wright, A. D., Smirl, J. D., Bryk, K., & van Donkelaar, P. (2017). A prospective transcranial Doppler ultrasound-based evaluation of the acute and cumulative effects of sport-related concussion on neurovascular coupling response dynamics. Journal of Neurotrauma, 34(22), 3097–3106. https://doi.org/10.1089/neu.2017.5020.
Wu, W.-C., Fernández-Seara, M., Detre, J. A., Wehrli, F. W., & Wang, J. (2007). A theoretical and experimental investigation of the tagging efficiency of pseudocontinuous arterial spin labeling. Magnetic Resonance in Medicine, 58(5), 1020–1027. https://doi.org/10.1002/mrm.21403.
Zhang, Y., Brady, M., & Smith, S. (2001). Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Transactions on Medical Imaging, 20(1), 45–57. https://doi.org/10.1109/42.906424.
Zhang, X., Petersen, E. T., Ghariq, E., De Vis, J. B., Webb, A. G., Teeuwisse, W. M., et al. (2013). In vivo blood T(1) measurements at 1.5 T, 3 T, and 7 T. Magnetic Resonance in Medicine, 70(4), 1082–1086. https://doi.org/10.1002/mrm.24550.
Acknowledgements
The authors would like to thank Cheryl Hamilton and Don Brien for their dedication and willingness to help with data collection. The authors would also like to acknowledge Dr. J. J. Wang at UCLA for sharing the pCASL sequence used in this study. The authors would also like to acknowledge Dr. Michael Germuska (Cardiff University) and Dr. Alex Bhogal (University Medical Center Utrecht) for their input regarding the pre-processing workflow used in this study to derive physiological measurements.
Funding
This work was supported by the Canadian Institutes of Health Research (CIHR) and the Natural Sciences and Engineering Research Council (NSERC) through a Collaborative Health Research Project Grant (#315705).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have conflicts of interest to disclose.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Figure S1
Scatter plots of cerebral blood flow and cerebrovascular reactivity for the regions where activation was different during the working memory task. Open circles represent the concussed group and filled show the control group. A) Lateral occipital cortex superior (LOCs) and B) inferior (LOCi), C) medial temporal gyrus (MTG), D) parietal operculum (POp), E) region of interest (ROI), F) superior frontal gyrus (SFG), G) ventral anterior cingulate cortex (ACC). (PNG 102 kb)
Rights and permissions
About this article
Cite this article
Coverdale, N.S., Fernandez-Ruiz, J., Champagne, A.A. et al. Co-localized impaired regional cerebrovascular reactivity in chronic concussion is associated with BOLD activation differences during a working memory task. Brain Imaging and Behavior 14, 2438–2449 (2020). https://doi.org/10.1007/s11682-019-00194-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-019-00194-5