Abstract
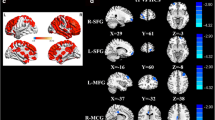
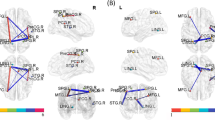
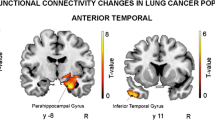
Previous studies have demonstrated that cognitive impairment is associated with neurophysiological changes in lung cancer following chemotherapy. This study aimed to investigate the intrinsic functional connectivity (FC) pattern within the default mode network (DMN) and its associations with cognitive impairment in patients with lung cancer revealed by resting-state functional magnetic resonance imaging (fMRI). Resting-state fMRI scans were acquired from 21 post-chemotherapy and 27 non-chemotherapy lung cancer patients and 30 healthy controls. All groups were age, gender and education-matched. The posterior cingulate cortex (PCC) was chosen as the seed region to detect the FC patterns and then determine whether these changes were related with specific cognitive performance. Compared with non-chemotherapy lung cancer patients, chemotherapy patients revealed decreased FC between the PCC and the right anterior cingulate cortex (ACC), left inferior parietal lobule (IPL), and left medial prefrontal cortex (mPFC), as well as increased FC with the left postcentral gyrus (PoCG). Relative to healthy controls, post-chemotherapy patients exhibited reduced FC between the PCC and the left ACC and left temporal lobe, as well as increased FC with the right PoCG. Moreover, the decreased FC of the PCC to bilateral ACC in post-chemotherapy patients was positively associated with reduced MoCA scores (left: r = 0.529, p = 0.029; right: r = 0.577, p = 0.015). The current study mainly demonstrated reduced resting-state FC pattern within the DMN regions that was linked with impaired cognitive function in lung cancer patients after chemotherapy. These findings illustrated the potential role of the DMN in lung cancer patients that will provide novel insight into the underlying neuropathological mechanisms in chemotherapy-induced cognitive impairment.



Similar content being viewed by others
References
Agrawal, V., Coroller, T. P., Hou, Y., Lee, S. W., Romano, J. L., Baldini, E. H., Chen, A. B., Jackman, D. M., Kozono, D., Swanson, S. J., Wee, J. O., Aerts, H. J. W. L., & Mak, R. H. (2016). Radiologic-pathologic correlation of response to chemoradiation in resectable locally advanced NSCLC. Lung Cancer, 102, 1–8.
Ahles, T. A., & Saykin, A. J. (2002). Breast cancer chemotherapy-related cognitive dysfunction. Clinical Breast Cancer, 3(3), S84–S90.
Arriagada, R., Le Chevalier, T., Borie, F., Riviere, A., Chomy, P., Monnet, I., et al. (1995). Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. Journal of the National Cancer Institute, 87(3), 183–190.
Ashburner, J., & Friston, K. J. (2005). Unified segmentation. Neuroimage, 26(3), 839–851. https://doi.org/10.1016/j.neuroimage.2005.02.018.
Bromis, K., Gkiatis, K., Karanasiou, I., Matsopoulos, G., Karavasilis, E., Papathanasiou, M., et al. (2017). Altered brain functional connectivity in small-cell lung Cancer patients after chemotherapy treatment: A resting-state fMRI study. Computational and Mathematical Methods in Medicine, 1403940(10), 17.
Bush, G., Luu, P., & Posner, M. I. (2000). Cognitive and emotional influences in anterior cingulate cortex. Trends in Cognitive Sciences, 4(6), 215–222.
Cees De Groot, J., De Leeuw, F.E., Oudkerk, M., Van Gijn, J., Hofman, A., Jolles, J., et al. (2000). Cerebral white matter lesions and cognitive function: the Rotterdam Scan Study. Annals of Neurology: Official Journal of the American Neurological Association and the Child Neurology Society, 47(2), 145–151.
Correa, D., Root, J., Kryza-Lacombe, M., Mehta, M., Karimi, S., Hensley, M., et al. (2017). Brain structure and function in patients with ovarian cancer treated with first-line chemotherapy: A pilot study. Brain Imaging and Behavior, 11(6), 1652–1663.
Damoiseaux, J. S., Rombouts, S. A., Barkhof, F., Scheltens, P., Stam, C. J., Smith, S. M., et al. (2006). Consistent resting-state networks across healthy subjects. Proceedings of the National Academy of Sciences of the United States of America, 103(37), 13848–13853.
de Groot, J.C., De Leeuw, F.-E., Oudkerk, M., Hofman, A., Jolles, J., and Breteler, M. (2001). Cerebral white matter lesions and subjective cognitive dysfunction The Rotterdam Scan Study. Neurology, 56(11), 1539–1545.
Deprez, S., Amant, F., Yigit, R., Porke, K., Verhoeven, J., Van den Stock, J., et al. (2011). Chemotherapy-induced structural changes in cerebral white matter and its correlation with impaired cognitive functioning in breast cancer patients. Human Brain Mapping, 32(3), 480–493.
Ettinger, D. S., Wood, D. E., Aisner, D. L., Akerley, W., Bauman, J., Chirieac, L. R., D'Amico, T. A., DeCamp, M. M., Dilling, T. J., Dobelbower, M., Doebele, R. C., Govindan, R., Gubens, M. A., Hennon, M., Horn, L., Komaki, R., Lackner, R. P., Lanuti, M., Leal, T. A., Leisch, L. J., Lilenbaum, R., Lin, J., Loo, B. W., Jr., Martins, R., Otterson, G. A., Reckamp, K., Riely, G. J., Schild, S. E., Shapiro, T. A., Stevenson, J., Swanson, S. J., Tauer, K., Yang, S. C., Gregory, K., & Hughes, M. (2017). Non-small cell lung Cancer, version 5.2017, NCCN clinical practice guidelines in oncology. Journal of the National Comprehensive Cancer Network, 15(4), 504–535.
Fox, M. D., & Raichle, M. E. (2007). Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nature Reviews. Neuroscience, 8(9), 700–711. https://doi.org/10.1038/nrn2201.
Fransson, P., & Marrelec, G. (2008). The precuneus/posterior cingulate cortex plays a pivotal role in the default mode network: Evidence from a partial correlation network analysis. Neuroimage, 42(3), 1178–1184. https://doi.org/10.1016/j.neuroimage.2008.05.059.
Galea, M., & Woodward, M. (2005). Mini-mental state examination (MMSE). The Australian Journal of Physiotherapy, 51(3), 198.
Grosshans, D. R., Meyers, C. A., Allen, P. K., Davenport, S. D., & Komaki, R. (2008). Neurocognitive function in patients with small cell lung cancer : Effect of prophylactic cranial irradiation. Cancer, 112(3), 589–595.
Hong, Q. Y., Wu, G. M., Qian, G. S., Hu, C. P., Zhou, J. Y., Chen, L. A., Li, W. M., Li, S. Y., Wang, K., Wang, Q., Zhang, X. J., Li, J., Gong, X., Bai, C. X., & on behalf of the Lung Cancer Group of the Chinese Thoracic Society; Chinese Alliance Against Lung Cancer. (2015). Prevention and management of lung cancer in China. Cancer, 121(Suppl 17), 3080–3088. https://doi.org/10.1002/cncr.29584.
Inagaki, M., Yoshikawa, E., Matsuoka, Y., Sugawara, Y., Nakano, T., Akechi, T., Wada, N., Imoto, S., Murakami, K., Uchitomi, Y., & and The Breast Cancer Survivors' Brain MRI Database Group. (2007). Smaller regional volumes of brain gray and white matter demonstrated in breast cancer survivors exposed to adjuvant chemotherapy. Cancer, 109(1), 146–156. https://doi.org/10.1002/cncr.22368.
Johnson, B. E., Grayson, J., Makuch, R. W., Linnoila, R. I., Anderson, M. J., Cohen, M. H., Glatstein, E., Minna, J. D., & Ihde, D. C. (1990). Ten-year survival of patients with small-cell lung cancer treated with combination chemotherapy with or without irradiation. Journal of Clinical Oncology, 8(3), 396–401.
Kaasa, S., Olsnes, B. T., Thorud, E., & Host, H. (1988). Reduced short-term neuropsychological performance in patients with nonsmall-cell lung cancer treated with cisplatin and etoposide. Antibiotics and Chemotherapy (1971), 41, 226–231.
Kam, J. W., Boyd, L. A., Hsu, C. L., Liu-Ambrose, T., Handy, T. C., Lim, H. J., et al. (2016). Altered neural activation during prepotent response inhibition in breast cancer survivors treated with chemotherapy: An fMRI study. Brain Imaging and Behavior, 10(3), 840–848.
Kesler, S. R. (2014). Default mode network as a potential biomarker of chemotherapy-related brain injury. Neurobiology of Aging, 35(2), 15.
Kesler, S. R., Wefel, J. S., Hosseini, S. H., Cheung, M., Watson, C. L., & Hoeft, F. (2013). Default mode network connectivity distinguishes chemotherapy-treated breast cancer survivors from controls. Proceedings of the National Academy of Sciences, 110(28), 11600–11605.
Kesler, S. R., Adams, M., Packer, M., Rao, V., Henneghan, A. M., Blayney, D. W., & Palesh, O. (2017). Disrupted brain network functional dynamics and hyper-correlation of structural and functional connectome topology in patients with breast cancer prior to treatment. Brain and Behavior, 7(3), e00643.
Kim, S. S., Gomez-Ramirez, M., Thakur, P. H., & Hsiao, S. S. (2015). Multimodal interactions between proprioceptive and cutaneous signals in primary somatosensory cortex. Neuron, 86(2), 555–566.
Lee, M. H., Smyser, C. D., & Shimony, J. S. (2013). Resting-state fMRI: A review of methods and clinical applications. American Journal of Neuroradiology, 34(10), 1866–1872.
Lowe, M. J., Mock, B. J., & Sorenson, J. A. (1998). Functional connectivity in single and multislice echoplanar imaging using resting-state fluctuations. Neuroimage, 7(2), 119–132. https://doi.org/10.1006/nimg.1997.0315.
Maldjian, J. A., Laurienti, P. J., Kraft, R. A., & Burdette, J. H. (2003). An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. Neuroimage, 19(3), 1233–1239.
Meyers, C. A., Byrne, K. S., & Komaki, R. (1995). Cognitive deficits in patients with small cell lung cancer before and after chemotherapy. Lung Cancer, 12(3), 231–235.
Miao, H., Chen, X., Yan, Y., He, X., Hu, S., Kong, J., Wu, M., Wei, Y., Zhou, Y., Wang, L., Wang, K., & Qiu, B. (2016). Functional connectivity change of brain default mode network in breast cancer patients after chemotherapy. Neuroradiology, 58(9), 921–928.
Molina, J. R., Yang, P., Cassivi, S. D., Schild, S. E., & Adjei, A. A. (2008). Non-small cell lung cancer: Epidemiology, risk factors, treatment, and survivorship. Mayo Clinic Proceedings, 83(5), 584–594.
Nanavaty, P., Alvarez, M. S., & Alberts, W. M. (2014). Lung cancer screening: Advantages, controversies, and applications. Cancer Control, 21(1), 9–14. https://doi.org/10.1177/107327481402100102.
Nasreddine, Z. S., Phillips, N. A., Bédirian, V., Charbonneau, S., Whitehead, V., Collin, I., et al. (2005). The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. Journal of the American Geriatrics Society, 53(4), 695–699.
Pomykala, K. L., de Ruiter, M. B., Deprez, S., McDonald, B. C., & Silverman, D. H. (2013). Integrating imaging findings in evaluating the post-chemotherapy brain. Brain Imaging and Behavior, 7(4), 436–452. https://doi.org/10.1007/s11682-013-9239-y.
Power, J. D., Barnes, K. A., Snyder, A. Z., Schlaggar, B. L., & Petersen, S. E. (2012). Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage, 59(3), 2142–2154.
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G. L. (2001). A default mode of brain function. Proceedings of the National Academy of Sciences, 98(2), 676–682.
Rosazza, C., & Minati, L. (2011). Resting-state brain networks: Literature review and clinical applications. Neurological Sciences, 32(5), 773–785.
Simo, M., Root, J. C., Vaquero, L., Ripolles, P., Jove, J., Ahles, T., et al. (2015). Cognitive and brain structural changes in a lung cancer population. Journal of Thoracic Oncology, 10(1), 38–45.
Simo, M., Vaquero, L., Ripolles, P., Gurtubay-Antolin, A., Jove, J., Navarro, A., et al. (2016). Longitudinal brain changes associated with prophylactic cranial irradiation in lung Cancer. Journal of Thoracic Oncology, 11(4), 475–486.
Simo, M., Rifa-Ros, X., Vaquero, L., Ripolles, P., Cayuela, N., Jove, J., et al. (2018). Brain functional connectivity in lung cancer population: An exploratory study. Brain Imaging and Behavior, 12(2), 369–382.
Tao, L., Lin, H., Yan, Y., Xu, X., Wang, L., Zhang, J., & Yu, Y. (2017). Impairment of the executive function in breast cancer patients receiving chemotherapy treatment: A functional MRI study. European Journal of Cancer Care, 26(6), e12553.
Torre, L. A., Bray, F., Siegel, R. L., Ferlay, J., Lortet-Tieulent, J., & Jemal, A. (2015). Global cancer statistics, 2012. CA: a Cancer Journal for Clinicians, 65(2), 87–108.
Wahlund, L.-O., Barkhof, F., Fazekas, F., Bronge, L., Augustin, M., Sjögren, M., et al. (2001). A new rating scale for age-related white matter changes applicable to MRI and CT. Stroke, 32(6), 1318–1322.
Wang, J., Zuo, X., & He, Y. (2010). Graph-based network analysis of resting-state functional MRI. Frontiers in Systems Neuroscience, 4, 16.
Wefel, J. S., Vardy, J., Ahles, T., & Schagen, S. B. (2011). International Cognition and Cancer Task Force recommendations to harmonise studies of cognitive function in patients with cancer. The Lancet Oncology, 12(7), 703–708.
Welzel, T., Niethammer, A., Mende, U., Heiland, S., Wenz, F., Debus, J., & Krempien, R. (2008). Diffusion tensor imaging screening of radiation-induced changes in the white matter after prophylactic cranial irradiation of patients with small cell lung cancer: First results of a prospective study. AJNR. American Journal of Neuroradiology, 29(2), 379–383.
Whitney, K. A., Lysaker, P. H., Steiner, A. R., Hook, J. N., Estes, D. D., & Hanna, N. H. (2008). Is"chemobrain" a transient state? A prospective pilot study among persons with non-small cell lung cancer. The Journal of Supportive Oncology, 6(7), 313–321.
Xuan, H., Chen Gan, W. L., Huang, Z., Wang, L., Jia, Q., Chen, Z., et al. (2017). Altered network efficiency of functional brain networks in patients with breast cancer after chemotherapy. Oncotarget, 8(62), 105648.
Yan, C.-G., Wang, X.-D., Zuo, X.-N., & Zang, Y.-F. (2016). DPABI: Data processing & analysis for (resting-state) brain imaging. Neuroinformatics, 14(3), 339–351.
Funding
This work was supported by a grant from the National Natural Science Foundation of China (No. 81601477), Jiangsu Provincial Special Program of Medical Science (No. BE2017614), Youth Medical Talents of Jiangsu Province (No. QNRC2016062), 14th “Six Talent Peaks” Project of Jiangsu Province (No. YY-079), and Nanjing Outstanding Youth Fund (No. JQX17006).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests
The authors declare that there is no potential conflict of interests regarding the publication of this paper.
Ethical approval
The current study was approved by the Research Ethics Committee of the Nanjing Medical University.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Y., Chen, YC., Hu, L. et al. Chemotherapy-induced functional changes of the default mode network in patients with lung cancer. Brain Imaging and Behavior 14, 847–856 (2020). https://doi.org/10.1007/s11682-018-0030-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-018-0030-y