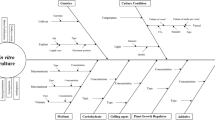
Summary
The aptitude for organogenesis from normal hormone-dependent cultures very commonly decreases as the tissues are serially subcultured. The reasons for the loss of regenerative ability may vary under different circumstances: genetic variation in the cell population, epigenetic changes, disappearance of an organogenesis-promoting substance, etc. The same reasons may be evoked for the progressive and eventually irreversible loss of organogenic totipotency in the course of neoplastic progressions from hormone-independent tumors and hyperhydric teratomas to cancers. As in animal cells, plant cells at the end of a neoplastic progression have probably undergone several independent genetic accidents with cumulative effects. They indeed are characterized by atypical biochemical cycles from which they are apparently unable to escape. The metabolic changes are probably not the primary defects that cause cancer, rather they may allow the cells to survive. How these changes, namely an oxidative stress, affect organogenesis is not known. The literature focuses on somatic mutations and epigenetic changes that cause aberrant regulation of cell cycle genes and their machinery.
Similar content being viewed by others
References
Alberts, B.; Bray, D.; Lewis, J.; Raff, M.; Roberts, K.; Watson, J. D. Cancer. In: Alberts, B.; Bray, D.; Lewis, J.; Raff, M.; Roberts, K.; Watson, J. D. eds. Molecular biology of the cell. New York: Garland Publishers; 1989; 1187; 1216.
Altman, A.; Kaur-Sawhney, R.; Galston W. A. Stabilization of oat leaf protoplasts through polyamine-mediated inhibition of senescence. Plant Physiol. 60:570–574; 1977.
Angelini, R.; Federico, R. Histochemical evidence of polyamine oxidation and generation of hydrogen peroxide in the cell wall. J. Plant Physiol. 135:212–217; 1989.
Anonymous. Habituation, hyperhydricity and plant cancer. Agricell Rep. 25:29, 1995.
Arbillot, J.; Le Saos, J.; Billard, J. P.; Boucaud, J.; Gaspar, Th. Changes in fatty acid and lipid composition in normal and habituated sugar beet calli. Phytochemistry 30:491–494; 1991.
Aribaud, M.; Kevers, C.; Martin-Tanguy, J.; Gaspar, Th. Low activity of amine-oxidases and accumulation of conjugated polyamines in disfavour of organogenic programs in Chrysanthemum leaf disc explants. Plant Cell Tiss. Org. Cult. 55:85–94; 1999.
Bayer, M. H. Genetic tumors: physiological aspects of tumor formation in interspecies hybrids. In: Kahl, G.; Schell, J. S., eds. Molecular biology of plant tumors. New York: Academic Press; 1982; 33–67.
Bennici, A.; Caffaro, L. Caryological behaviour during the first phases of dedifferentiation and habituation in Nicotiana bigelovii. Protoplasma 124:130–136; 1985.
Binns, A.; Meins, F. Jr. Chromosome number and the degree of cytokinin habituation of cultured tobacco pith cells. Protoplasma 103:179–187; 1980.
Bisbis, B.; Kevers, C.; Crèche, J.; Rideau, M.; Gaspar, Th. Differential growth dependency of normal and habituated sugarbeet cell lines upon endogenous ethylene production and exogenous ethylene application. Physiol. Plant 103:201–208; 1998.
Bisbis, B.; Kevers, C.; Gaspar, Th. Atypical TCA cycle and replenishment in a non-photosynthetic fully habituated sugarbeet callus overproducing polyamines. Plant Physiol. Biochem. 35:363–368; 1997.
Bornman, C.; Vogelman, T. C. Effect of rigidity of gel medium on benzyladenine-induced adventitious bud formation and vitrification in Picea abies. Physiol. Plant 61:505–512; 1984.
Braun, A. C. Plant tumors. Biochim. Biophys. Acta 516:167–191; 1978.
Braun, A. C.; Stonier, T. Morphology and physiology of plant tumors. Protoplasmatologia 10:1–93; 1958.
Burdon, R.H. Control of cell proliferation by reactive oxygen species. Biochem. Soc. Trans. 24:1028–1032; 1996.
Campell, B. R.; Town, C. D. Physiology of hormone autonomous tissue derived from radiation-induced tumors of Arabidopsis thaliana. Plant Physiol. 97:1166–1173; 1991.
Collin, H. A.; Edwards, S. Plant cell culture. Oxford: Bios Publishers Ltd; 1998.
Coumans, M.; Ribet, J. Peroxidases as a marker of rejuvenation and vitrification in globe artichoke. In: Welinder, K. G.; Rasmussen, S. K.; Penel, C.; Greppin, H. eds. Plant peroxidases, biochemistry and physiology. Geneva, Switzerland: University Publishers; 1993; 429–434.
Crèvecoeur, M.; Hagège, D.; Catesson, A. M.; Greppin, H.; Gaspar, Th. Ultrastructural characteristics of cells from normal and habituated sugar beet calli. Plant Physiol. Biochem. 30:87–95; 1992.
Crèvecoeur, M.; Kevers, C.; Greppin, H.; Gaspar, Th. A comparative and cytological characterization of normal and habituated sugarbeet calli. Biol. Plant 29:1–6; 1987.
Dang, C. V.; Semenza, G. L. Oncogenic alterations of metabolism. Trends Biochem. Sci. 24:68–72; 1999.
Dianzani, M. V. Lipid peroxidation and cancer. A critical reconsideration. Tumori 75:351–357; 1989.
De Greef, W.; Jacobs, M. In vitro culture of the sugarbeet: description of a cell line with high regeneration capacity. Plant Sci. Lett. 17:55–61; 1979.
Doonan, J.; Hunt, T. Why don't plants get cancer? Nature 380:481–482; 1996.
Drolet, G.; Dumbroff, E. B.; Legge, R. L.; Thompson, J. E. Radical scavenging properties of polyamines. Phytochemistry 25:367–371; 1986.
Droual, A. M.; Hamdi, S.; Crèche, J.; Kevers, C.; Rideau, M. Autonomy to plant growth regulators and gene expression in periwinkle culture in vitro. J. Plant Physiol. 153:623–630; 1998.
Feutry, S.; Poder, D.; Hagège, D. Enhancement of diacylglycerol level and inositol phosphate turnover in fully habituated sugarbeet cell line. Plant Physiol. Biochem. 33:115–120; 1995.
Finnegan, E. J.; Genger, R. K.; Peacock, W. J.; Dennis, E. S. DNA methylation in plants. Ann. Rev. Plant Physiol. Plant Mol. Biol. 49:223–247; 1998.
Floh, E. I. S.; Handro, W. Variation of histological patterns in tobacco callus during successive subcultures. Can. J. Bot. 63:1794–1800; 1985.
Franck, T.; Kevers, C.; Gaspar, Th. Protective enzymatic systems against activated oxygen species compared in normal and vitrified shoots of Prunus avium L. raised in vitro. Plant Growth Regul. 16:253–256; 1995.
Franck, T.; Kevers, C.; Penel, C.; Gaspar, Th.; Hausman, J. F.; Greppin, H. Reducing properties and markers of lipid peroxidation in normal and hyperhydrating shoots of Prunus avium L. J. Plant Physiol. 153:339–346; 1998.
Gaspar, Th. Vitrification in micropropagation. In: Bajaj, Y. P. S. eds. Hightech and micropropagation (Biotechnology in agriculture and forestry, Vol. 17). Berlin: Springer-Verlag; 1991; 117–126.
Gaspar, Th. The concept of cancer in in vitro plant cultures and the implication of habituation to hormones and hyperhydricity. Plant Tiss. Cult. and Biotechnol. 1:126–136; 1995.
Gaspar, Th. Plants can get cancer. Plant Physiol. Biochem. 36:203–204; 1998.
Gaspar, Th. Tumours, neoplastic progressions and cancers in plants. In: Sirnad, M.; Pec, R.; Beck, E. eds. Advances in regulation of plant growth and development. Prague: Peres Publishers; 1999; 183–192.
Gaspar, Th.; Bisbis, B.; Kevers, C.; Penel, C.; Greppin, H.; Le Dily, F.; Billard, J. P.; Huault, C.; Garnier, F.; Rideau, M.; Foidart, J. M. Atypical metabolisms and biochemical cycles imposing the cancerous state on plant cells. Plant Growth Regul. 24:135–144; 1998.
Gaspar, Th.; Hagège, D.; Kevers, C.; Penel, C.; Crèvecoeur, M.; Engelmann, I.; Greppin, H.; Foidart, J. M. When plant teratomas turn into cancers in the absence of pathogens. Physiol. Plant 83:696–701; 1991.
Gaspar, Th.; Kevers, C.; Bisbis, B.; Crèvecoeur, M.; Penel, C.; Greppin, H.; Le Dily, F.; Billard, J.P.; Huault, C.; Foidart, J. M. Cancer végétal in vitro: aspects morphogénétiques et biochimiques. In: Demarly, Y.; Dubois, J. eds. Quel avenir pour l'amélioration des plantes. Paris, France: John Libbey Eurotext: 1995; 165–171.
Gaspar, Th.; Kevers, C.; Bisbis, B.; Penel, C.; Greppin, H.; Garnier, F.; Rideau, M.; Huault, C.; Billard, J. P.; Foidart, J. M. Shemin pathway and peroxidase deficiency in a fully habituated and fully heterotrophic nonorganogenic sugarbeet callus: an adaptative strategy or the consequence of modified hormonal balances and sensitivities in these cancerous cells? A review and reassessment. Cell Prolif. 32:249–270; 1999.
Gaspar, Th.; Kevers, C.; Hausman, J. F. Indissociable chief factors in the inductive phase of adventitious rooting. In: Altman, A.; Waisel, Y. eds. Biology of root formation and development. New York: Plenum Press, 1997; 55–63.
George, E. F. Plant propagation by tissue culture. Edington, UK: Exegetics Ltd; 1993.
Hagège, D. Protooncogenes in plants: Widespread conserved genes for which roles? Plant Physiol. Biochem. 31:621–629; 1993.
Hagège, D.; Kevers, C.; Salabert, Ph.; Gaspar, Th. Protective systems against activated oxygen species compared in normal and fully habituated nonorganogenic sugarbeet callus. In Vitro Cell. Dev. Biol. 28P:143–147; 1992.
Hahne, B. Fleck, J.; Hahne, G. Mesophyll protoplasts of Avena sativa: characterization of the first cell cycles. Protoplasma 167:49–54; 1992.
Halliwell, B. Free radicals, proteins and DNA: oxidative damage versus redox regulation. Biochem. Soc. Trans. 24:1023–1027; 1996.
Hartwell, L. Defects in a cell cycle check point may be responsible for the genomic instability of cancer cells. Cell 71:543–546; 1992.
Hausman, J. F.; Kevers, C.; Evers, D.; Gaspar, Th. Conversion of putrescine into γ-aminobutyric acid, an essential pathway for root formation by poplar shoots in vitro. In: Altman, A.; Waisel, Y. eds. Biology of root formation and development. New York: Plenum Press, 1997; 133–139.
Hervagault, J. F.; Ortoleva, P. J.; Ross, J. A plausible model for reversal of neoplastic transformation in plants based on multiple steady states. Proc. Natl. Acad. Sci. USA 88:10797–10800; 1991.
Hildrum, K. I.; Scanlan, R. A.; Libbey, L. M.In: Walker, E. A.; Bogovski, P.; Gricinte, L. eds. Environmental N-nitroso compounds—analysis and formation. IARC;14:205–214; 1976.
Jackson, J. A.; Lyndon, R. F. Habituation: cultural curiosity or developmental determinant. Physiol. Plant 79:579–583; 1990.
John, A. Vitrification in Sitka spruce cultures. In: Withers, L.; Alderson, P. G. eds. Plant tissue culture and its agricultural applications. London: Butterworths; 1986; 167–174.
Kataeva, N. V.; Alexandrova, I. G.; Butenko, R. G.; Dragaviceva, E. V. Effect of applied and internal hormones on vitrification and apical necrosis of different plant cultures in vitro. Plant Cell Tiss. Org. Cult. 27:149–154; 1991.
Keller, A. V.; Frey Koonen, N.; Wingender, R.; Schnabl, H.: Ultrastructure of sunflower protoplast derived callus differing in their regenerative potential. Plant Cell Tiss. Org. Cult. 37:277–285; 1994.
Kevers, C.; Bisbis, B.; Faivre-Rampant, O.; Gaspar, Th. Putrescine metabolism in a fully habituated nonorganogenic sugar beet callus and its relationship with growth. J. Plant Physiol. 154: 503–508; 1999a.
Kevers, C.; Bisbis, B.; Franck, T.; Le Dily, F.; Huault, C.; Billard, J. P.; Foidart, J. M.; Gaspar, Th. On the possible causes of polyamine accumulation in in vitro plant tissues under neoplasic progression. In: Greppin, H.; Penel, C.; Simon, P. eds. Travelling shot on plant development. Geneva, Switzerland: University Publishers; 1997; 63–71.
Kevers, C.; Bisbis, B.; Le Dily, F.; Billard, J. P.; Huault, C.; Gaspar, Th. Darkness improves growth and delays necrosis in a non chlorophyllous habituated sugarbeet callus. Biochemical changes. In Vitro Cell. Dev. Biol.-Plant 31:122–126; 1995.
Kevers, C.; Bisbis, B.; Penel, C.; Greppin, H.; Dommès, J.; Gaspar, Th. Changes in the levels of hormones and related enzyme activities in the course of a neoplastic progression in sugarbeet cells in culture. A critical appraisal. Curr. Top. Phytochem (In press); 1999b.
Kevers, C.; Coumans, M.; De Greef, W.; Hofinger, M.; Gaspar, Th. Habituation in sugarbeet callus: auxin content, auxin protectors, peroxidase pattern and inhibitors. Physiol. Plant 51:281–286; 1981a.
Kevers, C.; Coumans, M.; De Greef, W.; Jacobs, M.; Gaspar, Th. Organogenesis in habituated sugarbeet callus: auxin content and protectors, peroxidase pattern and inhibitors. Z. Pflanzenphysiol 101:79–87; 1981b.
Kevers C.; Greimers, R.; Franck, T.; Bisbis, B.; Dommès, J.; Gaspar, Th. Flow cytometry estimation of nuclear size and ploidy level of habituated calli of sugarbeet. Biol. Plant 42:321–332; 1999c.
Köves, E.; Szabo, M. Ethylene production in habituated and auxin requiring tobacco callus cultures. Does ethylene play a role in habituation? Physiol. Plant 69:351–355; 1987.
Lambé, P.; Mutambel, H. S. N.; Fouché, J. G.; Deltour, R.; Foidart, J. M.; Gaspar Th. DNA methylation as a key process in regulation of organogenic totipotency and plant neoplastic progression? In Vitro Cell. Dev. Biol.-Plant 33:155–162; 1997.
Le Dily, F.; Billard, J. P.; Gaspar, Th.; Huault, C. Disturbed nitrogen metabolism associated with the hyperhydric status of fully habituated callus of sugarbeet. Physiol. Plant 88:129–134; 1993b.
Le Dily, F.; Billard, J. P.; Huault, C.; Kevers, C.; Gaspar, Th. Fully habituated sugarbeet callus: under permanent stress? In Vitro Cell Dev. Biol. 29P:149–154; 1993a.
Leshem, B. The carnation succulent plantlet, a stable teratological growth. Ann. Bot. 52:873–876; 1983.
Leshem, B.; Sachs, T. Vitrified Dianthus teratoma in vitro due to growth factor imblance. Ann. Bot. 56:613–617; 1985.
Liners, F.; Gaspar, Th.; Van Cutsem, P. Acetyl- and methyl-esterification of pectins of friable and compact calli: consequences for intercellular adhesions. Planta 192:545–556; 1994.
Lutz, A. Aptitudes morphogénétiques des cultures de tissues d'origine unicellulaire. In: Hirth, M. L.; Morel, G. eds. Les cultures de tissus de plantes. Coll. Int. CNRS no. 193. Paris: CNRS; 1971; 163–168.
Meins, F. Jr. Habituation of cultured plant cells. In: Kahl, G.; Schell, J. S. eds. Molecular biology of plant tumors. New York: Academic Press; 1982; 3–31.
Meins, F. Jr. Habituation: heritable variation in the requirement of cultured plant cells for hormones. Ann. Rev. Genet. 23:395–408; 1989.
Mérillon, J. M.; Filali, M.; Dupéron, P.; Montagu, M.; Chénieux, J. C.; Rideau, M. Effect of 2,4-dichlorophenoxyacetic acid and habituation on lipid and protein composition of microsomal membranes from periwinkle cell suspensions. Plant Physiol. Biochem. 33:443–451; 1995.
Mousdale, D. M. A.; Fidgeon, C.; Wilson, G. Auxin content and growth patterns in auxin-dependent and auxin-autotrophic plant cell and tissue cultures. Biol. Plant 27:257–264; 1985.
Nandi, S. K.; Palni, L. M. S.; Parker, C. W. Dynamics of endogenous cytokinins during the growth cycle of a hormone-autotrophic genetic tumor line of tobacco. Plant Physiol. 94:1084–1089; 1990.
Pengelly, W. L. Neoplastic progression in plants. In: Kaiser, H. E. eds. Cancer growth and progression, Dordrecht, The Netherlands: Kluwer Academic Publishers; 1989; 5; 15–23.
Persinger, S. M.; Town, C. D. Isolation and characterization of hormone-autonomous tumors of Arabidopsis thaliana. J. Exp. Bot. 42:1363–1370; 1991.
Pierik, R. L. M. In vitro culture of higher plants. Dordrecht, The Netherlands: Martinus Nijhoff Publishers; 1989.
Porciani, S.; Becciolini, A.; Lanini, A.; Balzi, M.; Boanini, P.; Mauri, P.; Mangeri, A. Polyamines and proliferative activity in tumor tissues. Cell Prolif. 26:490; 1993.
Robards, A. W.; Lucas, W. J. Plasmodesmata. Ann. Rev. Plant Physiol. 41:369–419; 1990.
Sacristan, M. D.; Melchers, G. The caryological analysis of plants regenerated from tumorous and other callus cultures of tobacco. Mol. Gen. Genet. 105:317–333; 1969.
Sacristan, M. D.; Melchers, G. Regeneration of plants from habituated and Agrobacterium-transformed single-cell clones of tobacco. Mol. Gen. Genet. 152:111–117; 1977.
Siedow, J. N.; Stitt, M. Plant metabolism; where are all those pathways leading us? Curr. Opin. Biol. 1:197–200; 1998.
Smith, T. A. Polyamines. Ann. Rev. Plant Physiol. 36:117–143; 1985.
Strnad, M.; Krystov, V. V.; Havlicek, L. Control of tumour development in plants and animals: a comparative treatise. In: Strnad, M.; Lec, L.; Beck, E. eds. Advances in, regulation of plant growth and development, Prague: Peres Publishers; 1999; 193–202.
Syono, K.; Fujita T. Habituation as a tumorous state that is interchangeable with a normal state in plant cells. Int. Rev. Cytol. 152:265–299; 1994.
Szabo, M.; Köves, E.; Somogyi, I. Development of auxin autotrophy in Nicotiana tabacum callus cultures. Physiol. Plant 90:348–352; 1994.
Tacchini, P.; Fink, A.; Xue, G. X.; Gaspar, Th.; Greppin, H. Analysis of proteins of a fully habituated nonorganogenic sugarbeet callus and a hormone-dependent one by high-performance 2-D gel electrophoresis. Plant Physiol. Biochem. 33:361–366; 1995.
Teutonico, R. A.; Dudley, M. W.; Orr, J. D.; Lynn, D. G.; Binns, A. N. Activity and accumulation of cell division-promoting phenolics in tobacco tissue cultures. Plant Physiol. 97:288–297; 1991.
Tucker, E. B. Analytical studies of dye coupling between plant cells. In: Robards, A. W.; Lucas, W. J.; Putts, H. J. eds. Parallels in cell to cell junctions in plants and animals. Berlin: Springer-Verlag; 1990; 239–247.
Vaca, C. E.; Wilhelm, J.; Harms-Ringahl, M. Interactions of lipid peroxidation products with DNA. A review. Mutat. Res. 195:137–149; 1998.
Winston, G. W. Physiochemical basis for free radical formation in cells: production and defenses. In: Alscher, R. G.; Cumming, J. R. eds. Stress responses in plants: adaptation and acclimation mechanisms. New York: Wiley-Liss Inc.; 1990; 57–86.
Ziv, M. Vitrification: morphological and physiological disorders of in vitro plants. In: Debergh, P.; Zimmerman, R. H. eds. Micropropagation, technology and application. Dordrecht, The Netherlands: Kluwer Academic Publishers; 1991; 45; 69.
Zoglauer, K.; Koschuckova, S.; Girg, P.; Göring, H. Phytohormone Autotrophie eines Spross-Kallus-Systems. In: Göring, H.; Paul, E. eds. Pflanzische Gewebekultur. Coll. Pflanzenphysiol. Berlin, Germany: Humboldt University; 1981; 133–139.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gaspar, T., Kevers, C., Bisbis, B. et al. Special symposium: In vitro plant recalcitrance loss of plant organogenic totipotency in the course of In vitro neoplastic progression. In Vitro Cell.Dev.Biol.-Plant 36, 171–181 (2000). https://doi.org/10.1007/s11627-000-0033-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11627-000-0033-3