Abstract
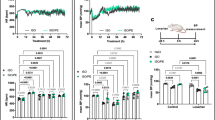
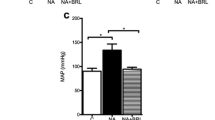
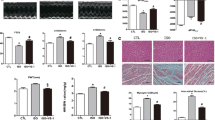
Heart disease is associated with increased sympathetic nerve activity and elevated levels of circulating catecholamines, resulting in chronic stimulation of the β-adrenergic receptors (β-AR) and consequent pathological cardiac remodeling. Experimentally, chronic administration of the β-AR agonist isoproterenol (ISO) has been most commonly used to model β-AR-induced cardiac remodeling. However, it remains unclear whether β-AR-mediated cardiac remodeling and dysfunction differs between sustained versus pulsatile (intermittent) exposure to a β-agonist. Here, we compare the effects of intermittent versus sustained administration of ISO on cardiac remodeling and function in mice. Animals were administered 5 mg (kg d)−1 ISO for 2 weeks either by daily subcutaneous injection, or continuous infusion via an implanted osmotic minipump. Cardiac function and remodeling were determined by echocardiography, micromanometry and histology. Moreover, Western blotting and quantitative real-time polymerase chain reaction (qRT-PCR) were utilized to define the proteins and genes involved. Both sustained and intermittent administration of ISO resulted in a similar degree of cardiac hypertrophy (16% and 19%, respectively). However, mice receiving ISO by daily injection developed more severe ventricular systolic and diastolic dysfunction and myocardial fibrosis compared with mice receiving ISO via the osmotic minipump. The disparity in results between the delivery methods is suggested to be due, at least in part, to increased expression of fibrogenic factors, including connective tissue growth factor (CTGF) and NADPH oxidase (NOX4), in mice receiving intermittent application of ISO. In summary, compared with sustained exposure to a β-AR agonist, intermittent β-AR stimulation leads to more severe cardiac dysfunction and fibrosis. These findings not only further our understanding of β-AR function in the setting of cardiac pathophysiology, but also highlight that significant differences can result dependent upon the mode of experimental β-AR stimulation in inducing cardiomyopathy.
Article PDF
Similar content being viewed by others
References
Floras J S. Sympathetic activation in human heart failure: diverse mechanisms, therapeutic opportunities. Acta Physiol Scand, 2003, 177: 391–398 12609011, 10.1046/j.1365-201X.2003.01087.x, 1:CAS:528:DC%2BD3sXitlOru7g%3D
Lohse M J, Engelhardt S, Eschenhagen T. What is the role of beta-adrenergic signaling in heart failure. Circ Res, 2003, 93: 896–906 14615493, 10.1161/01.RES.0000102042.83024.CA, 1:CAS:528:DC%2BD3sXoslykurk%3D
Singh K, Communal C, Sawyer D B, et al. Adrenergic regulation of myocardial apoptosis. Cardiovasc Res, 2000, 45: 713–719 10728393, 10.1016/S0008-6363(99)00370-3, 1:CAS:528:DC%2BD3cXltVGkuw%3D%3D
Fowler M B, Laser J A, Hopkins G L, et al. Assessment of the beta-adrenergic receptor pathway in the intact failing human heart: progressive receptor down-regulation and subsensitivity to agonist response. Circulation, 1986, 74: 1290–1302 3022962, 10.1161/01.CIR.74.6.1290, 1:STN:280:DyaL2s%2Flt1Smuw%3D%3D
Goldspink D F, Burniston J G, Ellison G M, et al. Catecholamine-induced apoptosis and necrosis in cardiac and skeletal myocytes of the rat in vivo: the same or separate death pathways. Exp Physiol, 2004, 89: 407–416 15131072, 10.1113/expphysiol.2004.027482, 1:CAS:528:DC%2BD2cXmslWktL0%3D
Communal C, Singh K, Pimentel D R, et al. Norepinephrine stimulates apoptosis in adult rat ventricular myocytes by activation of the beta-adrenergic pathway. Circulation, 1998, 98: 1329–1334 9751683, 1:CAS:528:DyaK1cXmslCntrY%3D
Bos R, Mougenot N, Findji L, et al. Inhibition of catecholamine-induced cardiac fibrosis by an aldosterone antagonist. J Cardiovasc Pharmacol, 2005, 45: 8–13 15613973, 10.1097/00005344-200501000-00003, 1:CAS:528:DC%2BD2cXhtFaitbzM
Grassi G, Quarti-Trevano F, Dell’oro R, et al. Essential hypertension and the sympathetic nervous system. Neurol Sci, 2008, 29: S33–36 18545892, 10.1007/s10072-008-0882-9
Flaa A, Eide I K, Kjeldsen S E, et al. Sympathoadrenal stress reactivity is a predictor of future blood pressure: an 18-year follow-up study. Hypertension, 2008, 52: 336–341 18574074, 10.1161/HYPERTENSIONAHA.108.111625, 1:CAS:528:DC%2BD1cXosF2jsL8%3D
Sharkey S W, Lesser J R, Zenovich A G, et al. Acute and reversible cardiomyopathy provoked by stress in women from the United States. Circulation, 2005, 111: 472–479 15687136, 10.1161/01.CIR.0000153801.51470.EB
Ueyama T, Kasamatsu K, Hano T, et al. Emotional stress induces transient left ventricular hypocontraction in the rat via activation of cardiac adrenoceptors: a possible animal model of ‘tako-tsubo’ cardiomyopathy. Circ J, 2002, 66: 712–713 12135146, 10.1253/circj.66.712
Ueyama T, Senba E, Kasamatsu K, et al. Molecular mechanism of emotional stress-induced and catecholamine-induced heart attack. J Cardiovasc Pharmacol, 2003, 41: S115–118 12688407, 1:CAS:528:DC%2BD3sXos1WqsA%3D%3D
Rona G, Chappel C I, Balazs T, et al. An infarct-like myocardial lesion and other toxic manifestations produced by isoproterenol in the rat. AMA Arch Pathol, 1959, 67: 443–455 13636626, 1:CAS:528:DyaG1MXnvVyrsw%3D%3D
Zbinden G, Moe R A. Pharmacological studies on heart muscle lesions induced by isoproterenol. Ann N Y Acad Sci, 1969, 156: 294–308 4400975, 10.1111/j.1749-6632.1969.tb16736.x, 1:CAS:528:DyaF1MXhtVCqs7g%3D
Kung H F, Blau M. Subcutaneous isoproterenol: a convenient rat model for early detection of myocardial necrosis. J Nucl Med, 1978, 19: 948–951 682027, 1:CAS:528:DyaE1cXlsF2mt7k%3D
Kitagawa Y, Yamashita D, Ito H, et al. Reversible effects of isoproterenol-induced hypertrophy on in situ left ventricular function in rat hearts. Am J Physiol Heart Circ Physiol, 2004, 287: H277–285 15016627, 10.1152/ajpheart.00073.2004, 1:CAS:528:DC%2BD2cXmtVelur0%3D
Benjamin I J, Jalil J E, Tan L B, et al. Isoproterenol-induced myocardial fibrosis in relation to myocyte necrosis. Circ Res, 1989, 65: 657–670 2527639, 1:CAS:528:DyaL1MXlsFWgurw%3D
Clerk A, Michael A, Sugden P H. Stimulation of the p38 mitogen-activated protein kinase pathway in neonatal rat ventricular myocytes by the G protein-coupled receptor agonists, endothelin-1 and phenylephrine: a role in cardiac myocyte hypertrophy. J Cell Biol, 1998, 142: 523–535 9679149, 10.1083/jcb.142.2.523, 1:CAS:528:DyaK1cXltFGqs7Y%3D
Bueno O F, De Windt L J, Tymitz K M, et al. The MEK1-ERK1/2 signaling pathway promotes compensated cardiac hypertrophy in transgenic mice. EMBO J, 2000, 19: 6341–6350 11101507, 10.1093/emboj/19.23.6341, 1:CAS:528:DC%2BD3MXjtF2htr4%3D
Ramirez M T, Sah V P, Zhao X L, et al. The MEKK-JNK pathway is stimulated by alpha1-adrenergic receptor and ras activation and is associated with in vitro and in vivo cardiac hypertrophy. J Biol Chem, 1997, 272: 14057–14061 9162028, 10.1074/jbc.272.22.14057, 1:CAS:528:DyaK2sXjsFKqur8%3D
Boluyt M O, Zheng J S, Younes A, et al. Rapamycin inhibits alpha 1-adrenergic receptor-stimulated cardiac myocyte hypertrophy but not activation of hypertrophy-associated genes. Evidence for involvement of p70 S6 kinase. Circ Res, 1997, 81: 176–186 9242178, 1:CAS:528:DyaK2sXkvFyksL8%3D
Sadoshima J, Izumo S. Rapamycin selectively inhibits angiotensin II-induced increase in protein synthesis in cardiac myocytes in vitro. Potential role of 70-kD S6 kinase in angiotensin II-induced cardiac hypertrophy. Circ Res, 1995, 77: 1040–1052 7586215, 1:CAS:528:DyaK2MXpsFCitr8%3D
Hu Y, Zhang Y, Venkitaramani D V, et al. Translation of striatal-enriched protein tyrosine phosphatase (STEP) after beta1-adrenergic receptor stimulation. J Neurochem, 2007, 103: 531–541 17623046, 10.1111/j.1471-4159.2007.04749.x, 1:CAS:528:DC%2BD2sXht1KqsLzK
Pesce L, Comellas A, Sznajder J I. Beta-adrenergic agonists regulate Na-K-ATPase via p70S6k. Am J Physiol Lung Cell Mol Physiol, 2003, 285: L802–807 12704019, 1:CAS:528:DC%2BD3sXosV2qsLg%3D
Dhalla N S, Temsah R M, Netticadan T. Role of oxidative stress in cardiovascular diseases. J Hypertens, 2000, 18: 655–673 10872549, 10.1097/00004872-200018060-00002, 1:CAS:528:DC%2BD3cXksFWrur4%3D
Zhang G X, Kimura S, Nishiyama A, et al. Cardiac oxidative stress in acute and chronic isoproterenol-infused rats. Cardiovasc Res, 2005, 65: 230–238 15621051, 10.1016/j.cardiores.2004.08.013, 1:CAS:528:DC%2BD2cXhtFCqt7fM
Ruperez M, Lorenzo O, Blanco-Colio L M, et al. Connective tissue growth factor is a mediator of angiotensin II-induced fibrosis. Circulation, 2003, 108: 1499–1505 12952842, 10.1161/01.CIR.0000089129.51288.BA, 1:CAS:528:DC%2BD3sXntlartbk%3D
Wang J, Xu N, Feng X, et al. Targeted disruption of Smad4 in cardiomyocytes results in cardiac hypertrophy and heart failure. Circ Res, 2005, 97: 821–828 16151019, 10.1161/01.RES.0000185833.42544.06, 1:CAS:528:DC%2BD2MXhtVymt7nK
Liao W, Wang S, Han C, et al. 14-3-3 proteins regulate glycogen synthase 3beta phosphorylation and inhibit cardiomyocyte hypertrophy. FEBS J, 2005, 272: 1845–1854 15819880, 10.1111/j.1742-4658.2005.04614.x, 1:CAS:528:DC%2BD2MXjslGktLg%3D
Perrino C, Naga P S V, Mao L, et al. Intermittent pressure overload triggers hypertrophy-independent cardiac dysfunction and vascular rarefaction. J Clin Invest, 2006, 116: 1547–1560 16741575, 10.1172/JCI25397, 1:CAS:528:DC%2BD28XlsFWrtbg%3D
Han Y S, Tveita T, Kondratiev T V, et al. Changes in cardiovascular beta-adrenoceptor responses during hypothermia. Cryobiology, 2008, 57: 246–250 18834873, 10.1016/j.cryobiol.2008.09.006, 1:CAS:528:DC%2BD1cXhsVChtb3L
Kralova E, Mokran T, Murin J, et al. Electrocardiography in two models of isoproterenol-induced left ventricular remodeling. Physiol Res, 2008, 57: S83–89 18373388, 1:CAS:528:DC%2BD1cXhtVSktbrP
Hohimer A R, Davis L E, Hatton D C. Repeated daily injections and osmotic pump infusion of isoproterenol cause similar increases in cardiac mass but have different effects on blood pressure. Can J Physiol Pharmacol, 2005, 83: 191–197 15791293, 10.1139/y04-137, 1:CAS:528:DC%2BD2MXjs12ru7c%3D
Freund C, Schmidt-Ullrich R, Baurand A, et al. Requirement of nuclear factor-kappaB in angiotensin II- and isoproterenol-induced cardiac hypertrophy in vivo. Circulation, 2005, 111: 2319–2325 15870116, 10.1161/01.CIR.0000164237.58200.5A, 1:CAS:528:DC%2BD2MXjsl2itrc%3D
Xu Q, Dalic A, Fang L, et al. Myocardial oxidative stress contributes to transgenic beta-adrenoceptor activation-induced cardiomyopathy and heart failure. Br J Pharmacol, 2011, 162: 1012–1028 20955367, 10.1111/j.1476-5381.2010.01043.x, 1:CAS:528:DC%2BC3MXhslWgsro%3D
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Ma, X., Song, Y., Chen, C. et al. Distinct actions of intermittent and sustained β-adrenoceptor stimulation on cardiac remodeling. Sci. China Life Sci. 54, 493–501 (2011). https://doi.org/10.1007/s11427-011-4183-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4183-9