Abstract
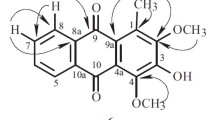
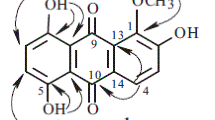
Two tetrahydroxanthones, 1,3,5S,8S-tetrahydroxy-5,6,7,8-tetrahydroxanthone (1) and 1,3,5R,8S-tetrahydroxy-5,6,7,8-tetrahydroxanthone (2), and six new tetrahydroxanthone glycosides, amarellins A–F (3–8), were isolated from the aerial parts of a Mongolian medicinal plant Gentianella amarella ssp. acuta (Gentianaceae). The structures of 1–8 were elucidated on the basis of spectroscopic analysis, chemical conversion, and ECD calculation. Amarellins A–C (3–5) were assigned as 8-O-β-d-glucoside, 8-O-β-d-xyloside, and 1-O-β-d-glucoside of 1, respectively, while amarellins D–F (6–8) were elucidated to be 8-O-β-d-xyloside, 1-O-β-d-glucoside, and 3-O-β-d-glucoside of 2, respectively.






Similar content being viewed by others
References
Struwe L (2014) Classification and evolution of the family Gentianaceae. In: Rybczyński JJ, Davey MR, Mikula A (eds) The Gentianaceae. Characterization and ecology, vol 1. Springer, Berlin, pp 13–35
Li MH, Zhou LS, Fang HY, Song XL, Zhang N (2010) Quantification of xanthones in a Mongolian health tea using high-performance liquid chromatography. J Med Plants Res 4:1704–1707
Lv LJ, Li MH (2009) Terpenoids, flavonoids and xanthones from Gentianella acuta (Gentianaceae). Biochem Syst Ecol 37:497–500
Tanaka N, Takekata M, Kurimoto SI, Kawazoe K, Murakami K, Damdinjav D, Dorjbal E, Kashiwada Y (2015) Algiolide A, secoiridoid glucoside from Mongolian medicinal plant Gentiana algida. Tetrahedron Lett 56:817–819
Kaldas M, Miura I, Hostettmann K (1978) Campestroside, a new tetrahydroxanthone glucoside from Gentiana campestris. Phytochemistry 17:295–297
Zhou HM, Liu YL, Blaskó G, Cordell GA (1989) Swertiabisxanthone-I from Swertia macrosperma. Phytochemistry 28:3569–3571
Urbain A, Marston A, Sintra Grilo L, Bravo J, Purev O, Purevsuren B, Batsuren D, Reist M, Carrupt PA, Hostettmann K (2008) Xanthones from Gentianella amarella ssp. acuta with acetylcholinesterase and monoamine oxidase inhibitory activities. J Nat Prod 71:895–897
Ishimaru K, Sudo H, Satake M, Matsunaga Y, Hasegawa Y, Takemoto S, Shimomura K (1990) Amarogentin, amaroswerin and four xanthones from hairy root cultures of Swertia japonica. Phytochemistry 29:1563–1565
Tomimori T, Komatsu M (1969) Studies on the constituents of Swertia japonica. VI. On the flavonoid and xanthone constituents of Swertia randaiensis. Hayata and S. swertopsis Makino. Yakugaku Zasshi 89:1276–1282
Faizi S, Zikr-ur-Rehman S, Ali M, Naz A (2006) Temperature and solvent dependent NMR studies on mangiferin and complete NMR spectral assignments of its acyl and methyl derivatives. Magn Reson Chem 44:838–844
Inouye H, Ueda S, Nakamura Y (1966) Structure of sweroside, a new glucoside from Swertia japonica. Tetrahedron Lett 7:5229–5234
Kubota T, Tomita Y (1961) The structure of swertiamarin. Tetrahedron Lett 2:176–182
Ikeshiro Y, Tomita Y (1984) A new iridoid glucoside of Swertia japonica. Planta Med 50:485–488
Kizu H, Shimana H, Tomimori T (1995) Studies on the constituents of Clematis species. VI. The constituents of Clematis stans Sieb. et Zucc. Chem Pharm Bull 43:2187–2194
Hase K, Li J, Basnet P, Xiong Q, Takamura S, Namba T, Kadota S (1997) Hepatoprotective principles of Swertia japonica Makino on d-galactosamine/lipopolysaccharide-induced liver injury in mice. Chem Pharm Bull 45:1823–1827
Du XG, Wang W, Zhang QY, Cheng J, Avula B, Khan IA, Guo DA (2012) Identification of xanthones from Swertia punicea using high-performance liquid chromatography coupled with electrospray ionization tandem mass spectrometry. Rapid Commun Mass Spectrom 26:2913–2923
Wang QH, Bao B, Chen YL (2014) Structure elucidation and NMR assignments of two unusual xanthones from Lomatogonium carinthiacum (Wulf) Reichb. Magn Reson Chem 52:37–39
Tanaka T, Nakashima T, Ueda T, Tomii K, Kouno I (2007) Facile discrimination of aldose enantiomers by reversed-phase HPLC. Chem Pharm Bull 55:899–901
Du XG, Wang W, Zhang SP, Pu XP, Zhang QY, Ye M, Zhao YY, Wang BR, Khan IA, Guo DA (2010) Neuroprotective xanthone glycosides from Swertia punicea. J Nat Prod 73:1422–1426
Zhou NJ, Geng CA, Huang XY, Ma YB, Zhang XM, Wang JL, Chen JJ (2015) Anti-hepatitis B virus active constituents from Swertia chirayita. Fitoterapia 100:27–34
Spartan’10, Wavefunction Inc., Irvine, CA
Gaussian 09, Revision C.01, Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski, JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2010) Gaussian, Inc., Wallingford, CT
Bruhn T, Schaumlöffel A, Hemberger Y, Bringmann G (2013) SpecDis, version 1.61. University of Würzburg, Germany
Acknowledgments
This work was partly supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lu, S., Tanaka, N., Kawazoe, K. et al. Tetrahydroxanthones from Mongolian medicinal plant Gentianella amarella ssp. acuta . J Nat Med 70, 780–788 (2016). https://doi.org/10.1007/s11418-016-1015-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-016-1015-z