Abstract
Purpose
Soil matric suction, or negative water potential, contributes to soil physical stability (e.g., slope). Plants retain soil matric suction as they take up and transpire water in soil. This study investigated the effects of the common plant root-fungi symbiosis on the changes of the low-range matric suction (i.e., 0–100 kPa), which is relevant to slope stability.
Methods
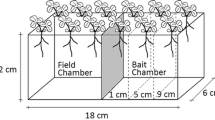
Bermuda grass (Cynodon dactylon) was grown in compacted soil (dry density ~ 1680 kg m−3) with and without the inoculation of an arbuscular mycorrhizal fungus (AMF) (Rhizophagus intraradices). Matric suction along the soil depth was measured during wetting and drying. Biomass, fungal colonization rate, and wet-aggregates size distribution were investigated subsequently.
Results
AMF association did not exert significant effects on the matric suction (range 0–80 kPa) distribution compared with the control, during wetting and drying. AMF significantly decreased the root biomass compared with the control. Significantly, higher proportions of 2–5-mm wet-aggregates were observed in the inoculated treatment than the control.
Conclusion
AMF can hinder root growth, and the choice of plant-fungus combination should not be neglected. Higher root biomass is preferred during drying in order to achieve a higher initial suction before the rainfall. Promoting root biomass would be more essential than shoot biomass during wetting (heavy rainfall). The application of AMF should be considered for soil erosion control during the early stage of restoration works.





Similar content being viewed by others
References
Abbott LK, Lumley S (2014) Assessing economic benefits of arbuscular mycorrhizal fungi as a potential indicator of soil health. In: Solaiman ZM, Abbott LK, Varma A (eds) Mycorrhizal fungi: use in sustainable agriculture and land restoration. Springer, Berlin Heidelberg, pp 17–31
Allen MF (2007) Mycorrhizal fungi: highways for water and nutrients in arid soils. Vadose Zone J 6:291–297. https://doi.org/10.2136/vzj2006.0068
Allen MF (2009) Bidirectional water flows through the soil–fungal–plant mycorrhizal continuum. New Phytol 182:290–293. https://doi.org/10.1111/j.1469-8137.2009.02815.x
ASTM (2007) Test methods for laboratory compaction characteristics of soil using standard effort (12 400 ft-lbf/ft3 (600 kN-m/m3)). In: ASTM Volume 04.08 Soil and Rock (I). ASTM International, West Conshohocken, pp D421–D5876
Augé RM (2001) Water relations, drought and vesicular-arbuscular mycorrhizal symbiosis. Mycorrhiza 11:3–42. https://doi.org/10.1007/s005720100097
Augé RM, Stodola AJW, Tims JE, Saxton AM (2001) Moisture retention properties of a mycorrhizal soil. Plant Soil 230:87–97. https://doi.org/10.1023/A:1004891210871
Barrett G, Campbell C, Fitter A, Hodge A (2011) The arbuscular mycorrhizal fungus Glomus hoi can capture and transfer nitrogen from organic patches to its associated host plant at low temperature. Appl Soil Ecol 48:102–105. https://doi.org/10.1016/j.apsoil.2011.02.002
Bárzana G, Aroca R, Paz JA, Chaumont F, Martinez-Ballesta MC, Carvajal M, Ruiz-Lozano JM (2012) Arbuscular mycorrhizal symbiosis increases relative apoplastic water flow in roots of the host plant under both well-watered and drought stress conditions. Ann Bot 109:1009–1017. https://doi.org/10.1093/aob/mcs007
Bast A, Wilcke W, Graf F, Lüscher P, Gärtner H (2014a) The use of mycorrhiza for eco-engineering measures in steep alpine environments: effects on soil aggregate formation and fine-root development. Earth Surf Process Landf 39:1753–1763. https://doi.org/10.1002/esp.3557
Bast A, Wilcke W, Lüscher P et al (2014b) Aggregate stability, root length and root thickness influenced by a mycorrhizal inoculum? - Results from a three-year eco-engineering field experiment on an alpine slope. Presented at the EGU General Assembly Conference Abstracts, p 15498
Bast A, Grimm M, Graf F et al (2015) Does mycorrhizal inoculation benefit plant survival, plant development and small-scale soil fixation? Results from a perennial eco-engineering field experiment in the Swiss Alps. Presented at the EGU General Assembly Conference Abstracts, p 9279
Bearden BN (2001) Influence of arbuscular mycorrhizal fungi on soil structure and soil water characteristics of vertisols. Plant Soil 229:245–258. https://doi.org/10.1023/A:1004835328943
Bearden BN, Petersen L (2000) Influence of arbuscular mycorrhizal fungi on soil structure and aggregate stability of a vertisol. Plant Soil 218:173–183. https://doi.org/10.1023/A:1014923911324
Bencherif K, Boutekrabt A, Dalpé Y, Lounès-Hadj Sahraoui A (2016) Soil and seasons affect arbuscular mycorrhizal fungi associated with Tamarix rhizosphere in arid and semi-arid steppes. Appl Soil Ecol 107:182–190. https://doi.org/10.1016/j.apsoil.2016.06.003
Bryla DR, Duniway JM (1997) Water uptake by safflower and wheat roots infected with arbuscular mycorrhizal fungi. New Phytol 136:591–601. https://doi.org/10.1046/j.1469-8137.1997.00781.x
Burri K, Gromke C, Graf F (2013) Mycorrhizal fungi protect the soil from wind erosion: a wind tunnel study. Land Degrad Dev 24:385–392. https://doi.org/10.1002/ldr.1136
Cornelissen JHC, Lavorel S, Garnier E, Díaz S, Buchmann N, Gurvich DE, Reich PB, Steege H, Morgan HD, Heijden MGA, Pausas JG, Poorter H (2003) A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Aust J Bot 51:335–380
Duddridge JA, Malibari A, Read DJ (1980) Structure and function of mycorrhizal rhizomorphs with special reference to their role in water transport. Nature 287:834–836. https://doi.org/10.1038/287834a0
Fredlund DG, Rahardjo H (1993) Soil mechanics for unsaturated soils, 1st edn. Wiley-Interscience, New York
Gafur S (2014) Contribution of dynamics of root colonisation by arbuscular mycorrhizal communities to ecosystem function. In: Solaiman ZM, Abbott LK, Varma A (eds) Mycorrhizal fungi: use in sustainable agriculture and land restoration. Springer, Berlin Heidelberg, pp 33–43
Gholamhoseini M, Ghalavand A, Dolatabadian A, Jamshidi E, Khodaei-Joghan A (2013) Effects of arbuscular mycorrhizal inoculation on growth, yield, nutrient uptake and irrigation water productivity of sunflowers grown under drought stress. Agric Water Manag 117:106–114. https://doi.org/10.1016/j.agwat.2012.11.007
Graf F, Frei M (2013) Soil aggregate stability related to soil density, root length, and mycorrhiza using site-specific Alnus incana and Melanogaster variegatus s.l. Ecol Eng 57:314–323. https://doi.org/10.1016/j.ecoleng.2013.04.037
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Calif Agric Exp Stn Circ 347:1–32
Jansa J, Mozafar A, Frossard E (2003) Long-distance transport of P and Zn through the hyphae of an arbuscular mycorrhizal fungus in symbiosis with maize. Agronomie 23:481–488. https://doi.org/10.1051/agro:2003013
Karandashov V, Bucher M (2005) Symbiotic phosphate transport in arbuscular mycorrhizas. Trends Plant Sci 10:22–29. https://doi.org/10.1016/j.tplants.2004.12.003
Khalvati MA, Hu Y, Mozafar A, Schmidhalter U (2005) Quantification of water uptake by arbuscular mycorrhizal hyphae and its significance for leaf growth, water relations, and gas exchange of barley subjected to drought stress. Plant Biol 7:706–712. https://doi.org/10.1055/s-2005-872893
Klironomos JN (2003) Variation in plant response to native and exotic arbuscular mycorrhizal fungi. Ecology 84:2292–2301
Klute A, Weaver RW, Mickelson SH et al (1994) Methods of soil analysis: Part 4, Physical Methods. Soil Science Society of America, Madison
Lam CC, Leung YK (1995) Extreme rainfall statistics and design rainstorm profiles at selected locations in Hong Kong. Technical Note No. 86
Leifheit EF, Veresoglou SD, Lehmann A, Morris EK, Rillig MC (2013) Multiple factors influence the role of arbuscular mycorrhizal fungi in soil aggregation—a meta-analysis. Plant Soil 347:523–537. https://doi.org/10.1007/s11104-013-1899-2
Leung AK, Ng CWW (2013) Analyses of groundwater flow and plant evapotranspiration in a vegetated soil slope. Can Geotech J 50:1204–1218. https://doi.org/10.1139/cgj-2013-0148
Leung HM, Ye ZH, Wong MH (2007) Survival strategies of plants associated with arbuscular mycorrhizal fungi on toxic mine tailings. Chemosphere 66:905–915. https://doi.org/10.1016/j.chemosphere.2006.06.037
Leung FTY, Yan WM, Hau BCH, Tham LG (2015) Root systems of native shrubs and trees in Hong Kong and their effects on enhancing slope stability. CATENA 125:102–110. https://doi.org/10.1016/j.catena.2014.10.018
Ma JF, Tamai K, Ichii M, Wu GF (2002) A rice mutant defective in Si uptake. Plant Physiol 130:2111–2117. https://doi.org/10.1104/pp.010348
Marulanda A, Azcón R, Ruiz-Lozano JM (2003) Contribution of six arbuscular mycorrhizal fungal isolates to water uptake by Lactuca sativa plants under drought stress. Physiol Plant 119:526–533. https://doi.org/10.1046/j.1399-3054.2003.00196.x
Mayr R, Godoy R (1990) Seasonal patterns in vesicular-arbuscular mycorrhiza in Melic-Beech Forest. Agric Ecosyst Environ 29:281–288. https://doi.org/10.1016/0167-8809(90)90288-O
McGonigle TP, Miller MH, Evans DG et al (1990) A new method which gives an objective measure of colonization of roots by vesicular—arbuscular mycorrhizal fungi. New Phytol 115:495–501. https://doi.org/10.1111/j.1469-8137.1990.tb00476.x
Nadian H, Smith SE, Alston AM, Murray RS (1996) The effect of soil compaction on growth and P uptake by Trifolium subterraneum: interactions with mycorrhizal colonisation. Plant Soil 182:39–49. https://doi.org/10.1007/BF00010993
Nadian H, Smith SE, Alston AM et al (1998) Effects of soil compaction on phosphorus uptake and growth of Trifolium subterraneum colonized by four species of vesicular–arbuscular mycorrhizal fungi. New Phytol 140:155–165. https://doi.org/10.1046/j.1469-8137.1998.00219.x
Ng CWW, Leung AK, Woon KX (2013a) Effects of soil density on grass-induced suction distributions in compacted soil subjected to rainfall. Can Geotech J 51:311–321. https://doi.org/10.1139/cgj-2013-0221
Ng CWW, Woon KX, Leung AK, Chu LM (2013b) Experimental investigation of induced suction distribution in a grass-covered soil. Ecol Eng 52:219–223. https://doi.org/10.1016/j.ecoleng.2012.11.013
Paszkowski U, Kroken S, Roux C, Briggs SP (2002) Rice phosphate transporters include an evolutionarily divergent gene specifically activated in arbuscular mycorrhizal symbiosis. Proc Natl Acad Sci U S A 99:13324–13329. https://doi.org/10.1073/pnas.202474599
Peng S, Guo T, Liu G (2013) The effects of arbuscular mycorrhizal hyphal networks on soil aggregations of purple soil in southwest China. Soil Biol Biochem 57:411–417. https://doi.org/10.1016/j.soilbio.2012.10.026
Pollen N, Simon A (2005) Estimating the mechanical effects of riparian vegetation on stream bank stability using a fiber bundle model. Water Resour Res 41:W07025. https://doi.org/10.1029/2004WR003801
Pollen-Bankhead N, Simon A (2010) Hydrologic and hydraulic effects of riparian root networks on streambank stability: is mechanical root-reinforcement the whole story. Geomorphology 116:353–362. https://doi.org/10.1016/j.geomorph.2009.11.013
Pozo MJ, Jung SC, López-Ráez JA, Azcón-Aguilar C (2010) Impact of arbuscular mycorrhizal symbiosis on plant response to biotic stress: the role of plant defence mechanisms. In: Koltai H, Kapulnik Y (eds) Arbuscular mycorrhizas: physiology and function. Springer, Netherlands, pp 193–207
Rahardjo H, Satyanaga A, Leong EC, Santoso VA, Ng YS (2014) Performance of an instrumented slope covered with shrubs and deep-rooted grass. Soils Found 54:417–425. https://doi.org/10.1016/j.sandf.2014.04.010
Redecker D, Schüßler A, Stockinger H, Stürmer SL, Morton JB, Walker C (2013) An evidence-based consensus for the classification of arbuscular mycorrhizal fungi (Glomeromycota). Mycorrhiza 23:515–531. https://doi.org/10.1007/s00572-013-0486-y
Rillig MC, Mummey DL (2006) Mycorrhizas and soil structure. New Phytol 171:41–53. https://doi.org/10.1111/j.1469-8137.2006.01750.x
Rillig MC, Wright SF, Eviner VT (2002) The role of arbuscular mycorrhizal fungi and glomalin in soil aggregation: comparing effects of five plant species. Plant Soil 238:325–333. https://doi.org/10.1023/A:1014483303813
Rillig MC, Ramsey PW, Morris S, Paul EA (2003) Glomalin, an arbuscular-mycorrhizal fungal soil protein, responds to land-use change. Plant Soil 253:293–299. https://doi.org/10.1023/A:1024807820579
Ruiz-Lozano JM (2003) Arbuscular mycorrhizal symbiosis and alleviation of osmotic stress. New perspectives for molecular studies. Mycorrhiza 13:309–317. https://doi.org/10.1007/s00572-003-0237-6
Ruiz-Lozano JM, Azcón R (1995) Hyphal contribution to water uptake in mycorrhizal plants as affected by the fungal species and water status. Physiol Plant 95:472–478. https://doi.org/10.1111/j.1399-3054.1995.tb00865.x
Ruth B, Khalvati M, Schmidhalter U (2011) Quantification of mycorrhizal water uptake via high-resolution on-line water content sensors. Plant Soil 342:459–468. https://doi.org/10.1007/s11104-010-0709-3
Schnepf A, Roose T, Schweiger P (2008) Impact of growth and uptake patterns of arbuscular mycorrhizal fungi on plant phosphorus uptake—a modelling study. Plant Soil 312:85–99. https://doi.org/10.1007/s11104-008-9749-3
Segal E, Ben-Gal A, Shani U (2006) Root water uptake efficiency under ultra-high irrigation frequency. Plant Soil 282:333–341. https://doi.org/10.1007/s11104-006-0003-6
Selig E, Ladd R (1978) Preparing test specimens using undercompaction. Geotech Test J 1:16. https://doi.org/10.1520/GTJ10364J
Smith SE, Read DJ (2008) Mycorrhizal symbiosis, 3rd edn. Academic Press, London
Stokes A, Atger C, Bengough AG, Fourcaud T, Sidle RC (2009) Desirable plant root traits for protecting natural and engineered slopes against landslides. Plant Soil 324:1–30. https://doi.org/10.1007/s11104-009-0159-y
Tisdall JM, Oades JM (1982) Organic matter and water-stable aggregates in soils. J Soil Sci 33:141–163. https://doi.org/10.1111/j.1365-2389.1982.tb01755.x
Wu TH (2013) Root reinforcement of soil: review of analytical models, test results, and applications to design. Can Geotech J 50:259–274. https://doi.org/10.1139/cgj-2012-0160
Wu TH, McKinnell WP III, Swanston DN (1979) Strength of tree roots and landslides on Prince of Wales Island, Alaska. Can Geotech J 16:19–33. https://doi.org/10.1139/t79-003
Funding
This study received financial support from the Collaborative Research Fund from the Research Grants Council, Hong Kong SAR (HKUST6/CRF/12R). This work was also supported by Guangdong Provincial Key Laboratory of Soil and Groundwater Pollution Control (No. 2017B030301012), State Environmental Protection Key Laboratory of Integrated Surface Water-Groundwater Pollution Control, and Department of Education of Guangdong Province (No. 2020KCXTD006).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Ravi Naidu
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• We investigated the effects of a mycorrhizal grass on soil matric suction.
• Mycorrhizal association did not exert significant effects on the matric suction.
• Root biomass would be more essential than shoot biomass in retaining suction.
• Higher proportions of 2–5-mm wet-aggregates observed in the mycorrhizal treatment.
• Bermuda grass associated with mycorrhizae is a potential method for erosion control.
Supplementary Information
ESM 1
(DOCX 25 kb)
Rights and permissions
About this article
Cite this article
Chen, X.W., Wong, J.T.F., Wang, JJ. et al. Effects of mycorrhizal Bermuda grass on low-range soil matric suction. J Soils Sediments 21, 990–1000 (2021). https://doi.org/10.1007/s11368-020-02839-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-020-02839-1