Abstract
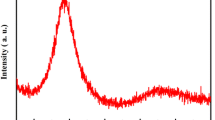
Real-time high-energy X-ray diffraction (HEXRD) was used to investigate the crystallization kinetics and phase selection sequence for constant-heating-rate devitrification of fully amorphous Cu50Zr50, using heating rates from 10 K/min to 60 K/min (10 °C/min to 60 °C/min). In situ HEXRD patterns were obtained by the constant-rate heating of melt-spun ribbons under synchrotron radiation. High-accuracy phase identification and quantitative assessment of phase fraction evolution though the duration of the observed transformations were performed using a Rietveld refinement method. Results for 10 K/min (10 °C/min) heating show the apparent simultaneous formation of three phases, orthorhombic Cu10Zr7, tetragonal CuZr2 (C11b), and cubic CuZr (B2), at 706 K (433 °C), followed immediately by the dissolution of the CuZr (B2) phase upon continued heating to 789 K (516 °C). Continued heating results in reprecipitation of the CuZr (B2) phase at 1002 K (729 °C), with the material transforming completely to CuZr (B2) by 1045 K (772 °C). The Cu5Zr8 phase, previously reported to be a devitrification product in C50Zr50, was not observed in the present study.






Similar content being viewed by others
References
A. Inoue: Acta Mater., 2001, vol. 49, pp. 2645-52.
W. Wang: Mater. Sci. Eng., R, 2004, vol. 44, pp. 45-89.
A. Inoue: Acta Mater., 2000, vol. 48, pp. 279-306.
S. Shin: Mater. Sci. Forum, 2004, vols. 449–452, pp. 945-48.
J.R. Morris, M. Xu, Y.Y. Ye, D.J. Sordelet, and M.J. Kramer: Acta Mater., 2007, vol. 55, pp. 5901-09.
Y.E. Kalay, L.S. Chumbley, and I.E. Anderson: J. Non-Cryst. Solids, 2008, vol. 354, pp. 3040-48.
Y.E. Kalay, C. Yeager, L.S. Chumbley, M.J. Kramer, and I.E. Anderson: J. Non-Cryst. Solids, 2010, vol. 356, pp. 1416-24.
M. Calin, J. Eckert, and L. Schultz: Scripta Mater., 2003, vol. 48, pp. 653-58.
U. Kuhn, J. Eckert, N. Mattern, and L. Schultz: Appl. Phys. Lett., 2002, vol. 80, pp. 2478-80.
J. Eckert, J. Das, S. Pauly, and C. Duhamel: Adv. Eng. Mater., 2007, vol. 9, pp. 443-53.
T. Abe, M. Shimono, M. Ode, and H. Onodera: Acta Mater., 2006, vol. 54, pp. 909-15.
W. Yang, F. Liu, H. Liu, H.F. Wang, Z. Chen, and G.C. Yang: J. Alloys Compd., 2009, vol. 484, pp. 702-07.
A.I. Zaitsev, N.E. Zaitseva, J.P. Alexeeva, S.F. Dunaev, and Y.S. Nechaev: Phys. Chem. Chem. Phys., 2003, vol. 5, pp. 4185-96.
W.H. Wang: Progr. Mater. Sci., 2007, vol. 52, pp. 540-96.
Y. Shen, E. Ma, and J. Xu: J. Mater. Sci. Tech., 2008, vol. 24, pp. 149-52.
T. Fukami, H. Yamamoto, D. Okai, Y. Yokoyama, Y. Yamasaki, and A. Inoue: Mater. Sci. Eng., B, 2006, vol. 131, pp. 1-8.
E. Kneller: Z. Metallkd., 1986, vol. 77, pp. 152-63.
K. Buschow: J. Appl. Phys., 1981, vol. 52, pp. 3319-23.
Z. Altounian: J. Appl. Phys., 1982, vol. 53, pp. 4755-60.
F. Jiang, D.H. Zhang, L.C. Zhang, Z.B. Zhang, L. He, J. Sun, and Z.F. Zhang: Mater. Sci. Eng., A, 2007, vol. 467, pp. 139-45.
J. Eckert, J. Das, S. Pauly, and C. Duhamel: J. Mater. Res., 2007, vol. 22, pp. 443-53.
S.H. Zhou and R.E. Napolitano: Acta Mater., 2010, vol. 58, pp. 2186-96.
Y. Calvayrac, J.P. Chevalier, M. Harmelin, A. Quivy, and J. Bigot: Phil. Mag., B, 1983, vol. 48, pp. 323-32.
G.V. Hillmann and W. Hofmann: Z. Metallkd., 1965, vol. 56, pp. 279-86.
P. Forey, J.L. Glimois, and J.L. Feron: J. Less-Common Met., 1986, vol. 124, pp. 21-27.
A.J. Perry and W. Hugi: J. Inst. Met., 1972, vol. 100, pp. 378-80.
J. Glimois, P. Forey, J.L. Feron: J. Less-Common Met., 1985, vol. 113, pp. 213-24.
D. Arias and J.P. Abriata: Bull. Alloy Phase Diagr., 1990, vol. 11, pp. 452-59.
L. Bsenko: J. Less-Common Met., 1975, vol. 40, pp. 365-66.
J. Gabathuler, P. White, and E. Parthe: Acta Crystallogr., B, 1975, vol. 31, pp. 608-10.
F. Eshelman and J.F. Smith: Acta Crystallogr., B, 1972, vol. 28, pp. 1594-1600.
C. Becle, B. Bourniquel, G. Develey, and M. Saillard: J. Less-Common Met., 1979, vol. 66, pp. 59-66.
L. Bsenko: Acta Crystallogr., B, 1976, vol. 32, pp. 2220-24.
J.M.C.B. Oliveira and I.R. Harris: J. Mater. Sci., 1983, vol. 18, pp. 3649-60.
E. Kneller, Y. Khan, and U. Gorres: Z. Metallkd., 1986, vol. 77, pp. 43-48.
J.M. Joubert, R. Cerny, K. Yvon, M. Latroche, and A. PercheronGuegan: Acta Crystallogr., C, 1997, vol. 53, pp. 1536-38.
M. Kirkpatrick, J.F. Smith, and W.L. Larsen: Acta Crystallogr., A, 1962, vol. 15, pp. 894-903.
J.L. Glimois, C. Becle, G. Develey, and J.M. Moreau: J. Less-Common Met., 1979, vol. 64, pp. 87-90.
E.M. Carvalho and I.R. Harris: J. Mater. Sci., 1980, vol. 15, pp. 1224-30.
D. Schryvers: Scripta Mater., 1997, vol. 36, pp. 1119-25.
M. Kirkpatrick, D.M. Bailey, and J.F. Smith: Acta Crystallogr., A, 1962, vol. 15, pp. 252-55.
G. Ghosh: Acta Mater., 2007, vol. 55, pp. 3347-74.
J.W. Visser: Acta Crystallogr., B, 1977, vol. 33, p. 316.
J.K. Brandon, R.Y. Brizard, P.C. Chieh, R.K. McMillan, and W.B. Pearson: Acta Crystallogr., B, 1974, vol. 30, pp. 1412-17.
M. Nevitt and J.W. Downey: Trans. TMS-AIME, 1962, vol. 224, pp. 195-96.
R.L. Freed and J.B. Vander Sande: J. Non-Cryst. Solids, 1978, vol. 27, pp. 9-28.
H. Okamoto: J. Phase Equilib. Diffus., 2008, vol. 29, p. 204.
S.H. Zhou and R.E. Napolitano: Scripta Mater., 2008, vol. 59, pp. 1143-46.
A.C. Larson and R.B. Von Dreele: LAUR, 2004, pp. 86-748.
B.H. Toby: J. Appl. Crystallogr., 2001, vol. 34, pp. 210-13.
R.E. Napolitano and H. Meco: Metall. Mater. Trans. A, 2004, vol. 35A, pp. 1539-53.
M.J. Kramer, H. Mecco, K.W. Dennis, E. Vargonova, R.W. McCallum, and R.E. Napolitano: J. Non-Cryst. Solids, 2007, vol. 353, pp. 3633-39.
H. Kissinger: Anal. Chem., 1957, vol. 29, pp. 1702-06.
H.J. Eifert, B. Elschner, and K.H.J. Buschow: Phys. Rev. B, 1982, vol. 25, pp. 7441-48.
M. Kasai, J. Saida, M. Matsushita, T. Osuna, E. Matsubara, and A. Inoue: J. Phys.: Condens. Matter., 2002, vol. 14, pp. 13867-77.
D.V. Louzguine-Luzgin, A.R. Yavari, and A. Inoue: Appl. Phys. Lett., 2005, vol. 86, pp. 1-3.
D.V. Louzguine-Luzgin, G. Xie, Q. Zhang, C. Suryanarayana, and A. Inoue: Metall. Mater. Trans. A, 2010 vol. 41A, pp. 1664-69.
Acknowledgments
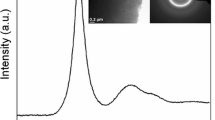
This work was supported by the U.S. Department of Energy, Office of Basic Energy Science, Division of Materials Sciences and Engineering. The research was performed at the Ames Laboratory. Ames Laboratory is operated for the U.S. Department of Energy by Iowa State University under Contract No. DE-AC02-07CH11358. Synchrotron experiments were performed at the Advanced Photon Source, Argonne National Laboratory, under Grant No. DE-AC02-06CH11357. The authors also would like to thank Dr. Y. Eren Kalay, from Ames Laboratory, for assistance with TEM imaging.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted April 12, 2010.
Appendix A
Appendix A
The modeled intensity, I M(Q,ϕ), where Q is \( \left( {Q = {\frac{4\pi \sin \theta }{\lambda }}} \right) \) for the general case can be written as follows:
where the sum is taken over all phases (p), and where L(λ,θ) is the Lorentz polarization factor \( \left( {L\left( {\lambda ,\theta } \right) = {\frac{{P_{h} (\lambda ) + \left( {1 - P_{h} \left( \lambda \right)} \right)\cos^{2} 2\theta }}{{2\sin^{2} \theta \cos \theta }}}} \right), \) A p (λ) is an absorption factor, which is not refined for constant wavelength data because the correction is indistinguishable from thermal motion effects, s p is a phase fraction weighting factor, P p (λ) is a preferred orientation factor, PV p (θ) is a pseudo-Voigt shape function (Type 2), which is a linear combination of Gaussian and Lorentzian functions, m p (θ) is a multiplicity factor, F p (θ) is the structure factor, and P h (λ) is the polarization fraction. For the work described here, we use a constant wavelength and set P h = 0.950. In addition, we take P p and m p as unity. Thus, for the case described here, the model becomes the following:
where the optimization simply involves fitting the remaining three parameters, s p , PV p (θ), and F p (θ), where s p is a scalar,
and
where η determines the weighting of Gaussian (G(θ)) and Lorentzian (L(θ)) components,[49] f p is an atomic scattering factor, \(\overset{\lower0.5em\hbox{$\smash{\scriptscriptstyle\rightharpoonup}$}}{r}_{n} \) gives the atomic positions and the sum is taken over the unit cell. The background intensity, I b (θ) were modeled using a background profile function as follows:
where B 1 is a constant, N is the number of coefficients, and the values of B i are determined by least squares during Rietveld refinement process.
An example of profile fitting using Rietveld refinement is shown by the sequence in Figure 7 for three diffraction peaks corresponding to the CuZr (B2) structure with a space group of Pm-3m with atomic positions of Cu (½, ½, ½) and Zr (0, 0, 0) at 1216 K (943 °C). The refinement is started from the raw data (Figure 7(a)) and done in proper order of applying Lorentz-polarization and background functions (Figure 7(b)), scale factor (Figure 7(c)), structure factor including lattice parameters (Figure 7(d)), Gaussian and Lorentzian peak profile functions (Figure 7(e)), and structure factor including thermal motion using isotropic thermal displacement parameters (Uiso) (Figure 7(f)).
An example of Rietveld refinement sequence of CuZr (B2) phase at 1216 K (943 °C) (a–f); starting with (a) raw data, and refining with (b) Lorentz-polarization factor and background function, (c) phase scalar factor, (d) structure factor including lattice parameters, (e) Gaussian and Lorentzian peak profile functions, and (f) isotropic thermal displacement parameters. The goodness of fit factors are given inset the figures
Rietveld refinement provides an accurate determination of structural properties. After achieving a satisfactory profile fitting, the lattice parameter of B2 is found to change from 3.2810 Å (at room temperature) to 3.29616 Å when heated to 1216 K (943 °C). A specific benefit of the Rietveld analysis is that the refined model provides a quantitative phase analysis, in which the weight fraction w i of each crystalline constituent phase can be determined from the corresponding phase scale parameter, s i according to the following relationship:
where Mu i is the unit cell mass. This analysis is based on the normalization condition \( \sum {w_{i} } = 1.0. \)
Rights and permissions
About this article
Cite this article
Kalay, I., Kramer, M.J. & Napolitano, R.E. High-Accuracy X-Ray Diffraction Analysis of Phase Evolution Sequence During Devitrification of Cu50Zr50 Metallic Glass. Metall Mater Trans A 42, 1144–1153 (2011). https://doi.org/10.1007/s11661-010-0531-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11661-010-0531-9