Abstract
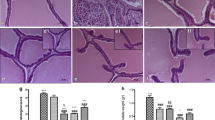
This study investigated the effect of diosgenin, a natural sapogenin possessing various pharmacological activities, on benign prostatic hyperplasia (BPH) in rats and the possible mechanisms. BPH was established in the castrated rats by subcutaneous injection of testosterone propionate. Animals were randomly divided into four groups (n=10 each): model group (0.5% sodium carboxymethyl cellulose); positive control group (3 mg/kg finasteride); two diosgenin groups (50 and 100 mg/kg). The drugs were intragastricaly given in each group for consecutive 3 weeks. Another 10 rats with no testicles cut off served as negative controls and they were subcutaneously injected with 0.1 mL olive oil per day and then treated with 0.5% sodium carboxymethylcellulose. After 3-week administration, the prostate index and serum PSA level were determined, and histopathological examination was carried out. The levels of MDA, SOD and GPx in prostates were also measured. Additionally, the expression of Bcl-2, Bax and p53 was examined using Western blotting. The results showed that the prostate index and serum PSA level were significantly decreased, and the pathological changes of the prostate gland were greatly improved in diosgenin groups as compared with the model group. Elevated activities of SOD and GPx, and reduced MDA level were also observed in diosgenin-treated rats. In addition, the expression of Bcl-2 in prostates was down-regulated, whereas that of Bax and p53 was up-regulated in diosgenin-treated rats. These results indicated that diosgenin was effective in inhibiting testosterone propionate-induced prostate enlargement and may be a candidate agent for the treatment of BPH.
Similar content being viewed by others
References
Berry SJ, Coffey DS, Walsh PC, et al. The development of human benign prostatic hyperplasia with age. J Urol, 1984,132(3):474–479
Djavan B. Lower urinary tract symptoms/benign prostatic hyperplasia: fast control of the patient’s quality of life. Urology, 2003,62(3 Suppl 1):6–14
Gandaglia G, Briganti A, Gontero P, et al. The role of chronic prostatic inflammation in the pathogenesis and progression of benign prostatic hyperplasia (BPH). BJU Int, 2013,112(4):432–441
Kyprianou N, Tu H, Jacobs SC. Apoptotic versus proliferative activities in human benign prostatic hyperplasia. Hum Pathol, 1996,27(7):668–675
Pace G, Di Massimo C, de Amicis D, et al. Oxidative stress in benign prostatic hyperplasia and prostate cancer. Urol Int, 2010,85(3):328–333
Kaplan SA, Lee JY, Meehan AG, et al. Long-term treatment with finasteride improves clinical progression of benign prostatic hyperplasia in men with an enlarged versus a smaller prostate: data from the MTOPS trial. J Urol, 2011,185(4):1369–1373
Oelke M, Höfner K, Berges RR, et al. Drug therapy of benign prostatic hyperplasia syndrome with alpha 1-receptor blockers. Basic principles and clinical results. Urologe A, 2002,41(5):425–441
Bullock TL, Andriole GL Jr. Emerging drug therapies for benign prostatic hyperplasia. Expert Opin Emerg Drugs, 2006,11(1):111–123
Carlin BI, Seftel AD, Resnick MI, et al. Finasteride induced gynecomastia. J Urol, 1997,158(2):547
Chung KS, Cheon SY, An HJ. Effects of resveratrol on benign prostatic hyperplasia by the regulation of inflamematory and apoptotic proteins. J Nat Prod, 2015,78(4):689–694
Sirab N, Robert G, Fasolo V, et al. Lipidosterolic extract of Serenoa repens modulates the expression of inflammation related-genes in benign prostatic hyperplasia epithelial and stromal cells. Int J Mol Sci, 2013,14(7):14301–14320
Xu DH, Wang LH, Mei XT, et al. Protective effects of seahorse extracts in a rat castration and testosterone-induced benign prostatic hyperplasia model and mouse oligospermatism model. Environ Toxicol Pharmacol, 2014,37(2):679–688
Shao B, Guo H, Cui Y, et al. Steroidal saponins from Smilax china and their anti-inflammatory activities. Phytochemistry, 2007,68(5):623–630
Ahmed LA, Obaid AA, Zaki HF, et al. Role of oxidative stress, inflammation, nitric oxide and transforming growth factor-beta in the protective effect of diosgenin in monocrotalin-induced pulmonary hypertension in rats. Eur J Pharmacol, 2014,740:379–387
Kim DS, Jeon BK, Lee YE, et al. Diosgenin induces apoptosis in HepG2 cells through generation of reactive oxygen species and mitochondrial pathway. Evid Based Complement Alternat Med, 2012,2012:981675
Chen PS, Shih YW, Huang HC, et al. Diosgenin, a steroidal saponin, inhibits migration and invasion of human prostate cancer PC-3 cells by reducing matrix metalloproteinases expression. PLoS One, 2011,6(5): e20164
Chen J, Xiong CM, Song SS, et al. Fraction of macroporous resin from Smilax china L. inhibits testosterone propionate-induced prostatic hyperplasia in castrated rats. J Med Food, 2012,15(7):646–650
van Coppenolle F, Le Bourhis X, Carpentier F, et al. Pharmacological effects of the lipidosterolic extract of Serenoa repens (Permixon®) on rat prostate hyperplasia induced by hyperprolactinemia: comparison with finasteride. Prostate, 2000,43(1):49–58
Ali MI, Kondreddi HD, Veeresh B. Protective effect of 2-hydroxy-4-methoxy benzoic acid on testosterone induced benign prostatic hyperplasia in Wister rats. Eur J Pharmacol, 2013,698(1-3):397–403
Atawia RT, Tadros MG, Khalifa AE, et al. Role of the phytoestrogenic, pro-apoptotic and anti-oxidative properties of silymarin in inhibiting experimental benign prostatic hyperplasia in rats. Toxicol Lett, 2013,219(2):160–169
Ahmad M, Suhail N, Mansoor T, et al. Evaluation of oxidative stress and DNA damage in benign prostatic hyperplasia patients and comparison with controls. Indian J Clin Biochem, 2012,27(4):385–388
Fleshner NE, Klotz LH. Diet, androgens, oxidative stress and prostate cancer susceptibility. Cancer Metastasis Rev, 1998-1999,17(4):325–330
Aruoma OI. Free radicals, oxidative stress, and antioxidants in human health and disease. J Am Oil Chem Soc, 1998,75(2):199–212
Jang H, Bae WJ, Yuk SM, et al. Seoritae extract reduces prostate weight and suppresses prostate cell proliferation in a rat model of benign prostate hyperplasia. Evid Based Complement Alternat Med, 2014,2014:475876
Minutoli L, Altavilla D, Marini H, et al. Inhibitors of apoptosis proteins in experimental benign prostatic hyperplasia: effects of serenoa repens, selenium and lycopene. J Biomed Sci, 2014,21:19
Adams JM, Cory S. The Bcl-2 protein family: arbiters of cell survival. Science, 1998,281(5381):1322–1326
Amaral JD, Xavier JM, Steer CJ, et al. The role of p53 in apoptosis. Discov Med, 2010,9(45):145–152
Roshan S, Liu YY, Banafa A, et al. Fucoidan induces apoptosis of HepG2 cells by down-regulating p-Stat3. J Huazhong Univ Sci Technol [Med Sci], 2014,34(3):330–336
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by the National Nature Science Foundation of China (No. 81173065).
Rights and permissions
About this article
Cite this article
Chen, J., Zhang, Hf., Xiong, Cm. et al. Inhibitory effect of diosgenin on experimentally induced benign prostatic hyperplasia in rats. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 36, 806–810 (2016). https://doi.org/10.1007/s11596-016-1666-8
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-016-1666-8