Summary
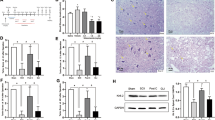
The effects of tacrolimus postconditioning on protein-serine-threonine kinases (Akt) phosphorylation and apoptotic cell death in rats after spinal cord ischemia-reperfusion injury were investigated. Ninety male SD rats were randomly divided into sham operation group, ischemia-reperfusion group and tacrolimus postconditioning group. The model of spinal cord ischemia was established by means of catheterization through femoral artery and balloon dilatation. The spinal cord was reperfused 20 min after ischemia via removing saline out of balloon. The corresponding spinal cord segments were excised and determined for Akt activity in spinal cord tissue by using Western blotting at 5, 15, and 60 min after reperfusion respectively. Spinal cord tissue sections were stained immunohistochemically for detection of the phosphorylated Akt expression at 15 min after reperfusion. Flow cytometry was applied to assess apoptosis of neural cells, and dry-wet weights method was employed to measure water content in spinal cord tissue at 24 h after reperfusion. The results showed that the activities of Akt in tarcolimus postconditioning group were significantly higher than those in ischemia-reperfusion group at 5, 15, and 60 min after reperfusion (P<0.05, P<0.01). The Akt activities reached the peak at 15 min after reperfusion in ischemia-reperfusion group and tacrolimus postconditioning group. The percentage of apoptotic cells and water content in spinal cord tissue were significantly reduced (P<0.01) in tacrolimus postconditioning group as compared with those in ischemia-reperfusion group at 24 h after reperfusion. It is concluded that tacrolimus post-conditioning can increase Akt activity in spinal cord tissue of rats, inhibit apoptosis of neural cells as well as tissue edema, and thereby alleviate spinal cord ischemia-reperfusion injury.
Similar content being viewed by others
References
Svensson LG, Crawford ES, Hess KR, et al. Experience with 1509 patients undergoing thoracoabdominal aortic operations. J Vasc Surg, 1993,17(2):357–368
Pan F, Zhu C, Cheng Y, et al. The neuroprotective effect of tacrolimus postconditioning combined with rehabilitation training in spinal cord ischemia-reperfusion injury in rats. Chin J Phys Med Rehabil (Chinese), 2013,35(3): 172–176
Taira Y, Marsala M. Effect of proximal arterial perfusion pressure on function, spinal cord blood flow, and histo pathologic changes after increasing intervals of aortic occlusion in the rat. Stroke, 1996,27(10):1850–1858
Pan F, Cheng Y, Tao H, et al. Comparison of neuroprotective effect of two postconditioning methods on spinal cord ischemia-reperfusion injury in rats. Chin J Exp Surg (Chinese), 2013,30(7):1413–1415
Mallei A, Aden SA, Bachis A, et al. The nitrosteroid NCX 1015, a prednisolone derivative, improves recovery of function in rats after spinal cord injury. Brain Res, 2005,1062(1–2):16–25
Bigini P, Atzori C, Fumagalli E, et al. Lack of caspase-dependent apoptosis in spinal motor neurons of the wobbler mouse. Neurosci Lett, 2007,426(2):106–110
Pan F, Chen A, Guo F, et al. Effect of FK506 on expression of hepatocyte growth factor in murine spinal cord following peripheral nerve injury. J Huazhong Univ Sci Technol [Med Sci], 2008,28(2):159–162
Li J, Lee B, Lee AS. Endoplasmic reticulum stress-induced apoptosis: multiple pathways and activation of p53-up-regulated modulator of apoptosis (PUMA) and NOXA by p53. J Biol Chem, 2006,281(11): 7260–7270
Hausenloy DJ, Yellon DM. Preconditioning and postconditioning: underlying mechanisms and clinical application. Atherosclerosis, 2009,204(2):334–341
Boengler K, Heusch G, Schulz R. Mitochondria in postconditioning. Antioxid Redox Signal, 2011,14(5): 863–880
Alba-Betancourt C, Luna-Acosta JL, Ramírez-Martínez CE, et al. Neuro-protective effects of growth hormone (GH) after hypoxia-ischemia injury in embryonic chicken cerebellum. Gen Comp Endocrinol, 2013,183:17–31
Li L, Zuo Z. Isoflurane postconditioning induces neuroprotection via Akt activation and attenuation of increased mitochondrial membrane permeability. Neuroscience, 2011,199:44–50
Mockford KA, Girn HR, Homer-Vanniasinkam S. Postconditioning: current controversies and clinical implications. Eur J Vasc Endovasc Surg, 2009,37(4):437–442
Penna C, Mancardi D, Rastaldo R, et al. Cardioprotection: a radical view free radicals in pre and post conditioning. Biochim Biophys Acta, 2009,1787(7):781–793
Ovize M, Baxter GF, Di Lisa F, et al. Postconditioning and protection from reperfusion injury: where do we stand? Position paper from the Working Group of Cellular Biology of the Heart of the European Society of Cardiology. Cardiovasc Res, 2010,87(3):406–423
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by the Hubei Provincial Natural Science Foundation of China (No. 2012FFB04406).
Rights and permissions
About this article
Cite this article
Pan, F., Cheng, Yx., Zhu, Cl. et al. Tacolimus postconditioning alleviates apoptotic cell death in rats after spinal cord ischemia-reperfusion injury via up-regulating protein-serine-threonine kinases phosphorylation. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 33, 852–856 (2013). https://doi.org/10.1007/s11596-013-1210-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-013-1210-z