Abstract
Objective
To assess the efficacy and safety of Gastrosis No.1 compound in the treatment of functional dyspepsia with Spleen (Pi) and Stomach (Wei) deficiency-cold syndrome.
Methods
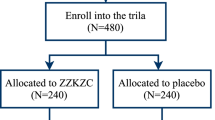
A randomized, double-blind, placebo-controlled trial was performed in 5 centers. Patients with functional dyspepsia (FD) of Spleen-deficiency and qi-stagnation syndrome (162 cases) were randomly assigned to groups given Chinese herbal medicine (CHM) Gastrosis No.1 compound or placebo in a 2:1 ratio. This trial included a 4-week treatment period and a 4-week follow-up period. The outcomes were the dyspepsia symptom scores (measured by total dyspepsia symptom scale and single dyspepsia symptom scale) and syndromes of traditional Chinese medicine score (measured by traditional Chinese medicine syndrome scale). The outcomes were noted at weeks 0, 4 and 8.
Results
Compared with patients in the placebo group, patients in the CHM group showed significant improvement in the dyspepsia symptom scores as rated by patients and investigators (P <0.01), and also showed improvement in syndromes of traditional Chinese medicine score (P <0.01). No serious adverse event was reported. Safety tests obtained after 4 weeks of treatment showed no abnormal values.
Conclusion
CHM Gastrosis No.1 compound was effective and safe in the treatment of functional dyspepsia with Spleen and Stomach deficiency-cold syndrome
Similar content being viewed by others
References
Shaib Y, El-Serag HB. The prevalence and risk factors of functional dyspepsia in a multiethnic population in the United States. Am J Gastroenterol 2004;99:2210–2216.
Bernersen B, Johnsen R, Straume B. Non-ulcer dyspepsia and peptic ulcer: the distribution in a population and their relation to risk factors. Gut 1996;38:822–825.
Hirakawa K, Adachi K, Amano K, Katsube T. Prevalence of non-ulcer dyspepsia in the Japanese population. J Gastroenterol Hepatol 1999;14:1083–1087.
Lu CL, Lang HC, Chang FY. Prevalence and health/social impacts of functional dyspepsia in Taiwan: a study based on the Rome criteria questionnaire survey assisted by endoscopic exclusion among a physical check-up population. Scand J Gastroenterol 2005;40:402–411.
Brook RA, Kleinman NL, Choung RS. Excess comorbidity prevalence and cost associated with functional dyspepsia in an employed population. Dig Dis Sci 2011;13:109–118.
Brook RA, Kleinman NL, Choung RS. Functional dyspepsia impacts absenteeism and direct and indirect costs. Clin Gastroenter Hepatol 2010;8:498–503.
Tack J, Lee KJ. Pathophysiology and treatment of functional dyspepsia. J Clin Gastroenterol 2005;39(5 Suppl 3):S211–216.
Lee KJ, Tack J. Duodenal implications in the pathophysiology of functional dyspepsia. J Neurogastroenterol Motil 2010;16:251–257.
Zeng F, Qin W, Liang F. Abnormal resting brain activity in patients with functional dyspepsia is related to symptom severity. Gastroenterology 2011;141:499–506.
Savarino E, Zentilin P, Dulbecco P. The role of acid in functional dyspepsia. Am J Gastroenterol 2011;106:1168.
Zhang SS, Su DM, Zhao LQ. Systematic review on the efficacy of TCM in the treatment of functional dyspepsia. Chin J Integr Tradit West Med Digestion (Chin) 2011;23:32–34.
Rome Foundation. Guidelines-Rome III diagnostic criteria for functional gastrointestinal disorders. J Gastrointestin Liver Dis 2006;15:307–312.
Zheng XY. Guiding principle of clinical research on new drugs of Chinese medicine in the treament of stuffiness and fullness. In guiding principle of clinical research on new drugs of Chinese medicine (trial implementation). Edited by: Yu XH. Bejing: Chinese Medical Science and Technology Press; 2002:54–58.
Liu X. Experimental design and data processing (fourth). Chin J Difficult Complicated Cases (Chin) 2003;2:55–57.
Zhang YP, Luo JH, Zhou C. The clinical curative effect of Jianpi Oral Liquid on 87 cases of Spleen and Stomach deficiency-weakness syndrome. Chin Tradit Patent Med (Chin) 1999;21:239–242.
Holtmann G, Talley NJ, Liebregts T, Adam B, Parow C. A placebo-controlled trial of itopride in functional dyspepsia. New Engl J Med 2006;354:832–840.
Oshima T, Miwa H. Treatment of functional dyspepsia: where to go and what to do. J Gastroenterol 2006;41:647–653.
Mönkemüller K, Malfertheiner P. Drug treatment of functional dyspepsia. World J Gastroenterol 2006;12:2694–2700.
Hojo M, Miwa H, Yokoyama. Treatment of functional dyspepsia with antianxiety or antidepressive agents: systematic review. J Gastroenterol 2005;40:1036–1042.
Talley NJ, Locke GR, Lahr BD. Predictors of the placebo response in functional dyspepsia. Aliment Pharmacol Ther 2006;23:923–936.
Zhao BG, Sun QF. Modern pharmacological studies and clinic application of Lizhong Bolus. Chin Tradit Patent Med (Chin) 2010;32:1957–1960.
Guo JC, Zhang AJ, Zheng XH, Zhou QF. Experimental study of complex Dangshen ventilation oral solution on intestinal function recovery after operation on abdominal region. J Emerg Tradit Chin Med (Chin) 2007;16:1238–1240.
Song D, Wang ZT, Li LY, Zhong GY. Protective effect of lobetyolin on gastric mucosa of experimental gastric ulcer in rats. J Emerg Tradit Chin Med (Chin) 2008;17:963–964.
Chan LW, Cheah EL, Saw CL. Antimicrobial and antioxidant activities of Cortex Magnoliae Officinalis and some other medicinal plants commonly used in south-east Asia. Chin Med 2008;28:3–15.
Liu CB, Zhang SC, Li QT. The development of modern pharmacological research of rhizoma cyperi. Guangming J Chin Med (Chin) 2009;24:787–788.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Eleventh Five-Year National Key Technology R&D Program (No. 2007BAI20B092)
Rights and permissions
About this article
Cite this article
Zhang, Ss., Zhao, Lq., Wang, Hb. et al. Efficacy of Gastrosis No.1 compound on functional dyspepsia of spleen and stomach deficiency-cold syndrome: A multi-center, double-blind, placebo-controlled clinical trial. Chin. J. Integr. Med. 19, 498–504 (2013). https://doi.org/10.1007/s11655-013-1503-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-013-1503-x