Abstract
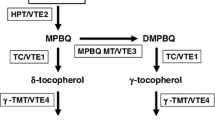
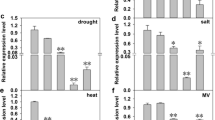
Tocopherols synthesized exclusively by photosynthetic organisms are major antioxidants in biomembranes. In plants, tocopherol cyclase (TC/VTE1) catalyzes the conversion of 2,3-dimethyl-5-phytyl-1,4-benzoquinone (DMPBQ) to γ-tocopherol. In the present study, OsVTE1, which encodes a rice tocopherol cyclase ortholog, was cloned and characterized. OsVTE1 was induced significantly by abiotic stresses such as high salt, H2O2, drought, cold and by the plant hormones ABA and salicylic acid. The tissue-specific expression pattern and OsVTE1-promoter GUS activity assay showed that OsVTE1 was mainly expressed in the leaf, and also could be detected in the root, stem and panicle. Compared with control plants, transgenic plants with Os-VTE1 RNA interference (OsVTE1-RNAi) were more sensitive to salt stress whereas, in contrast, transgenic plants overexpressing OsVTE1 (OsVTE1-OX) showed higher tolerance to salt stress. The DAB in vivo staining showed that OsVTE1-OX plants accumulated less H2O2 than did control plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Munne-Bosch S, Alegre L. The function of tocopherols and tocotrienols in plants. Crit Rev Plant Sci, 2002, 21: 31–57, 1:CAS:528:DC%2BD38XhvFGhtro%3D
Bramley P M, Elmadfa I, Kafatos A, et al. Vitamin E. J Sci Food Agric, 2000, 80: 913–938, 1:CAS:528:DC%2BD3cXjsFals7k%3D, 10.1002/(SICI)1097-0010(20000515)80:7<913::AID-JSFA600>3.0.CO;2-3
Szymańska R, Kruk J. Occurrence and function of tocochromanols in plants, animals and men. Postepy Biochem, 2007, 53: 174–181, 17969879
Neely W C, Martin J M, Barker S A. Products and relative reaction rates of the oxidation of tocopherols with singlet molecular oxygen. Photochem Photobiol, 1988, 48: 423–428, 1:CAS:528:DyaL1MXjsV2isA%3D%3D, 10.1111/j.1751-1097.1988.tb02840.x, 3231678
Grusak M A, DellaPenna D. Improving the nutrient composition of plants to enhance human nutrition and health. Annu Rev Plant Physiol Plant Mol Biol, 1999, 50: 133–161, 1:CAS:528:DyaK1MXkt1yksLk%3D, 10.1146/annurev.arplant.50.1.133, 15012206
Desel C, Hubbermann E M, Schwarz K, et al. Nitration of γ-tocopherol in plant tissues. Planta, 2007, 226: 1311–1322, 1:CAS:528:DC%2BD2sXhtVWjsbvM, 10.1007/s00425-007-0552-9, 17619201
Cheng Z, Sattler S, Maeda H, et al. Highly divergent methyltransferases catalyze a conserved reaction in tocopherol and plastoquinone synthesis in cyanobacteria and photosynthetic eukaryotes. Plant Cell, 2003, 15: 2343–2356, 1:CAS:528:DC%2BD3sXotlGmtLg%3D, 10.1105/tpc.013656, 14508009
Sattler S E, Cahoon E B, Coughlan S J, et al. Characterization of tocopherol cyclases from higher plants and cyanobacteria: Evolutionary implications for tocopherol synthesis and function. Plant Physiol, 2003, 132: 2184–2195, 1:CAS:528:DC%2BD3sXmsVanur8%3D, 10.1104/pp.103.024257, 12913173
Arango Y, Heise K P. Tocopherol synthesis from homogentisate in Capsicum annuum L. (yellow pepper) chromoplast membranes: Evidence for tocopherol cyclase. Biochem J, 1998, 336: 531–533, 1:CAS:528:DyaK1MXjvVCjtw%3D%3D, 9841861
Porfirova S, Bergmuller E, Tropf S, et al. Isolation of an Arabidopsis mutant lacking vitamin E and identification of a cyclase essential for all tocopherol biosynthesis. Proc Natl Acad Sci USA, 2002, 99: 12495–12500, 1:CAS:528:DC%2BD38XntlCkt7g%3D, 10.1073/pnas.182330899, 12213958
Shintani D, DellaPenna D. Elevating the vitamin E content of plants through metabolic engineering. Science, 1998, 282: 2098–2100, 1:CAS:528:DyaK1cXotVyisrc%3D, 10.1126/science.282.5396.2098, 9851934
Kanwischer M, Porfirova S, Bergmuller E, et al. Alterations in tocopherol cyclase activity in transgenic and mutant plants of Arabidopsis affect tocopherol content, tocopherol composition, and oxidative stress. Plant Physiol, 2005, 137: 713–723, 1:CAS:528:DC%2BD2MXhs1Kqtro%3D, 10.1104/pp.104.054908, 15665245
Bolwell G P, Wojtaszek P. Mechanisms for the generation of reactive oxygen species in plant defence-a broad perspective. Physiol Mol Plant Pathol, 1997, 51: 347–366, 1:CAS:528:DyaK1cXjsVSgurg%3D, 10.1006/pmpp.1997.0129
Lamb C, Dixon R A. The oxidative burst in plant disease resistance. Annu Rev Plant Physiol Plant Mol Biol, 1997, 48: 251–275, 1:CAS:528:DyaK2sXjs1entr8%3D, 10.1146/annurev.arplant.48.1.251, 15012264
Noctor G, Foyer C H. Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol, 1998, 49: 249–279, 1:CAS:528:DyaK1cXjvVShtrc%3D, 10.1146/annurev.arplant.49.1.249, 15012235
Karpinski S, Reynolds H, Karpinska B, et al. Systemic signaling and acclimation in response to excess excitation energy in Arabidopsis. Science, 1999, 284: 654–657, 1:CAS:528:DyaK1MXislyrtbc%3D, 10.1126/science.284.5414.654, 10213690
Orozco-Cardenas M, Ryan C A. Hydrogen peroxide is generated systemically in plant leaves by wounding and systemin via the octa decanoid pathway. Proc Natl Acad Sci USA, 1999, 96: 6553–6557, 1:CAS:528:DyaK1MXksFKkur0%3D, 10.1073/pnas.96.11.6553, 10339626
Dat J, Vandenabeele S, Vranová E, et al. Dual action of the active oxygen species during plant stress responses. Cell Mol Life Sci, 2000, 57: 779–795, 1:CAS:528:DC%2BD3cXksFyisrk%3D, 10.1007/s000180050041, 10892343
Alscher R G, Erturk N, Heath L S. Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J Exp Bot, 2002, 53: 1331–1341, 1:CAS:528:DC%2BD38XktFSlsL4%3D, 10.1093/jexbot/53.372.1331, 11997379
Shao H B, Chu L Y. Plant molecular biology in China: Opportunities and Challenges. Plant Mol Biol Rep, 2005, 23: 345–358, 1:CAS:528:DC%2BD28XmvFGisLc%3D, 10.1007/BF02788883
Shao H B, Chu L Y, Wu G, et al. Changes of some anti-oxidative physiological indices under soil water deficits among 10 wheat (Triticum aestivum L.) genotypes at tillering stage. Colloids Surf B Biointerfaces, 2007, 54: 143–149, 1:CAS:528:DC%2BD2sXpvVOqtA%3D%3D, 10.1016/j.colsurfb.2006.09.004, 17196377
Wu G, Wei Z K, Shao H B. The mutual responses of higher plants to environment: physiological and microbiological aspects. Colloids Surf B Biointerfaces, 2007, 59: 113–119, 1:CAS:528:DC%2BD2sXovFygtL4%3D, 10.1016/j.colsurfb.2007.05.003
Pourcel L, Routaboul J M, Cheynier V. Flavonoid oxidation in plants: from biochemical properties to physiological functions. Trends Plant Sci, 2007, 12: 29–36, 1:CAS:528:DC%2BD2sXntlymsQ%3D%3D, 10.1016/j.tplants.2006.11.006, 17161643
Lee K, Lee S M, Park S R, et al. Overexpression of Arabidopsis homogentisate phytyltransferase or tocopherol cyclase elevates vitamin E content by increasing gamma-tocopherol level in lettuce (Lactuca sativa L.). Mol Cells, 2007, 24: 301–306, 1:CAS:528:DC%2BD2sXht12jtb7M, 17978586
Liu X, Hua X, Guo J, et al. Enhanced tolerance to drought stress in transgenic tobacco plants overexpressing VTE1 for increased tocopherol production from Arabidopsis thaliana. Biotechnol Lett, 2008, 30: 1275–1280, 1:CAS:528:DC%2BD1cXmslSqu78%3D, 10.1007/s10529-008-9672-y, 18317702
Zhang J S, Gu J, Liu F H, et al. A gene encoding a truncated large subunit of rubisco is transcribed and salt-inducible in rice. Theor Appl Genet, 1995, 91: 361–366, 1:CAS:528:DyaK2MXotl2qtb0%3D, 10.1007/BF00220900
Xie C, Zhang Z G, Zhang J S, et al. Spatial expression and characterization of a putative ethylene receptor protein NTHK1 in tobacco. Plant Cell Physiol, 2002, 43: 810–815, 1:CAS:528:DC%2BD38XlsFOitr8%3D, 10.1093/pcp/pcf095, 12154144
Hiei Y, Ohta S, Komari T, et al. Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J, 1994, 6: 271–282, 1:CAS:528:DyaK2MXhtlGntLc%3D, 10.1046/j.1365-313X.1994.6020271.x, 7920717
Inskeep W P, Bloom P R. Extinction coefficients of chlorophyll a and b in N,N-dimethylformamide and 80% acetone. Plant Physiol, 1985, 77: 483–485, 1:CAS:528:DyaL2MXht12jtrc%3D, 10.1104/pp.77.2.483, 16664080
Jefferson R A, Kavanagh T A, Bevan M W. GUS fusions: Beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J, 1987, 6: 3901–3907, 1:CAS:528:DyaL1cXovV2itQ%3D%3D, 3327686
Schreiber U, Hormann H, Neubauer C, et al. Assessment of photosystem II photochemical quantum yield by chlorophyll fluorescence quenching analysis. Aust J Plant Physiol, 1995, 22: 209–220, 1:CAS:528:DyaK2MXmt1Oisro%3D, 10.1071/PP9950209
Hasegawa P M, Bressan R A, Zhu J K, et al. Plant cellular and molecular responses to high salinity. Annu Rev Plant Physiol Plant Mol Biol, 2000, 51: 463–499, 1:CAS:528:DC%2BD3cXlsVymt7s%3D, 10.1146/annurev.arplant.51.1.463, 15012199
Brigelius-Flohe R, Traber M G. Vitamin E: function and metabolism. FASEB J, 1999, 13: 1145–1155, 1:CAS:528:DyaK1MXktlOrtbs%3D, 10385606
Ricciarelli R, Zingg J M, Azzi A. Vitamin E: protective role of a Janus molecule. FASEB J, 2001, 15: 2314–2325, 1:CAS:528:DC%2BD3MXoslSjurk%3D, 10.1096/fj.01-0258rev, 11689457
Collakova E, DellaPenna D. The role of homogentisate phytyltransferase and other tocopherol pathway enzymes in the regulation of tocopherol synthesis during abiotic stress. Plant Physiol, 2003, 133: 930–940, 1:CAS:528:DC%2BD3sXosVaqurk%3D, 10.1104/pp.103.026138, 14512521
Havaux M, Eymery F, Porfirova S, et al. Vitamin E protects against photoinhibition and photooxidative stress in Arabidopsis thaliana. Plant Cell, 2005, 17: 3451–3469, 1:CAS:528:DC%2BD2MXhtlersL3N, 10.1105/tpc.105.037036, 16258032
Collakova E, DellaPenna D. Isolation and functional analysis of homogentisate phytyltransferase from Synechocystis sp. PCC 6803 and Arabidopsis. Plant Physiol, 2001, 127: 1113–1124, 1:CAS:528:DC%2BD3MXos1KmsLc%3D, 10.1104/pp.010421, 11706191
Foyer C H, Noctor G. Redox sensing and signalling associated with reactive oxygen in chloroplasts, peroxisomes and mitochondria. Physiol Plant, 2003, 119: 355–364, 1:CAS:528:DC%2BD3sXoslekt74%3D, 10.1034/j.1399-3054.2003.00223.x
Abbasi A R, Hajirezaei M, Hofius D, et al. Specific roles of alpha- and gamma-tocopherol in abiotic stress responses of transgenic tobacco. Plant Physiol, 2007, 143: 1720–1738, 1:CAS:528:DC%2BD2sXksFWjtLc%3D, 10.1104/pp.106.094771, 17293434
Kruk J, Holländer-Czytko H, Oettmeier W, et al. Tocopherol as singlet oxygen scavenger in photosystem II. J Plant Physiol, 2005, 162: 749–757, 1:CAS:528:DC%2BD2MXntVSmtbs%3D, 10.1016/j.jplph.2005.04.020, 16008099
Bergmüller E, Porfirova S, Dörmann P. Characterization of an Arabidopsis mutant deficient in gamma-tocopherol methyltransferase. Plant Mol Biol, 2003, 52: 1181–1190, 10.1023/B:PLAN.0000004307.62398.91, 14682617
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Ouyang, S., He, S., Liu, P. et al. The role of tocopherol cyclase in salt stress tolerance of rice (Oryza sativa). Sci. China Life Sci. 54, 181–188 (2011). https://doi.org/10.1007/s11427-011-4138-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4138-1