Abstract
CD8+ T cells can contribute to neuroinflammation by secretion of inflammatory cytokines like interferon γ (IFNγ) and tumor necrosis factor α (TNFα). Astrocytes, a glial cell in the brain, can be stimulated by IFNγ and TNFα to secrete the inflammatory cytokines, monocyte chemotactic protein 1 (MCP-1), interleukin 6 (IL-6), and interferon-γ inducible protein 10 (IP-10). Δ9-Tetrahydrocannabinol (THC), the primary psychoactive cannabinoid in Cannabis sativa, possesses potent anti-inflammatory activity. The objective of this investigation was to assess the effects of THC treatment on CD8+ T cell-mediated activation of astrocytes. CD3/CD28/IFNα- stimulated CD8+ T cells were treated with vehicle (0.03% EtOH) or THC and cocultured with U251 astrocytes. IP-10+, MCP-1+, and IL-6+ astrocytes were quantified by flow cytometry. LegendPlex™ was used to measure cytokine secretion by CD8+ T cells and flow cytometry was employed to quantify IFNγ, TNFα, and lysosomal-associated membrane protein 1 (LAMP-1) expression. Recombinant TNFα and IFNγ were used to stimulate MCP-1, IP-10, IL-6 responses in U251 astrocytes, which were measured by flow cytometry. Treatment with THC reduced CD8+ T cell-mediated induction of IP-10 and IL-6 responses in U251 astrocytes but had no effect on MCP-1. THC treatment differentially affected T cell effector functions such that IFNγ and degranulation responses were sensitive to THC-mediated ablation while TNFα was not. Lastly, THC treatment reduced the IFNγ-induced IP-10 response but had no effect on TNFα-induced MCP-1 response in U251 astrocytes. The results suggest that cannabinoid treatment can selectively reduce certain CD8+ T cell responses that contribute to stimulation of astrocytes.
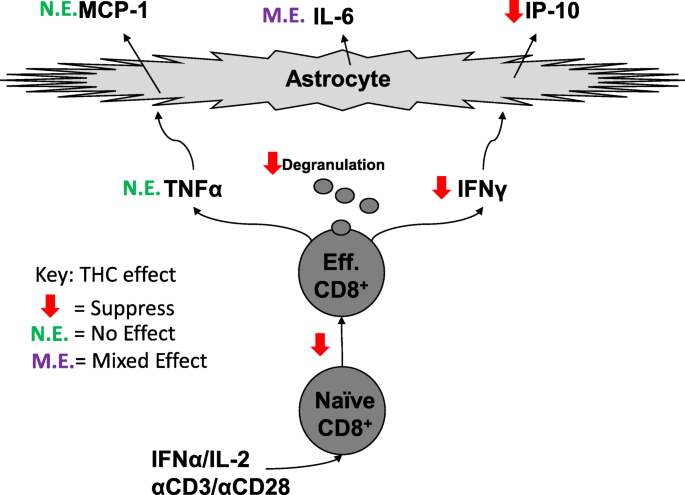
Treatment with THC can abate CD8+ T cell-dependent neuroinflammatory processes by inhibiting CD8+ cell differentiation into effector cells, suppressing CD8+ effector cell function, and reducing activation of astrocytes by CD8+ T cell-derived inflammatory cytokines.





Similar content being viewed by others
References
Alcocer-Varela J, Aleman-Hoey D, Alarcon-Segovia D (1992) Interleukin-1 and interleukin-6 activities are increased in the cerebrospinal fluid of patients with CNS lupus erythematosus and correlate with local late T-cell activation markers. Lupus 1:111–117
Appay V, Nixon DF, Donahoe SM, Gillespie GM, Dong T, King A, Ogg GS, Spiegel HM, Conlon C, Spina CA (2000) HIV-specific CD8+ T cells produce antiviral cytokines but are impaired in cytolytic function. J Exp Med 192:63–76
Balashov KE, Rottman JB, Weiner HL, Hancock WW (1999) CCR5+ and CXCR3+ T cells are increased in multiple sclerosis and their ligands MIP-1α and IP-10 are expressed in demyelinating brain lesions. Proc Natl Acad Sci 96:6873–6878
Betts MR, Brenchley JM, Price DA, De Rosa SC, Douek DC, Roederer M, Koup RA (2003) Sensitive and viable identification of antigen-specific CD8+ T cells by a flow cytometric assay for degranulation. J Immunol Methods 281:65–78
Bezzi P, Domercq M, Brambilla L, Galli R, Schols D, De Clercq E, Vescovi A, Bagetta G, Kollias G, Meldolesi J (2001) CXCR4-activated astrocyte glutamate release via TNFα: amplification by microglia triggers neurotoxicity. Nat Neurosci 4:702
Blake D, Robson P, Ho M, Jubb R, McCabe C (2006) Preliminary assessment of the efficacy, tolerability and safety of a cannabis-based medicine (Sativex) in the treatment of pain caused by rheumatoid arthritis. Rheumatology 45:50–52
Blanco P, Palucka AK, Gill M, Pascual V, Banchereau J (2001) Induction of dendritic cell differentiation by IFN-α in systemic lupus erythematosus. Science 294:1540–1543
Bouaboula M, Bourrié B, Rinaldi-Carmona M, Shire D, Le Fur G, Casellas P (1995) Stimulation of cannabinoid receptor CB1 induces krox-24 expression in human astrocytoma cells. J Biol Chem 270:13973–13980
Chen W, Kaplan BL, Pike ST, Topper LA, Lichorobiec NR, Simmons SO, Ramabhadran R, Kaminski NE (2012) Magnitude of stimulation dictates the cannabinoid-mediated differential T cell response to HIVgp120. J Leukoc Biol 92:1093–1102
Comabella M, Lünemann J, Rio J, Sanchez A, Lopez C, Julia E, Fernandez M, Nonell L, Camina-Tato M, Deisenhammer F (2009) A type I interferon signature in monocytes is associated with poor response to interferon-β in multiple sclerosis. Brain 132:3353–3365
Conant K, Garzino-Demo A, Nath A, McArthur JC, Halliday W, Power C, Gallo RC, Major EO (1998) Induction of monocyte chemoattractant protein-1 in HIV-1 tat-stimulated astrocytes and elevation in AIDS dementia. Proc Natl Acad Sci 95:3117–3121
Croitoru-Lamoury J, Guillemin GJ, Boussin FD, Mognetti B, Gigout LI, Chéret A, Vaslin B, Le Grand R, Brew BJ, Dormont D (2003) Expression of chemokines and their receptors in human and simian astrocytes: evidence for a central role of TNFα and IFNγ in CXCR4 and CCR5 modulation. Glia 41:354–370
Croxford JL, Pryce G, Jackson SJ, Ledent C, Giovannoni G, Pertwee RG, Yamamura T, Baker D (2008) Cannabinoid-mediated neuroprotection, not immunosuppression, may be more relevant to multiple sclerosis. J Neuroimmunol 193:120–129
Curran NM, Griffin BD, O'Toole D, Brady KJ, Fitzgerald SN, Moynagh PN (2005) The synthetic cannabinoid R (+) WIN 55,212-2 inhibits the interleukin-1 signaling pathway in human astrocytes in a cannabinoid receptor-independent manner. J Biol Chem 280:35797–35806
Deshmane SL, Kremlev S, Amini S, Sawaya BE (2009) Monocyte chemoattractant protein-1 (MCP-1): an overview. J Interf Cytokine Res 29:313–326
Ehrhart J, Obregon D, Mori T, Hou H, Sun N, Bai Y, Klein T, Fernandez F, Tan J, Shytle RD (2005) Stimulation of cannabinoid receptor 2 (CB 2) suppresses microglial activation. J Neuroinflammation 2:29
Eisenstein TK, Meissler JJ (2015) Effects of cannabinoids on T-cell function and resistance to infection. J NeuroImmune Pharmacol 10:204–216
Farkas L, Beiske K, Lund-Johansen F, Brandtzaeg P, Jahnsen FL (2001) Plasmacytoid dendritic cells (natural interferon-α/β-producing cells) accumulate in cutaneous lupus erythematosus lesions. Am J Pathol 159:237–243
Fine SM, Angel RA, Perry SW, Epstein LG, Rothstein JD, Dewhurst S, Gelbard HA (1996) Tumor necrosis factor α inhibits glutamate uptake by primary human astrocytes IMPLICATIONS FOR PATHOGENESIS OF HIV-1 DEMENTIA. J Biol Chem 271:15303–15306
Giorgi JV, Liu Z, Hultin LE, Cumberland WG, Hennessey K, Detels R (1993) Elevated levels of CD38+ CD8+ T cells in HIV infection add to the prognostic value of low CD4+ T cell levels: results of 6 years of follow-up. The Los Angeles center, multicenter AIDS cohort study. J Acquir Immune Defic Syndr 6:904–912
Giorgi JV, Hultin LE, McKeating JA, Johnson TD, Owens B, Jacobson LP, Shih R, Lewis J, Wiley DJ, Phair JP (1999) Shorter survival in advanced human immunodeficiency virus type 1 infection is more closely associated with T lymphocyte activation than with plasma virus burden or virus chemokine coreceptor usage. J Infect Dis 179:859–870
González H, Pacheco R (2014) T-cell-mediated regulation of neuroinflammation involved in neurodegenerative diseases. J Neuroinflammation 11:201
Gulzar N, Copeland KF (2004) CD8+ T-cells: function and response to HIV infection. Curr HIV Res 2:23–37
Henriquez J, Rizzo M, Crawford R, Gulick P, Kaminski N (2018) Interferon-α-mediated activation of T cells from healthy and HIV-infected individuals is suppressed by Δ 9-Tetrahydrocannabinol i. J Pharmacol Exp Ther
Henriquez JE, Rizzo MD, Schulz MA, Crawford RB, Gulick P, Kaminski NE (2017) Δ9-Tetrahydrocannabinol suppresses secretion of IFNα by Plasmacytoid dendritic cells from healthy and HIV-infected individuals. JAIDS Journal of Acquired Immune Deficiency Syndromes 75:588–596
Hersperger AR, Makedonas G, Betts MR (2008) Flow cytometric detection of perforin upregulation in human CD8 T cells. Cytometry Part A 73:1050–1057
Huber JP, David Farrar J (2011) Regulation of effector and memory T-cell functions by type I interferon. Immunology 132:466–474
Jackson SJ, Baker D, Cuzner ML, Diemel LT (2004) Cannabinoid-mediated neuroprotection following interferon-gamma treatment in a three-dimensional mouse brain aggregate cell culture. Eur J Neurosci 20:2267–2275
Janzer RC, Raff MC (1987) Astrocytes induce blood–brain barrier properties in endothelial cells. Nature 325:253
Jennings RN, Grayson JM, Barton ES (2014) Type I interferon signaling enhances CD8+ T cell effector function and differentiation during MHV68 infection. J Virol JVI 02360–02314
Kambayashi T, Assarsson E, Lukacher AE, Ljunggren H-G, Jensen PE (2003) Memory CD8+ T cells provide an early source of IFN-γ. J Immunol 170:2399–2408
Karmaus PW, Chen W, Kaplan BL, Kaminski NE (2012) Δ 9-Tetrahydrocannabinol suppresses cytotoxic T lymphocyte function independent of CB 1 and CB 2, disrupting early activation events. J NeuroImmune Pharmacol 7:843–855
Kaul M, Garden GA, Lipton SA (2001) Pathways to neuronal injury and apoptosis in HIV-associated dementia. Nature 410:988
Kessing CF, Spudich S, Valcour V, Cartwright P, Chalermchai T, Fletcher JL, Takata H, Nichols C, Josey BJ, Slike B (2017) High number of activated CD8+ T cells targeting HIV antigens are present in cerebrospinal fluid in acute HIV infection. JAIDS Journal of Acquired Immune Deficiency Syndromes 75:108–117
Klein TW (2005) Cannabinoid-based drugs as anti-inflammatory therapeutics. Nat Rev Immunol 5:400–411
Kolumam GA, Thomas S, Thompson LJ, Sprent J, Murali-Krishna K (2005) Type I interferons act directly on CD8 T cells to allow clonal expansion and memory formation in response to viral infection. J Exp Med 202:637–650
Lapteva L, Nowak M, Yarboro CH, Takada K, Roebuck-Spencer T, Weickert T, Bleiberg J, Rosenstein D, Pao M, Patronas N (2006) Anti–N-methyl-D-aspartate receptor antibodies, cognitive dysfunction, and depression in systemic lupus erythematosus. Arthritis & Rheumatism: Official Journal of the American College of Rheumatology 54:2505–2514
Lo Y-C, Edidin MA, Powell JD (2013) Selective activation of antigen-experienced T cells by anti-CD3 constrained on nanoparticles. J Immunol 191:5107–5114
Ma X, Reynolds SL, Baker BJ, Li X, Benveniste EN, Qin H (2010) IL-17 enhancement of the IL-6 signaling cascade in astrocytes. J Immunol ji_1000142
Meade T, Manolios N, Cumming SR, Conaghan PG, Katz P (2018) Cognitive impairment in rheumatoid arthritis: a systematic review. Arthritis care & research 70:39–52
Nagarkatti P, Pandey R, Rieder SA, Hegde VL, Nagarkatti M (2009) Cannabinoids as novel anti-inflammatory drugs. Future Med Chem 1:1333–1349
Newby BN, Brusko TM, Zou B, Atkinson MA, Clare-Salzler M, Mathews CE (2017) Type 1 interferons potentiate human CD8+ T cell cytotoxicity through a STAT4 and granzyme B dependent pathway. Diabetes:db170106
Nguyen KB, Watford WT, Salomon R, Hofmann SR, Pien GC, Morinobu A, Gadina M, O'shea JJ, Biron CA (2002) Critical role for STAT4 activation by type 1 interferons in the interferon-γ response to viral infection. Science 297:2063–2066
Palmer AM, Marion DW, Botscheller ML, Swedlow PE, Styren SD, DeKosky ST (1993) Traumatic brain injury-induced excitotoxicity assessed in a controlled cortical impact model. J Neurochem 61:2015–2024
Pitt D, Werner P, Raine CS (2000) Glutamate excitotoxicity in a model of multiple sclerosis. Nat Med 6:67
Ransohoff RM, Hamilton TA, Tani M, Stoler MH, Shick HE, Major JA, Estes ML, Thomas DM, Tuohy VK (1993) Astrocyte expression of mRNA encoding cytokines IP-10 and JE/MCP-1 in experimental autoimmune encephalomyelitis. FASEB J 7:592–600
Reiss CS (2010) Cannabinoids and viral infections. Pharmaceuticals 3:1873–1886
Rodríguez-Carrio J, de Paz B, López P, Prado C, Alperi-López M, Ballina-García FJ, Suárez A (2014) IFNα serum levels are associated with endothelial progenitor cells imbalance and disease features in rheumatoid arthritis patients. PLoS One 9:e86069
Sheng WS, Hu S, Min X, Cabral GA, Lokensgard JR, Peterson PK (2005) Synthetic cannabinoid WIN55, 212-2 inhibits generation of inflammatory mediators by IL-1β-stimulated human astrocytes. Glia 49:211–219
Skulina C, Schmidt S, Dornmair K, Babbe H, Roers A, Rajewsky K, Wekerle H, Hohlfeld R, Goebels N (2004) Multiple sclerosis: brain-infiltrating CD8+ T cells persist as clonal expansions in the cerebrospinal fluid and blood. Proc Natl Acad Sci 101:2428–2433
Tanasescu R, Constantinescu CS (2010) Cannabinoids and the immune system: an overview. Immunobiology 215:588–597
Trajkovic V, Vuckovic O, Stosic-Grujicic S, Miljkovic D, Popadic D, Markovic M, Bumbasirevic V, Backovic A, Cvetkovic I, Harhaji L (2004) Astrocyte-induced regulatory T cells mitigate CNS autoimmunity. Glia 47:168–179
Walter L, Stella N (2004) Cannabinoids and neuroinflammation. Br J Pharmacol 141:775–785
Williams R, Yao H, Dhillon NK, Buch SJ (2009) HIV-1 tat co-operates with IFN-γ and TNF-α to increase CXCL10 in human astrocytes. PLoS One 4:e5709
Yi J-H, Hazell AS (2006) Excitotoxic mechanisms and the role of astrocytic glutamate transporters in traumatic brain injury. Neurochem Int 48:394–403
Yuzefpolskiy Y, Baumann FM, Kalia V, Sarkar S (2015) Early CD8 T-cell memory precursors and terminal effectors exhibit equipotent in vivo degranulation. Cellular & molecular immunology 12:400
Zhang S, Xie R, Wan F, Ye F, Guo D, Lei T (2013) Identification of U251 glioma stem cells and their heterogeneous stem-like phenotypes. Oncol Lett 6:1649–1655
Acknowledgements
We would like thank Dr. Bill Atchison and the University of Puerto Rico, Ponce for enabling Karina Matos-Fernandez to participate in and contribute to this research. The authors would also like to thank Jiajun Zhou, for formatting figures in preparation for article submission and Mrs. Kimberly Hambleton, for assistance in the submission of this article.
Funding
This work was supported by the National Institute on Drug Abuse Grants DA047180 and National Institute of Environmental Health Sciences Training Grants T32-ES007255 and R25-ES025060.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Declaration of Conflicting Interests
The Authors declare no conflicting interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Henriquez, J.E., Bach, A.P., Matos-Fernandez, K.M. et al. Δ9-Tetrahydrocannabinol (THC) Impairs CD8+ T Cell-Mediated Activation of Astrocytes. J Neuroimmune Pharmacol 15, 863–874 (2020). https://doi.org/10.1007/s11481-020-09912-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-020-09912-z




