Abstract
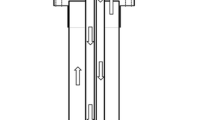
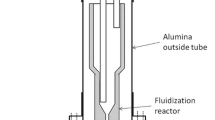
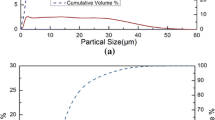
Synthesis of silicon carbide (SiC) by carbothermal reduction of quartz in a CH4–H2–Ar gas mixture was investigated in a laboratory fixed-bed reactor in the temperature range of 1573 K to 1823 K (1300 °C to 1550 °C). The reduction process was monitored by an infrared gas analyser, and the reduction products were characterized by LECO, XRD, and SEM. A mixture of quartz–graphite powders with C/SiO2 molar ratio of 2 was pressed into pellets and used for reduction experiments. The reduction was completed within 2 hours under the conditions of temperature at or above 1773 K (1500 °C), methane content of 0.5 to 2 vol pct, and hydrogen content ≥70 vol pct. Methane partially substituted carbon as a reductant in the SiC synthesis and enhanced the reduction kinetics significantly. An increase in the methane content above 2 vol pct caused excessive carbon deposition which had a detrimental effect on the reaction rate. Hydrogen content in the gas mixture above 70 vol pct effectively suppressed the cracking of methane.











Similar content being viewed by others
References
S.E. Saddow: Silicon carbide biotechnology: a biocompatible semiconductor for advanced biomedical devices and applications, Elsevier Science, Amsterdam, 2011, pp. 5–10.
S.E. Saddow and A. Agarwal: Advances in Silicon Carbide Processing and Applications, Artech House, Norwood, 2004, pp. 2–27.
H.P. Martin, R. Ecke, and E. Miiller: J. Eur. Ceram. Soc., 1998. vol. 18, pp. 1737–42.
K. Järrendahl and R. Davis: Semiconductors & Semimetals, 1998, vol. 52, pp. 1–20.
Y.L. Chiew and K.Y. Cheong: Mater. Sci. Eng. B, 2011, vol. 176, pp. 951–64.
H.N. Baumann: J. Electrochem. Soc., 1952, vol. 99, pp. 109–14.
G.S. Gupta, P. Vasanth Kumar, V.R. Rudolph, and M. Gupta: Metall. Mater. Trans. A, 2001, vol. 32, pp. 1301–8.
Y. Yang, K. Yang, Z.M. Lin, and J.T. Li: Mat. Res. Bull, 2007, vol. 42, pp. 1625–32.
Y. Yang, K. Yang, Z.M. Lin, and J.T. Li: J. Europ. Ceram. Soc., 2009, vol. 29, pp. 175–80.
O. Kordina, C. Hallin, A. Ellison, A.S. Bakin, I.G. Ivanov, A. Henry, R. Yakimova, M. Touminen, A. Vehanen, and E. Janzen: Appl. Phys. Lett., 1996, vol. 69, pp. 1456–8.
A. Henry, I.G. Ivanov, T. Egilsson, C. Hallin, A. Ellison, O. Kordina, U. Lindefelt, and E. Janzen: Diam. Relat. Mater., 1997, vol. 6, 1289–92.
Q.G. Fu, H.J. Li, X.H. Shi, K.Z. Li, J. Wei, and Z.B. Hu: Matetr. Chem. Phys., 2006, vol. 100, pp: 108–11.
O. Ostrovski and G. Zhang: AIChE J., 2006, vol. 52, pp. 300–10.
G. Zhang and O. Ostrovski: Metall. Mater. Trans. B, 2000, vol. 31B, pp. 129–39.
N. Anacleto, O. Ostrovski, and S. Ganguly: ISIJ Int., 2004, vol. 44, 1480–7.
N. Anacleto and O. Ostrovski: Metall. Mater. Trans. B, 2004, vol. 35B: 609–15.
J. Zhang and O. Ostrovski: Ironmak. Steelmak., 2002, vol. 29, pp. 15–21.
J. Zhang and O. Ostrovski: ISIJ Int., 2001, vol. 41, pp. 333–9.
H.C. Lee, S. Dhage, M.S. Akhtar, D.H. Kwak, W.J. Lee, C.Y. Kim and O.B. Yang: Curr. Appl. Phys., 2010, vol. 10, pp. S218–21.
S. Cetinkaya and S. Eroglu: Int. J. Refract. Met. H., 2011, vol. 29, pp. 566–72.
A. Agarwal and U. Pal: Metall. Mater. Trans. B, 1999, vol. 30, pp. 295–306.
B.M. Moshtaghioun, A. Monshi, M.H. Abbasi, and F. Karimzadeh: Int. J. Refract. Met. H., 2011, vol. 29, pp. 645–50.
E. DalMartello, G. Tranell, S. Gaal, O.S. Raaness, K. Tang and L. Arnberg: Metall. Mater. Trans. B, 2011, vol. 42B, pp. 939–50.
X. Li, G. Zhang, K. Tang, O. Ostrovski, and R. Tronstad: Metall. Mater. Trans. B, 2015. vol. 46, pp. 1343–52.
A. Schei and S.H. Halvorsen: Proceedings from the Kjetil Motzfeldt Symposium, Trondheim, Norway. 1991. pp. 41–46.
A.C.D. Chaklader and A.L. Roberts: J. Am. Ceram. Soc., 1961, vol. 44, pp. 35–41.
X. Wan, G. Zhang, O. Ostrovski, and H. Aral: Proceedings of the Thirteenth International Ferroalloys Congress, Karaganda, Kazakhstan. pp. 739–48.
G.A. Bootsma, W.F. Knippenberg, and G. Verspui: J. Cryst. Growth, 1971, vol. 11, pp. 297–309.
H.J. Choi and J.G. Lee: J. Mater. Sci., 1995, vol. 30. pp. 1982–6.
G. Urretavizcaya and J.M. Petro Lopez: J. Mater. Res., 1994, vol. 9, pp. 2981–6.
Acknowledgments
This research was supported under the Australian Research Council’s Linkage Projects funding scheme (Project No. LP100100866) and the Norwegian Research Council under the project Kiselrox (Project No. 228722/O30). The authors would also like to thank the Mark Wainwright Analytical Centre at the University of New South Wales for LECO analysis and the Electron Microscopy Centre (EMC) at the University of Wollongong for the electron microscopy characterization.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted January 27, 2015.
Rights and permissions
About this article
Cite this article
Li, X., Zhang, G., Tang, K. et al. Carbothermal Reduction of Quartz in Methane–Hydrogen–Argon Gas Mixture. Metall Mater Trans B 46, 2384–2393 (2015). https://doi.org/10.1007/s11663-015-0407-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-015-0407-x