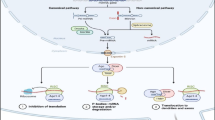
Abstract
Hepatocellular carcinoma (HCC) is common and one of the most aggressive of all human cancers. Recent studies have indicated that miRNAs, a class of small noncoding RNAs that regulate gene expression post-transcriptionally, directly contribute to HCC by targeting many critical regulatory genes. Several miRNAs are involved in hepatitis B or hepatitis C virus replication and virus-induced changes, whereas others participate in multiple intracellular signaling pathways that modulate apoptosis, cell cycle checkpoints, and growth-factor-stimulated responses. When disturbed, these pathways appear to result in malignant transformation and ultimately HCC development. Recently, miRNAs circulating in the blood have acted as possible early diagnostic markers for HCC. These miRNA also could serve as indicators with respect to drug efficacy and be prognostic in HCC patients. Such biomarkers would assist stratification of HCC patients and help direct personalized therapy. Here, we summarize recent advances regarding the role of miRNAs in HCC development and progression. Our expectation is that these and ongoing studies will contribute to the understanding of the multiple roles of these small noncoding RNAs in liver tumorigenesis.
Article PDF
Similar content being viewed by others
References
Berry D A, Herbst R S, Rubin E H. Reports from the 2010 Clinical and Translational Cancer Research Think Tank meeting: design strategies for personalized therapy trials. Clin Cancer Res, 2012, 18: 638–644
Liu L, Miao R, Yang H, et al. Prognostic factors after liver resection for hepatocellular carcinoma: a single-center experience from China. Am J Surg, 2012, 203: 741–750
El-Serag H B, Marrero J A, Rudolph L, et al. Diagnosis and treatment of hepatocellular carcinoma. Gastroenterology, 2008,134: 1752–1763
Huynh H. Tyrosine kinase inhibitors to treat liver cancer. Expert Opin Emerg Drugs, 2010, 15: 13–26
Razumilava N, Gores G J. Sorafenib for HCC: a pragmatic perspective. Oncology (Williston Park), 2011, 25: 300, 302
Blum H E. Hepatocellular carcinoma: therapy and prevention. World J Gastroenterol, 2005, 11: 7391–7400
Cho Y K. A randomized trial comparing radiofrequency ablation and surgical resection for HCC conforming to the Milan criteria. Ann Surg, 2011, 254: 838–839
Chen J W, Kow L, Verran D J, et al. Poorer survival in patients whose explanted hepatocellular carcinoma (HCC) exceeds Milan or UCSF Criteria. An analysis of liver transplantation in HCC in Australia and New Zealand. HPB (Oxford), 2009, 11: 81–89
Calin G A, Croce C M. MicroRNA signatures in human cancers. Nat Rev Cancer, 2006, 6: 857–866
Lu X, Zhao H, Yang H, et al. A prospective clinical study on early recurrence of hepatocellular carcinoma after hepatectomy. J Surg Oncol, 2009, 100: 488–493
Clinkenbeard E L, Butler J E, Spear B T. Pericentral activity of AFP enhancer E3 and glutamine synthetase upstream enhancer in the adult liver are regulated by beta-catenin. Hepatology, 2012
Petri A, Lindow M, Kauppinen S. MicroRNA silencing in primates: towards development of novel therapeutics. Cancer Res, 2009, 69: 393–395
Mott J L. MicroRNAs involved in tumor suppressor and oncogene pathways: implications for hepatobiliary neoplasia. Hepatology, 2009, 50: 630–637
Feng Y, Yu X. Cardinal roles of miRNA in cardiac development and disease. Sci China Life Sci, 2011, 54: 1113–1120
Zhang Y, Dong D, Yang B. Atrial remodeling in atrial fibrillation and association between microRNA network and atrial fibrillation. Sci China Life Sci, 2011, 54: 1097–1102
Feng W, Feng Y. MicroRNAs in neural cell development and brain diseases. Sci China Life Sci, 2011, 54: 1103–1112
Luo J, Teng M, Fan J, et al. Marek’s disease virus-encoded microRNAs: genomics, expression and function. Sci China Life Sci, 2010, 53: 1174–1180
Zuo J, Wang Y, Liu H, et al. MicroRNAs in tomato plants. Sci China Life Sci, 2011, 54: 599–605
He S, Yang Z, Skogerbo G, et al. The properties and functions of virus encoded microRNA, siRNA, and other small noncoding RNAs. Crit Rev Microbiol, 2008, 34: 175–188
Visone R, Petrocca F, Croce C M. Micro-RNAs in gastrointestinal and liver disease. Gastroenterology, 2008, 135: 1866–1869
Montalto G, Cervello M, Giannitrapani L, et al. Epidemiology, risk factors, and natural history of hepatocellular carcinoma. Ann N Y Acad Sci, 2002, 963: 13–20
Yang C, Wei W. The miRNA expression profile of the uveal melanoma. Sci China Life Sci, 2011, 54: 351–358
Wu J, Wang C, Du Z, et al. Identification of Pns12 as the second silencing suppressor of Rice gall dwarf virus. Sci China Life Sci, 2011, 54: 201–208
Wang X, Zhao H, Xu Q, et al. HPtaa database-potential target genes for clinical diagnosis and immunotherapy of human carcinoma. Nucleic Acids Res, 2006, 34: D607–612
Zhang X, Zhang H, Ye L. Effects of hepatitis B virus X protein on the development of liver cancer. J Lab Clin Med, 2006, 147: 58–66
Wang Y, Cui F, Lv Y, et al. HBsAg and HBx knocked into the p21 locus causes hepatocellular carcinoma in mice. Hepatology, 2004, 39: 318–324
Lara-Pezzi E, Majano P L, Yanez-Mo M, et al. Effect of the hepatitis B virus HBx protein on integrin-mediated adhesion to and migration on extracellular matrix. J Hepatol, 2001, 34: 409–415
Xia L M, Huang W J, Wu J G, et al. HBx protein induces expression of MIG and increases migration of leukocytes through activation of NF-kappaB. Virology, 2009, 385: 335–342
Chung T W, Lee Y C, Kim C H. Hepatitis B viral HBx induces matrix metalloproteinase-9 gene expression through activation of ERK and PI-3K/AKT pathways: involvement of invasive potential. FASEB J, 2004, 18: 1123–1125
Zhang F, Wang Q, Ye L, et al. Hepatitis B virus X protein upregulates expression of calpain small subunit 1 via nuclear factor-kappaB/p65 in hepatoma cells. J Med Virol, 2010, 82: 920–928
Zhang X, Liu S, Hu T, et al. Up-regulated microRNA-143 transcribed by nuclear factor kappa B enhances hepatocarcinoma metastasis by repressing fibronectin expression. Hepatology, 2009, 50: 490–499
Yin Y, Zhao Y, Wang J, et al. antiCODE: a natural sense-antisense transcripts database. BMC Bioinformatics, 2007, 8: 319
Gao W, Yu Y, Cao H, et al. Deregulated expression of miR-21, miR-143 and miR-181a in non small cell lung cancer is related to clinicopathologic characteristics or patient prognosis. Biomed Pharmacother, 2010, 64: 399–408
Kitade Y, Akao Y. MicroRNAs and their therapeutic potential for human diseases: microRNAs, miR-143 and -145, function as anti-oncomirs and the application of chemically modified miR-143 as an anti-cancer drug. J Pharmacol Sci, 2010, 114: 276–280
Dasari V R, Kaur K, Velpula K K, et al. Upregulation of PTEN in glioma cells by cord blood mesenchymal stem cells inhibits migration via downregulation of the PI3K/Akt pathway. PLoS ONE, 2010, 5: e10350
Du B, Ma L M, Huang M B, et al. High glucose down-regulates miR-29a to increase collagen IV production in HK-2 cells. FEBS Lett, 2010, 584: 811–816
Narbus C M, Israelow B, Sourisseau M, et al. HepG2 cells expressing microRNA miR-122 support the entire hepatitis C virus life cycle. J Virol, 2011, 85: 12087–12092
Cui Y, Su W Y, Xing J, et al. MiR-29a inhibits cell proliferation and induces cell cycle arrest through the downregulation of p42.3 in human gastric cancer. PLoS ONE, 2011, 6: e25872
Dudek H, Datta S R, Franke T F, et al. Regulation of neuronal survival by the serine-threonine protein kinase Akt. Science, 1997, 275: 661–665
Meng F, Henson R, Wehbe-Janek H, et al. MicroRNA-21 regulates expression of the PTEN tumor suppressor gene in human hepatocellular cancer. Gastroenterology, 2007, 133: 647–658
Garofalo M, Di Leva G, Romano G, et al. miR-221&222 regulate TRAIL resistance and enhance tumorigenicity through PTEN and TIMP3 downregulation. Cancer Cell, 2009, 16: 498–509
Kong G, Zhang J, Zhang S, et al. Upregulated microRNA-29a by hepatitis B virus X protein enhances hepatoma cell migration by targeting PTEN in cell culture model. PLoS ONE, 2011, 6: e19518
Korenaga M, Wang T, Li Y, et al. Hepatitis C virus core protein inhibits mitochondrial electron transport and increases reactive oxygen species (ROS) production. J Biol Chem, 2005, 280: 37481–37488
Irshad M, Dhar I. Hepatitis C virus core protein: an update on its molecular biology, cellular functions and clinical implications. Med Princ Pract, 2006, 15: 405–416
Pedersen I M, Cheng G, Wieland S, et al. Interferon modulation of cellular microRNAs as an antiviral mechanism. Nature, 2007, 449: 919–922
Hou W, Tian Q, Zheng J, et al. MicroRNA-196 represses Bach1 protein and hepatitis C virus gene expression in human hepatoma cells expressing hepatitis C viral proteins. Hepatology, 2010, 51: 1494–1504
Shan Y, Lambrecht R W, Ghaziani T, et al. Role of Bach-1 in regulation of heme oxygenase-1 in human liver cells: insights from studies with small interfering RNAS. J Biol Chem, 2004, 279: 51769–51774
Hornstein E, Mansfield J H, Yekta S, et al. The microRNA miR-196 acts upstream of Hoxb8 and Shh in limb development. Nature, 2005, 438: 671–674
Kitamuro T, Takahashi K, Ogawa K, et al. Bach1 functions as a hypoxia-inducible repressor for the heme oxygenase-1 gene in human cells. J Biol Chem, 2003, 278: 9125–9133
Shan Y, Zheng J, Lambrecht R W, et al. Reciprocal effects of micro-RNA-122 on expression of heme oxygenase-1 and hepatitis C virus genes in human hepatocytes. Gastroenterology, 2007, 133: 1166–1174
Zhu Z, Wilson A T, Mathahs M M, et al. Heme oxygenase-1 suppresses hepatitis C virus replication and increases resistance of hepatocytes to oxidant injury. Hepatology, 2008, 48: 1430–1439
Elbirt K K, Bonkovsky H L. Heme oxygenase: recent advances in understanding its regulation and role. Proc Assoc Am Physicians, 1999, 111: 438–447
Lohmann V, Korner F, Koch J, et al. Replication of subgenomic hepatitis C virus RNAs in a hepatoma cell line. Science, 1999, 285: 110–113
Chang J, Nicolas E, Marks D, et al. miR-122, a mammalian liver-specific microRNA, is processed from hcr mRNA and may downregulate the high affinity cationic amino acid transporter CAT-1. RNA Biol, 2004, 1: 106–113
Lanford R E, Hildebrandt-Eriksen E S, Petri A, et al. Therapeutic silencing of microRNA-122 in primates with chronic hepatitis C virus infection. Science, 2010, 327: 198–201
Wang G, Dong X, Hu J, et al. Long-term ex vivo monitoring of in vivo microRNA activity in liver using a secreted luciferase sensor. Sci China Life Sci, 2011, 54: 418–425
Jopling C L, Norman K L, Sarnow P. Positive and negative modulation of viral and cellular mRNAs by liver-specific microRNA miR-122. Cold Spring Harb Symp Quant Biol, 2006, 71: 369–376
Young D D, Connelly C M, Grohmann C, et al. Small molecule modifiers of microRNA miR-122 function for the treatment of hepatitis C virus infection and hepatocellular carcinoma. J Am Chem Soc, 2010, 132: 7976–7981
Moradpour D, Penin F, Rice C M. Replication of hepatitis C virus. Nat Rev Microbiol, 2007, 5: 453–463
Song Y, Friebe P, Tzima E, et al. The hepatitis C virus RNA 3′-untranslated region strongly enhances translation directed by the internal ribosome entry site. J Virol, 2006, 80: 11579–11588
Bradrick S S, Walters R W, Gromeier M. The hepatitis C virus 3′-untranslated region or a poly(A) tract promote efficient translation subsequent to the initiation phase. Nucleic Acids Res, 2006, 34: 1293–1303
Isken O, Baroth M, Grassmann C W, et al. Nuclear factors are involved in hepatitis C virus RNA replication. RNA, 2007, 13: 1675–1692
Niepmann M. Activation of hepatitis C virus translation by a liver-specific microRNA. Cell Cycle, 2009, 8: 1473–1477
Norman K L, Sarnow P. Modulation of hepatitis C virus RNA abundance and the isoprenoid biosynthesis pathway by microRNA miR-122 involves distinct mechanisms. J Virol, 2010, 84: 666–670
Jopling C L, Yi M, Lancaster A M, et al. Modulation of hepatitis C virus RNA abundance by a liver-specific MicroRNA. Science, 2005, 309: 1577–1581
Appel N, Bartenschlager R. A novel function for a micro RNA: negative regulators can do positive for the hepatitis C virus. Hepatology, 2006, 43: 612–615
Banaudha K, Kaliszewski M, Korolnek T, et al. MicroRNA silencing of tumor suppressor DLC-1 promotes efficient hepatitis C virus replication in primary human hepatocytes. Hepatology, 2011, 53: 53–61
Nakada C, Matsuura K, Tsukamoto Y, et al. Genome-wide microRNA expression profiling in renal cell carcinoma: significant down-regulation of miR-141 and miR-200c. J Pathol, 2008, 216: 418–427
Liu X, Wang T, Wakita T, et al. Systematic identification of microRNA and messenger RNA profiles in hepatitis C virus-infected human hepatoma cells. Virology, 2010, 398: 57–67
Yang Z, Ren F, Liu C, et al. dbDEMC: a database of differentially expressed miRNAs in human cancers. BMC Genomics, 2010, 11: S5
Li H P, Leu Y W, Chang Y S. Epigenetic changes in virus-associated human cancers. Cell Res, 2005, 15: 262–271
Huang J, Wang Y, Guo Y, et al. Down-regulated microRNA-152 induces aberrant DNA methylation in hepatitis B virus-related hepatocellular carcinoma by targeting DNA methyltransferase 1. Hepatology, 2010, 52: 60–70
Datta J, Kutay H, Nasser M W, et al. Methylation mediated silencing of microRNA-1 gene and its role in hepatocellular carcinogenesis. Cancer Res, 2008, 68: 5049–5058
Law P T, Wong N. Emerging roles of microRNA in the intracellular signaling networks of hepatocellular carcinoma. J Gastroenterol Hepatol, 2011, 26: 437–449
Shimizu S, Takehara T, Hikita H, et al. The let-7 family of microRNAs inhibits Bcl-xL expression and potentiates sorafenib-induced apoptosis in human hepatocellular carcinoma. J Hepatol, 2010, 52: 698–704
Tsang W P, Kwok T T. Epigallocatechin gallate up-regulation of miR-16 and induction of apoptosis in human cancer cells. J Nutr Biochem, 2010, 21: 140–146
Tsuruta T, Kozaki K, Uesugi A, et al. miR-152 is a tumor suppressor microRNA that is silenced by DNA hypermethylation in endometrial cancer. Cancer Res, 2011, 71: 6450–6462
Tsang T Y, Tang W Y, Chan J Y, et al. P-glycoprotein enhances radiation-induced apoptotic cell death through the regulation of miR-16 and Bcl-2 expressions in hepatocellular carcinoma cells. Apoptosis, 2011, 16: 524–535
Sang X, Zhao H, Lu X, et al. Prediction and identification of tumor-specific noncoding RNAs from human UniGene. Med Oncol, 2010, 27: 894–898
Chen W S, Leung C M, Pan H W, et al. Silencing of miR-1-1 and miR-133a-2 cluster expression by DNA hypermethylation in colorectal cancer. Oncol Rep, 2012, 28: 1069–1076
Chung G E, Yoon J H, Myung S J, et al. High expression of microRNA-15b predicts a low risk of tumor recurrence following curative resection of hepatocellular carcinoma. Oncol Rep, 2010, 23: 113–119
Zhang Y. Progress, challenges and new concepts in microRNAs. Sci China Life Sci, 2011, 54: 1096
Pineau P, Volinia S, McJunkin K, et al. miR-221 overexpression contributes to liver tumorigenesis. Proc Natl Acad Sci USA, 2010, 107: 264–269
Xu C, Liu S, Fu H, et al. MicroRNA-193b regulates proliferation, migration and invasion in human hepatocellular carcinoma cells. Eur J Cancer, 2010, 46: 2828–2836
Furuta M, Kozaki K I, Tanaka S, et al. miR-124 and miR-203 are epigenetically silenced tumor-suppressive microRNAs in hepatocellular carcinoma. Carcinogenesis, 2010, 31: 766–776
Zhao W Y, Wang D D, Song M Q, et al. Role of microRNA-223 and its target gene oncogene c-myc in hepatocellular carcinoma pathogenesis (in Chinese). Zhonghua Gan Zang Bing Za Zhi, 2011, 19: 114–117
Cheng J, Zhou L, Xie Q F, et al. The impact of miR-34a on protein output in hepatocellular carcinoma HepG2 cells. Proteomics, 2010, 10: 1557–1572
Hu L, Ibrahim S, Liu C, et al. Thrombin induces tumor cell cycle activation and spontaneous growth by down-regulation of p27Kip1, in association with the up-regulation of Skp2 and MiR-222. Cancer Res, 2009, 69: 3374–3381
Fornari F, Milazzo M, Chieco P, et al. MiR-199a-3p regulates mTOR and c-Met to influence the doxorubicin sensitivity of human hepatocarcinoma cells. Cancer Res, 2010, 70: 5184–5193
Henry J C, Park J K, Jiang J, et al. miR-199a-3p targets CD44 and reduces proliferation of CD44 positive hepatocellular carcinoma cell lines. Biochem Biophys Res Commun, 2010, 403: 120–125
Orian-Rousseau V, Chen L, Sleeman J P, et al. CD44 is required for two consecutive steps in HGF/c-Met signaling. Genes Dev, 2002, 16: 3074–3086
Mitchell P S, Parkin R K, Kroh E M, et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci USA, 2008, 105: 10513–10518
Chen X, Ba Y, Ma L, et al. Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res, 2008, 18: 997–1006
Gilad S, Meiri E, Yogev Y, et al. Serum microRNAs are promising novel biomarkers. PLoS ONE, 2008, 3: e3148
Wang Q Z, Xu W, Habib N, et al. Potential uses of microRNA in lung cancer diagnosis, prognosis, and therapy. Curr Cancer Drug Targets, 2009, 9: 572–594
Sukata T, Sumida K, Kushida M, et al. Circulating microRNAs, possible indicators of progress of rat hepatocarcinogenesis from early stages. Toxicol Lett, 2011, 200: 46–52
Zhou J, Yu L, Gao X, et al. Plasma microRNA panel to diagnose hepatitis B virus-related hepatocellular carcinoma. J Clin Oncol, 2011, 29: 4781–4788
Gui J, Tian Y, Wen X, et al. Serum microRNA characterization identifies miR-885-5p as a potential marker for detecting liver pathologies. Clin Sci (Lond), 2011, 120: 183–193
Li L M, Hu Z B, Zhou Z X, et al. Serum microRNA profiles serve as novel biomarkers for HBV infection and diagnosis of HBV-positive hepatocarcinoma. Cancer Res, 2010, 70: 9798–9807
Qu K Z, Zhang K, Li H, et al. Circulating microRNAs as biomarkers for hepatocellular carcinoma. J Clin Gastroenterol, 2011, 45: 355–360
Li J, Wang Y, Yu W, et al. Expression of serum miR-221 in human hepatocellular carcinoma and its prognostic significance. Biochem Biophys Res Commun, 2011, 406: 70–73
Miao R Y, Zhao H T, Yang H Y, et al. Postoperative adjuvant antiviral therapy for hepatitis B/C virus-related hepatocellular carcinoma: a meta-analysis. World J Gastroenterol, 2010, 16: 2931–2942
Xu J, Wu C, Che X, et al. Circulating microRNAs, miR-21, miR-122, and miR-223, in patients with hepatocellular carcinoma or chronic hepatitis. Mol Carcinog, 2011, 50: 136–142
Pavlidis N, Briasoulis E, Hainsworth J, et al. Diagnostic and therapeutic management of cancer of an unknown primary. Eur J Cancer, 2003, 39: 1990–2005
Landgraf P, Rusu M, Sheridan R, et al. A mammalian microRNA expression atlas based on small RNA library sequencing. Cell, 2007, 129: 1401–1414
Barshack I, Meiri E, Rosenwald S, et al. Differential diagnosis of hepatocellular carcinoma from metastatic tumors in the liver using microRNA expression. Int J Biochem Cell Biol, 2010, 42: 1355–1362
Wong C C, Wong C M, Tung E K, et al. The microRNA miR-139 suppresses metastasis and progression of hepatocellular carcinoma by down-regulating Rho-kinase 2. Gastroenterology, 2011, 140: 322–331
Sato F, Hatano E, Kitamura K, et al. MicroRNA profile predicts recurrence after resection in patients with hepatocellular carcinoma within the Milan Criteria. PLoS ONE, 2011, 6: e16435
Wong Q W, Ching A K, Chan A W, et al. MiR-222 overexpression confers cell migratory advantages in hepatocellular carcinoma through enhancing AKT signaling. Clin Cancer Res, 2010, 16: 867–875
Ji J, Zhao L, Budhu A, et al. Let-7g targets collagen type I alpha2 and inhibits cell migration in hepatocellular carcinoma. J Hepatol, 2010, 52: 690–697
Budhu A, Jia H L, Forgues M, et al. Identification of metastasis-related microRNAs in hepatocellular carcinoma. Hepatology, 2008, 47: 897–907
Cheng A L, Kang Y K, Chen Z, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol, 2009, 10: 25–34
Liu L, Cao Y, Chen C, et al. Sorafenib blocks the RAF/MEK/ERK pathway, inhibits tumor angiogenesis, and induces tumor cell apoptosis in hepatocellular carcinoma model PLC/PRF/5. Cancer Res, 2006, 66: 11851–11858
Cominetti M R, Martin A C, Ribeiro J U, et al. Inhibition of platelets and tumor cell adhesion by the disintegrin domain of human ADAM9 to collagen I under dynamic flow conditions. Biochimie, 2009, 91: 1045–1052
Josson S, Anderson C S, Sung S Y, et al. Inhibition of ADAM9 expression induces epithelial phenotypic alterations and sensitizes human prostate cancer cells to radiation and chemotherapy. Prostate, 2011, 71: 232–240
Peduto L. ADAM9 as a potential target molecule in cancer. Curr Pharm Des, 2009, 15: 2282–2287
Xu Q, Liu X, Cai Y, et al. RNAi-mediated ADAM9 gene silencing inhibits metastasis of adenoid cystic carcinoma cells. Tumour Biol, 2010, 31: 217–224
Zhou C, Liu J, Li Y, e al. microRNA-1274a, a modulator of sorafenib induced a disintegrin and metalloproteinase 9 (ADAM9) down-regulation in hepatocellular carcinoma. FEBS Lett, 2011, 585: 1828–1834
Bai S, Nasser M W, Wang B, et al. MicroRNA-122 inhibits tumorigenic properties of hepatocellular carcinoma cells and sensitizes these cells to sorafenib. J Biol Chem, 2009, 284: 32015–32027
Ji J, Shi J, Budhu A, et al. MicroRNA expression, survival, and response to interferon in liver cancer. N Engl J Med, 2009, 361: 1437–1447
Calin G A, Dumitru C D, Shimizu M, et al. Frequent deletions and down-regulation of microRNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci USA, 2002, 99: 15524–15529
He L, Thomson J M, Hemann M T, et al. A microRNA polycistron as a potential human oncogene. Nature, 2005, 435: 828–833
O’Donnell K A, Wentzel E A, Zeller K I, et al. c-Myc-regulated microRNAs modulate E2F1 expression. Nature, 2005, 435: 839–843
Kutay H, Bai S, Datta J, et al. Downregulation of miR-122 in the rodent and human hepatocellular carcinomas. J Cell Biochem, 2006, 99: 671–678
Jiang Q, Wang Y, Hao Y, et al. miR2Disease: a manually curated database for microRNA deregulation in human disease. Nucleic Acids Res, 2009, 37: D98–104
Sarver A L, Phalak R, Thayanithy V, et al. S-MED: sarcoma microRNA expression database. Lab Invest, 2010, 90: 753–761
Calin G A, Sevignani C, Dumitru C D, et al. Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci USA, 2004, 101: 2999–3004
Chen H, Chen Q, Fang M, et al. microRNA-181b targets MLK2 in HL-60 cells. Sci China Life Sci, 2010, 53: 101–106
Lu J, Getz G, Miska E A, et al. MicroRNA expression profiles classify human cancers. Nature, 2005, 435: 834–838
Kanehisa M, Araki M, Goto S, et al. KEGG for linking genomes to life and the environment. Nucleic Acids Res, 2008, 36: D480–484
Xiangji L, Feng X, Qingbao C, et al. Knockdown of HBV surface antigen gene expression by a lentiviral microRNA-based system inhibits HBV replication and HCC growth. J Viral Hepat, 2011, 18: 653–660
Jin W B, Wu F L, Kong D, et al. HBV-encoded microRNA candidate and its target. Comput Biol Chem, 2007, 31: 124–126
Ely A, Naidoo T, Mufamadi S, et al. Expressed anti-HBV primary microRNA shuttles inhibit viral replication efficiently in vitro and in vivo. Mol Ther, 2008, 16: 1105–1112
Chen G, Wang C, Shi T. Overview of available methods for diverse RNA-Seq data analyses. Sci China Life Sci, 2011, 54: 1121–1128
Qiu L, Fan H, Jin W, et al. miR-122-induced down-regulation of HO-1 negatively affects miR-122-mediated suppression of HBV. Biochem Biophys Res Commun, 2010, 398: 771–777
Li S, Yu B, Wang Y, et al. Identification and functional annotation of novel microRNAs in the proximal sciatic nerve after sciatic nerve transection. Sci China Life Sci, 2011, 54: 806–812
Hao M, Zheng S, Ding H, et al. Regulation of microRNA-122 on HBV replication by targeting HBx sequence (in Chinese). Sheng Wu Yi Xue Gong Cheng Xue Za Zhi, 2011, 28: 784–789, 803
Su C, Hou Z, Zhang C, et al. Ectopic expression of microRNA-155 enhances innate antiviral immunity against HBV infection in human hepatoma cells. Virol J, 2011, 8: 354
de Veer M J, Sledz C A, Williams B R. Detection of foreign RNA: implications for RNAi. Immunol Cell Biol, 2005, 83: 224–228
Grimm D, Streetz K L, Jopling C L, et al. Fatality in mice due to oversaturation of cellular microRNA/short hairpin RNA pathways. Nature, 2006, 441: 537–541
Kim D H, Rossi J J. Strategies for silencing human disease using RNA interference. Nat Rev Genet, 2007, 8: 173–184
Shah Y M, Morimura K, Yang Q, et al. Peroxisome proliferator-activated receptor alpha regulates a microRNA-mediated signaling cascade responsible for hepatocellular proliferation. Mol Cell Biol, 2007, 27: 4238–4247
Lan F F, Wang H, Chen Y C, et al. Hsa-let-7g inhibits proliferation of hepatocellular carcinoma cells by downregulation of c-Myc and upregulation of p16(INK4A). Int J Cancer, 2011, 128: 319–331
Skawran B, Steinemann D, Becker T, et al. Loss of 13q is associated with genes involved in cell cycle and proliferation in dedifferentiated hepatocellular carcinoma. Mod Pathol, 2008, 21: 1479–1489
Wu G, Yu F, Xiao Z, et al. Hepatitis B virus X protein downregulates expression of the miR-16 family in malignant hepatocytes in vitro. Br J Cancer, 2011, 105: 146–153
Connolly E, Melegari M, Landgraf P, et al. Elevated expression of the miR-17-92 polycistron and miR-21 in hepadnavirus-associated hepatocellular carcinoma contributes to the malignant phenotype. Am J Pathol, 2008, 173: 856–864
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at Springerlink.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Zhao, X., Yang, Z., Li, G. et al. The role and clinical implications of microRNAs in hepatocellular carcinoma. Sci. China Life Sci. 55, 906–919 (2012). https://doi.org/10.1007/s11427-012-4384-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-012-4384-x