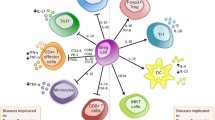
Summary
B cells are a heterogeneous population, which have distinct functions of antigen presentation, activating T cells, and secreting antibodies, cytokines as well as protease. It is supposed that the balance among these B cells subpopulation (resting B cells, activated B cells, Bregs, and other differentiated B cells) will determine the ultimate role of B cells in tumor immunity. There has been increasing evidence supporting opposite roles of B cells in tumor immunity, though there are no general acceptable phenotypes for them. Recent years, a new designated subset of B cells identified as Bregs has emerged from immunosuppressive and/or regulatory functions in tumor immune responses. Therefore, transferring activated B cells would be possible to become a promising strategy against tumor via conquering the immunosuppressive status of B cells in future. Understanding the potential mechanism of double-edge role of B cells will help researchers utilize activated B cells to improve their anti-tumor response. Moreover, the molecular pathways related to B cell differentiation are involved in its tumor-promoting effect, such as NF-κB, STAT3, BTK. So, we review the molecular and signaling pathway mechanisms of B cells involved in both tumor-promoting and tumor-suppressive immunity, in order to help researchers optimize B cells to fight cancer better.
Similar content being viewed by others
References
Hu X, Zhang J, Wang J, et al. Landscape of B cell immunity and related immune evasion in human cancers. Nat Genet, 2019,51(3):560–567
Tokunaga R, Naseem M, Lo JH, et al. B cell and B cell-related pathways for novel cancer treatments. Cancer Treat Rev, 2019,73:10–19
Bonizzi G, Karin M. The two NF-kappaB activation pathways and their role in innate and adaptive immunity. Trends Immunol, 2004,25(6):280–288
Sun SC. The non-canonical NF-kappaB pathway in immunity and inflammation. Nat Rev Immunol, 2017,17(9):545–558
Ammirante M, Luo JL, Grivennikov S, et al. B-cell-derived lymphotoxin promotes castration-resistant prostate cancer. Nature, 2010,464(7286):302–305
Ammirante M, Kuraishy AI, Shalapour S, et al. An IKKalpha-E2F1-BMI1 cascade activated by infiltrating B cells controls prostate regeneration and tumor recurrence. Genes Dev, 2013,27(13):1435–1440
Wuerzberger-Davis SM, Chen Y, Yang DT, et al. Nuclear export of the NF-kappaB inhibitor IkappaBalpha is required for proper B cell and secondary lymphoid tissue formation. Immunity, 2011,34(2):188–200
Huynh J, Etemadi N, Hollande F, et al. The JAK/STAT3 axis: A comprehensive drug target for solid malignancies. Semin Cancer Biol, 2017,45:13–22
Yang C, Lee H, Pal S, et al. B cells promote tumor progression via STAT3 regulated-angiogenesis. PLoS One, 2013,8(5):e64159
Zhang C, Xin H, Zhang W, et al. CD5 Binds to Interleukin-6 and Induces a Feed-Forward Loop with the Transcription Factor STAT3 in B Cells to Promote Cancer. Immunity, 2016,44(4):913–923
Ren Y, Yu K, Sun S, et al. JSI124 inhibits breast cancer cell growth by suppressing the function of B cells via the downregulation of signal transducer and activator of transcription 3. Oncol Lett, 2014,8(2):928–932
Olkhanud PB, Damdinsuren B, Bodogai M, et al. Tumor-evoked regulatory B cells promote breast cancer metastasis by converting resting CD4(+) T cells to T-regulatory cells. Cancer Res, 2011,71(10):3505–3515
Lee-Chang C, Bodogai M, Martin-Montalvo A, et al. Inhibition of breast cancer metastasis by resveratrol-mediated inactivation of tumor-evoked regulatory B cells. J Immunol, 2013,191(8):4141–4151
Gunderson AJ, Kaneda MM, Tsujikawa T, et al. Bruton Tyrosine Kinase-Dependent Immune Cell Cross-talk Drives Pancreas Cancer. Cancer Discov, 2016,6(3):270–285
Seiler T, Dreyling M. Bruton’s tyrosine kinase inhibitors in B-cell lymphoma: current experience and future perspectives. Expert Opin Investig Drugs, 2017,26(8): 909–915
Hamze M, Desmetz C, Guglielmi P. B cell-derived cytokines in disease. Eur Cytokine Netw, 2013,24(1):20–26
Schioppa T, Moore R, Thompson RG, et al. B regulatory cells and the tumor-promoting actions of TNF-alpha during squamous carcinogenesis. Proc Natl Acad Sci USA, 2011,108(26):10 662–10 667
Inoue S, Leitner WW, Golding B, et al. Inhibitory effects of B cells on antitumor immunity. Cancer Res, 2006,66(15):7741–7747
Tao H, Lu L, Xia Y, et al. Antitumor effector B cells directly kill tumor cells via the Fas/FasL pathway and are regulated by IL-10. Eur J Immunol, 2015,45(4):999–1009
Horikawa M, Minard-Colin V, Matsushita T, et al. Regulatory B cell production of IL-10 inhibits lymphoma depletion during CD20 immunotherapy in mice. J Clin Invest, 2011,121(11):4268–4280
Gorosito Serran M, Fiocca Vernengo F, Beccaria CG, et al. The regulatory role of B cells in autoimmunity, infections and cancer: Perspectives beyond IL10 production. FEBS Lett, 2015,589(22):3362–3369
Pylayeva-Gupta Y, Das S, Handler JS, et al. IL35-Producing B Cells Promote the Development of Pancreatic Neoplasia. Cancer Discov, 2016,6(3):247–255
Ou Z, Wang Y, Liu L, et al. Tumor microenvironment B cells increase bladder cancer metastasis via modulation of the IL-8/androgen receptor (AR)/MMPs signals. Oncotarget, 2015,6(28):26 065–26 078
Lee KE, Spata M, Bayne LJ, et al. Hif1a Deletion Reveals Pro-Neoplastic Function of B Cells in Pancreatic Neoplasia. Cancer Discov, 2016,6(3):256–269
Seton-Rogers S. Pancreatic cancer: Spotlight on B cells. Nat Rev Cancer, 2016,16(2):67
Forte G, Sorrentino R, Montinaro A, et al. Inhibition of CD73 improves B cell-mediated anti-tumor immunity in a mouse model of melanoma. J Immunol, 2012,189(5):2226–2233
Kemp TJ, Moore JM, Griffith TS. Human B cells express functional TRAIL/Apo-2 ligand after CpG-containing oligodeoxynucleotide stimulation. J Immunol, 2004,173(2):892–899
Iglesia MD, Vincent BG, Parker JS, et al. Prognostic B-cell signatures using mRNA-seq in patients with subtype-specific breast and ovarian cancer. Clin Cancer Res, 2014,20(14):3818–3829
Tadmor T, Zhang Y, Cho HM, et al. The absence of B lymphocytes reduces the number and function of T-regulatory cells and enhances the anti-tumor response in a murine tumor model. Cancer Immunol Immunother, 2011,60(5):609–619
Zhang Y, Eliav Y, Shin SU, et al. B lymphocyte inhibition of anti-tumor response depends on expansion of Treg but is independent of B-cell IL-10 secretion. Cancer Immunol Immunother, 2013,62(1):87–99
Shah S, Divekar AA, Hilchey SP, et al. Increased rejection of primary tumors in mice lacking B cells: inhibition of anti-tumor CTL and TH1 cytokine responses by B cells. Int J Cancer, 2005,117(4):574–586
Guy TV, Terry AM, Bolton HA, et al. Pro- and anti-tumour effects of B cells and antibodies in cancer: a comparison of clinical studies and preclinical models. Cancer Immunol Immunother, 2016,65(8):885–896
Kim SJ, Caton M, Wang C, et al. Increased IL-12 inhibits B cells’ differentiation to germinal center cells and promotes differentiation to short-lived plasmablasts. J Exp Med, 2008,205(10):2437–2448
Xia Y, Tao H, Hu Y, et al. IL-2 augments the therapeutic efficacy of adoptively transferred B cells which directly kill tumor cells via the CXCR4/CXCL12 and perforin pathways. Oncotarget, 2016,7(37):60 461–60 474
Li Q, Lao X, Pan Q, et al. Adoptive transfer of tumor reactive B cells confers host T-cell immunity and tumor regression. Clin Cancer Res, 2011,17(15):4987–4995
Li Q, Grover AC, Donald EJ, et al. Simultaneous targeting of CD3 on T cells and CD40 on B or dendritic cells augments the antitumor reactivity of tumor-primed lymph node cells. J Immunol, 2005,175(3):1424–1432
Li Q, Teitz-Tennenbaum S, Donald EJ, et al. In vivo sensitized and in vitro activated B cells mediate tumor regression in cancer adoptive immunotherapy. J Immunol, 2009,183(5):3195–3203
DiLillo DJ, Yanaba K, Tedder TF. B cells are required for optimal CD4+ and CD8+ T cell tumor immunity: therapeutic B cell depletion enhances B16 melanoma growth in mice. J Immunol, 2010,184(7):4006–4016
Kim S, Fridlender ZG, Dunn R, et al. B-cell depletion using an anti-CD20 antibody augments antitumor immune responses and immunotherapy in nonhematopoetic murine tumor models. J Immunother, 2008,31(5):446–457
Schwartz M, Zhang Y, Rosenblatt JD. B cell regulation of the anti-tumor response and role in carcinogenesis. J Immunother Cancer, 2016,4:40
Sarvaria A, Madrigal JA, Saudemont A. B cell regulation in cancer and anti-tumor immunity. Cell Mol Immunol, 2017,14(8):662–674
Hagn M, Jahrsdorfer B. Why do human B cells secrete granzyme B? Insights into a novel B-cell differentiation pathway. Oncoimmunology, 2012,1(8):1368–1375
Bao Y, Cao X. The immune potential and immunopathology of cytokine-producing B cell subsets: a comprehensive review. J Autoimmun, 2014,55:10–23
Wennhold K, Weber TM, Klein-Gonzalez N, et al. CD40-activated B cells induce anti-tumor immunity in vivo. Oncotarget, 2017,8(17):27 740–27 753
Shimabukuro-Vornhagen A, Schlosser HA, Gryschok L, et al. Characterization of tumor-associated B-cell subsets in patients with colorectal cancer. Oncotarget, 2014,5(13):4651–4664
Wang JZ, Zhang YH, Guo XH, et al. The double-edge role of B cells in mediating antitumor T-cell immunity: Pharmacological strategies for cancer immunotherapy. Int Immunopharmacol, 2016,36:73–85
Shalapour S, Font-Burgada J, Di Caro G, et al. Immunosuppressive plasma cells impede T-cell-dependent immunogenic chemotherapy. Nature, 2015, 521(7550):94–98
Author information
Authors and Affiliations
Corresponding authors
Additional information
Conflict of Interest Statement
The authors declare there is no conflicts of interest regarding the publication of this paper.
Rights and permissions
About this article
Cite this article
Zhao, Kl., Yang, Xj., Jin, Hz. et al. Double-edge Role of B Cells in Tumor Immunity: Potential Molecular Mechanism. CURR MED SCI 39, 685–689 (2019). https://doi.org/10.1007/s11596-019-2092-5
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-019-2092-5