Abstract
Early ventricular fibrillation (EVF) predicts mortality in ST-segment elevation myocardial infarction (STEMI) patients. Data are lacking about prognosis and management of non-ST-segment elevation myocardial infarction (NSTEMI) EMI with EVF, especially at higher age. In the daily clinical practice, there is no clear prognosis of patients surviving EVF. The present study aimed to investigate the risk factors and factors influencing the prognosis of NSTEMI patients surviving EVF, especially at higher age. Clinical data, including 30-day and 1-year mortality of 6179 NSTEMI patients, were examined; 2.44% (n=151) survived EVF and were further analyzed using chi-square test and uni- and multivariate analyses. Patients were divided into two age groups below and above the age of 70 years. Survival time was compared with Kaplan-Meier analysis. EVF was an independent risk factor for mortality in NSTEMI patients below (HR: 2.4) and above the age of 70 (HR: 2.1). Mortality rates between the two age groups of NSTEMI patients with EVF did not differ significantly: 30-day mortality was 24% vs 40% (p=0.2709) and 1-year mortality was 39% vs 55% (p=0.2085). Additional mortality after 30 days to 1 year was 15% vs 14.6% (p=0.9728). Clinical characteristics of patients with EVF differed significantly from those without in both age groups. EVF after revascularization—within 48 h—had 11.2 OR for 30-day mortality above the age of 70. EVF in NSTEMI was an independent risk factor for mortality in both age groups. Invasive management and revascularization of NSTEMI patients with EVF is highly recommended. Closer follow-up and selection of patients (independent of age) for ICD implantation in the critical first month is essential.
Similar content being viewed by others
Introduction
Ventricular arrhythmias are potentially lethal complications of acute coronary syndromes. According to the VALLIANT Trial, the risk of sudden cardiac death is the highest within 1 month after the infarction [1]. Early primary ventricular fibrillation (EVF) occurs within 48–72 h after the symptoms’ onset and it is independent of the reoccurring ischemia and heart failure. In daily clinical practice, the prognosis of patients surviving EVF is not clear.
However, the GISSI-2 Trial showed the relevance of EVF as an independent predictor for in-hospital mortality [2]. Results regarding the risk factors and the effect of EVF on the short- and long-term prognoses in ST-segment elevation myocardial infarction (STEMI) patients have been controversial. EVF is a predictor for both 30-day and 1-year mortality rates in STEMI patients treated with primary percutaneous coronary intervention [3]. In contrast, another study suggested that EVF was associated with higher in-hospital mortality but did not affect the long-term prognosis [4]. Other earlier studies also found non-significant impact of EVF on prognosis [5], and recent studies did not confirm these results [6]. Medina-Rodriguez et al. found that EVF before intensive care unit admission was an independent predictor of in-hospital mortality in a cohort of patients in whom fibrinolysis was the main method of revascularization therapy [7]. A similar prognostic impact in patients treated with percutaneous coronary intervention (PCI) was not present in that study, suggesting that PCI has a long-term therapeutic benefit in EVF patients. In a large unselected population of STEMI patients treated with PPCI, ventricular fibrillation during the first 48 h after STEMI was associated with increased in-hospital mortality but no influence on the long-term prognosis for surviving patients was established [8]. In contrast, Kosmidou et al. have reported that ventricular arrhythmias occurring before coronary angiography and revascularization in patients with STEMI were strongly associated with an increased 3-year likelihood of death and stent thrombosis [6].
Jabbari et al. investigated the independent risk factors that contribute to the occurrence of ventricular fibrillation (VF) before PPCI in STEMI patients. They found that traditional coronary artery disease (CAD) risk factors such as diabetes, hypertension, and hypercholesterinemia did not predict risk while higher age, family history of sudden cardiac death, use of statins, and higher alcohol intake were independent risk factors [9]. The extent of the CAD also contributed to higher mortality. Larger studies found that EVF is associated with the final infarct size [10, 11]. However, the findings of Gheeraer et al. contradicted these results, reporting that the region at risk and the site of the occlusion are not independent risk factors for out-of-hospital VF [12]. Literature data suggests that in STEMI, EVF is an independent risk factor for in-hospital and short-term mortality, and its effect on long-term mortality is unclear.
Many previous studies have investigated the risk factors contributing to EVF and the prognosis of EVF in unselected myocardial infarction population. A high proportion of acute coronary syndrome cases is NSTEMI. In NSTEMI patients, the prognosis of EVF and factors influencing the prognosis are less clarified. NSTEMI patients are more likely to exhibit complicated cases, have more comorbidities, and have higher mortality rates in general. In addition, mean age in NSTEMI is higher, and as a result of all these factors, it is essential to evaluate this patient population. Since NSTEMI prevalence increases with age, the importance of evaluating this patient population is particularly well justified. Current guidelines have few instructions about the management of NSTEMI patients with EVF, largely due to a lack of study evaluation in this population, especially in patients at higher age.
To help combat this lack of data about the prognosis and management of NSTEMI patients surviving EVF, the present study using our large database (~ 12,000 patients) has been undertaken. Our goal was to investigate the risk factors contributing to early VF in the elderly and identify factors influencing the prognosis of NSTEMI subjects using a retrospective study design.
Methods
Study population and data collection
A total of 11,582 patients with acute coronary syndrome have been revascularized between 2005 and 2013 at our institution. These consecutive patients were enrolled in the Városmajor Myocardial Infarction Registry (VMAJOR-MI Registry), in which all the available demographic data and clinical data are summarized. Demographic data include gender, date of birth, date of admission, and date of death. Clinical patient data include laboratory findings (troponin T, CK-MB, creatinine, glucose, cholesterol, LDL-cholesterol), type of infarction (STEMI, NSTEMI), results from echocardiography, left ventricle ejection fraction (LV-EF), and coronary angiography. The initiating acute event has been characterized by the following factors: complicated by EVF, cardiogenic shock, on-site resuscitation, heart failure, invasive respiratory treatment. Data from EVF-positive patients was supplemented by information on laboratory parameters such as potassium levels, white blood cell (WBC) count, and C-reactive protein (CRP) levels, as well as by information on detailed coronary status such as the number of vessels affected, and the number of vessels treated by PCI.
From this detailed VMAJOR-Registry, we enrolled only patients having NSTEMI. Patients with ST-segment elevation myocardial infarction were excluded from our analysis. We divided patients into two groups based on whether or not their myocardial infarction led to EVF. Patients were further grouped based on age (above or below the age of 70 years). Figure 1 describes the enrolment and grouping process in detail.
Diagnosis of NSTEMI was made based on typical symptoms such as chest discomfort, upper extremity discomfort, dyspnea, fatigue, and the elevation of necrosis markers. EVF was defined as ventricular fibrillation requiring defibrillation in the first 48 h after AMI. Patients in the EVF group included those who suffered VF before or after revascularization as long as it was within 48 h. Other types of ventricular arrhythmias, such as ventricular tachycardia, were not examined.
Among NSTEMI patients, only patients undergoing coronary angiography, defined as percutaneous coronary intervention (PCI) in the first 12 h of symptom onset, were included in the study [13]. Patients undergoing coronary artery bypass grafting surgery or who were managed conservatively were not eligible for the study. Coronary stenosis was evaluated from multiplane projections and a luminal diameter reduction of >50% was considered significant. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki and was approved in advance by the locally appointed ethics committee (30088-2/2014/EKU). The primary outcome of the study was all-cause mortality. The National Health Care Institute provided accurate details on the above endpoint with occurrence dates.
Statistical methods
Analysis was performed using Statistica 13.2 software and MedCalc statistical software. Continuous variables were expressed as mean and standard deviation (mean ± St. D); categorical variables were summarized as the sample size (n) and frequencies. Student’s t-test was used for comparison of normally distributed data and Mann-Whitney U-test with non-normal distribution. Categorical variables were compared using chi-square test.
Cox proportional hazard model was used to identify whether EVF was an independent risk factor for mortality. The model included the general risk factors such as age, gender, diabetes mellitus, left ventricle function, severity of acute event—heart failure, cardiogenic shock, invasive respiratory treatment—and EVF.
Cox regression analysis was performed in order to identify clinical characteristics associated with mortality. The model included all available risk factors of acute coronary syndromes and ventricular fibrillation such as gender, age, body mass index (BMI), diabetes mellitus, LV-EF, complications of the acute event (on-site CPR, cardiogenic shock, heart failure), coronary angiographic results (coronary status—vessels affected, PCI results—stent implantation on how many vessels), and the time of the VF (before, during, or after the revascularization but within 48 h). Hazard ratios (HR) with corresponding 95% confidence intervals (CI) were calculated using Cox proportional hazard model. Survival time of the different patient groups was compared using Kaplan-Meier survival analysis. All statistical analysis was two-tailed; the level of significance was p<0.05.
Results
The clinical characteristics of the 3140 NSTEMI patients below the age of 70 are presented in Table 1. Significant differences were found between EVF-positive and EVF-negative cases. EVF-positive NSTEMI patients were more likely to have poor left ventricle function (LV-EF <40%) (39.22% vs 14.95%), larger infarct size characterized by higher troponin (2073.9 ng/L vs 902.3 ng/L), and higher CK-MB (158.7 U/L vs 74.8 U/L), and were more likely to have diabetes (55.56% vs 30.34%). They suffered more severe infarction with more complications such as cardiogenic shock (18.18% vs 2.15%) and increased need for invasive respiratory treatment (43.94% vs 5.6%). Given these significant differences, it is not surprising that EVF-positive patients also had higher mortality rates than control patients. 30-day mortality was 24% vs 4.6% and 1-year mortality was 39% vs 10.6% in EVF vs. non-EVF patients <70, respectively. However, additional mortality (mortality between 30 days and 1 year) did not differ significantly.
Table 2 shows the differences between EVF and non-EVF groups in patients above the age of 70 years. In the older age group, similarly to the younger patient group, subjects surviving EVF were more likely to have reduced left ventricle ejection fraction (44.4% vs 22.8%) and diabetes (53% vs 37%). They also had more severe complications after the acute event including cardiogenic shock (18% vs 4%) and the need for invasive respiratory treatment (56% vs 9%). As seen in the <70 patient group, the patients aged >70 also exhibited increased 30-day and 1-year mortality in the EVF group vs non-EVF group (40% vs 10% for 30 days and 55% vs 28% for 1 year).
When we compared mortality rates for NSTEMI patients surviving EVF in patients below 70 years vs above 70 years, no significant difference has been found in 30-day mortality (24% vs 40% p=0.2709), in 1-year mortality (39% vs 55% p=0.2085), or in mortality between 30 days and 1 year (15% vs 14.6% p=0.9728).
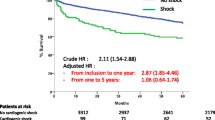
EVF patients at younger (Fig. 2), as well as at older age, >70 years (Fig. 3), had significantly (p<0.0001) lower survival probability compared to non-EVF ones. Figure 4 shows survival probability in the 4 patient groups (based on age and EVF).
Cox regression analysis showed that in patients <70, EVF is an independent risk factor for all mortality (HR: 2.38) (Table 3), in addition to other factors such as diabetes mellitus (HR: 2.02), heart failure (HR: 3.66), cardiogenic shock (HR: 8.99), and invasive respiratory treatment (HR: 5.4). Similarly, in patients above the age of 70 years, EVF is also an independent risk factor for mortality (HR: 2.1) as well as diabetes mellitus (HR: 1.5), heart failure (HR: 2.4), cardiogenic shock (HR: 4.85), and invasive respiratory treatment (HR: 3.2) (Table 4).
Seeing that EVF is an independent risk for mortality in NSTEMI in both age groups, we furtherly evaluated the risk factors for mortality in the NSTEMI with EVF patient group. Factors influencing mortality in NSTEMI patients surviving EVF are presented in Table 5. In patients below the age of 70 years, these factors include diabetes mellitus (HR: 1.9), cardiogenic shock (HR: 6.1), heart failure (HR: 2.65), and CPR (HR: 2.5). However, at higher age (above 70 years), the only factor influencing mortality was cardiogenic shock (HR: 2.3). The extent of the coronary artery disease did not affect mortality in either age group.
To help understand whether the timing of EVF, with respect to the timing of the coronary revascularization, had an effect on prognosis, we asked whether mortality was different in patients who experienced EVF either before or after PCI. In most cases in the <70 age group, EVF developed before revascularization (75%, 51/68), but the timing had no influence on either the short- or long-term mortality (Table 6). In the higher age group (>70 years), most EVF also developed before revascularization (74%, 61/82). In contrast to the younger group, in patients >70 years, EVF that occurs after revascularization was associated with a higher risk of 30-day mortality (OR 11.2), although 1-year mortality was not significantly different (Table 7).
Discussion
According to the literature, the incidence of ventricular arrhythmias including EVF in the acute phase of MI is approximately 2–8% [7, 14], which is consistent with data in our patient group resented here (2.4%). Despite the fact that the incidence of ventricular arrhythmias is higher in STEMI than in NSTEMI (10% vs 2.1%) respectively [15], mortality rates in the EVF patient group are significantly higher regardless of the infarction type versus non-EVF patients. However, the FAST-MI program discovered that 6-month mortality has decreased over the past 20 years [16]. Since 2010, mortality in STEMI patients has continued to decline; however, mortality in NSTEMI patients has remained stable [16], highlighting the need for further investigation into factors that affect NSTEMI mortality. In a small group of invasively treated NSTEMI patients, Gupta et al. investigated the incidence of and predictors for malignant arrhythmias [17]. In their population, VF occurred in 7.6% of the patients, a much higher fraction than in our study in which 2.4% (151/6179) experienced EVF. Similar to our study, they also reported that 30-day mortality was significantly higher in patients with vs. without VF (38% vs 3%), and their 30-day mortality rate among EVF patients was comparable to what we observed (33%). However, their EVF-negative patients had higher 30-day mortality compared to our results despite using invasive therapy. Similarly, Al-Khatib et al. also reported increased 30-day and 6-month mortality in spite of using effective therapy [15]. They found that in-hospital VF and VT were independently associated with 30-day and 6-month mortality even after excluding patients with heart failure and cardiogenic shock and those who died within 24 h [15]. The MERLIN-TIMI 36 Trial also highlighted the significance of non-sustained VT in NSTEMI. Although non-sustained VT is common after NSTEMI, short episodes of VT are independently associated with a higher risk of sudden cardiac death [18].
In the intervention era (2000–2012), the number of patients who receive coronary angiography and PCI after VT/VF has increased, resulting in a higher survival rate—survival in all acute myocardial infarction has risen from 46.9 to 60.1%, in STEMI survival has risen from 59.2 to 74.3%, and in NSTEMI survival has risen from 43.3 to 56.8% [19]. In spite of clear evidence showing that coronary angiography and PCI increase survival, in daily practice, some proportion of patients, mostly NSTEMI, do not undergo revascularization. At our high-volume cardiology institute with an invasive approach, STEMI and NSTEMI patients are treated invasively. Yet, in spite of this invasive strategy, here, we report that NSTEMI patients surviving EVF still have higher short- and long-term mortality rates compared to those without EVF regardless of whether they are above or below 70 years of age. Kaplan-Meier analysis (Fig. 4) showed that in the first 3 years after the acute myocardial infarction, younger patients with EVF had worse survival probability than those older ones without EVF. However, past the 3 years’ mark, age becomes more important regards life expectancy. This finding supports the fact that age is one of the main determining factors of survival. Besides the clinical fact that older patients are more likely to have numerous comorbidities, cellular and molecular mechanisms may contribute to increased mortality in older adults, e.g., impaired cellular stress and age-related oxidative stress. Studies confirm this idea that hypoxia, oxidative stress, worsens the prognosis of cardiovascular patients. Trimetazidine was found to be a cytoprotective agent [20] which improves the quality of life and left ventricle function in elderly patients with ischemic heart disease [21, 22]. Importantly, our analysis demonstrated that in addition to other well-characterized comorbidities, EVF is also an independent risk factor for mortality in both age groups. This is consistent with other studies showing that patients with EVF (compared to non-EVF patients) have more frequently reduced LV-EF and triple-vessel coronary artery disease (CAD) resulting in higher 30-day mortality [23]. Interestingly, the prognosis had no correlation with the extent of the coronary artery disease either at lower or at higher age in NSTEMI patients surviving EVF. In NSTEMI patients with EVF at younger age, several factors had an influence on mortality, such as LV-EF, diabetes mellitus, cardiogenic shock, heart failure, and on-site resuscitation. In contrast to that at higher age, the only factor was a cardiogenic shock.
One interesting finding of our study was the timing of EVF (with respect to the timing of intervention) impacted the prognosis in older but no younger NSTEMI patients. The impact of timing on outcomes has also been examined by others. For example, according to Jabbari et al., there is no difference in the 30-day mortality in STEMI patients depending on VF before or during PCI [24].
In general, NSTEMI patients have worse prognosis than STEMI patients, and cases that are complicated with EVF have even poorer outcomes. The mortality risk was the highest within the first 30 days; we found that 40% of the aged patients died within the first month. There are only a few differences in clinical factors influencing who will develop EVF in the two age groups. The fact that EVF develops based on acute myocardial ischemia is not surprising. Our results suggest that this pathomechanism is the same at higher age. Invasive management of NSTEMI patients is essential, and even with invasive management, EVF was an independent risk factor for mortality. These findings suggest that closer follow-up—using telemedicine in the aged patient population with decreased mobility—in the critical first 30 days is essential. It is important to select patients, independently from age, who would benefit from an early implantable cardioverter-defibrillator implantation before discharge. With more outpatient visits, more precise medication setup for secondary prevention would be beneficial independently from age.
Limitation section
The present study was a single-center retrospective observational study with limited available data. Factors affecting the prognosis such as ICD implantations, medication intake, and compliance could not be investigated.
References
Solomon SD, Zelenkofske S, McMurray JJV, Finn PV, Velazquez E, Ertl G, et al. Sudden death in patients with myocardial infarction and left ventricular dysfunction, heart failure, or both. N Engl J Med. 2005;352(25):2581–8.
Volpi A, Cavalli A, Santoro L, Negri E. Incidence and prognosis of early primary ventricular fibrillation in acute myocardial infarction--results of the Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico (GISSI-2) database. Am J Cardiol. 1998;82(3):265–71.
García-García C, Oliveras T, Rueda F, Pérez-Fernández S, Ferrer M, Serra J, et al. Primary ventricular fibrillation in the primary percutaneous coronary intervention ST-segment elevation myocardial infarction era (from the “Codi IAM” Multicenter Registry). Am J Cardiol. 2018;122(4):529–36.
Demirel F, Rasoul S, Elvan A, Ottervanger JP, Dambrink JHE, Gosselink ATM, et al. Impact of out-of-hospital cardiac arrest due to ventricular fibrillation in patients with ST-elevation myocardial infarction admitted for primary percutaneous coronary intervention: Impact of ventricular fibrillation in STEMI patients. Eur Heart J Acute Cardiovasc Care. 2015;4(1):16–23.
O’Doherty M, et al. Five hundred patients with myocardial infarction monitored within one hour of symptoms. Br Med J (Clin Res Ed). 1983;286(6375):1405–8.
Kosmidou I, Embacher M, McAndrew T, Dizon JM, Mehran R, Ben-Yehuda O, et al. early ventricular tachycardia or fibrillation in patients with ST elevation myocardial infarction undergoing primary percutaneous coronary intervention and impact on mortality and stent thrombosis (from the Harmonizing Outcomes with Revascularization and Stents in Acute Myocardial Infarction Trial). Am J Cardiol. 2017;120(10):1755–60.
Medina-Rodríguez KE, Almendro-Delia M, García-Alcántara Á, Arias-Garrido JJ, Rodríguez-Yáñez JC, Alonso-Muñoz G, et al. Prognostic implication of early ventricular fibrillation among patients with ST elevation myocardial infarction. Coron Artery Dis. 2017;28(7):570–6.
Demidova MM, Smith JG, Höijer CJ, Holmqvist F, Erlinge D, Platonov PG. Prognostic impact of early ventricular fibrillation in patients with ST-elevation myocardial infarction treated with primary PCI. Eur Heart J Acute Cardiovasc Care. 2012;1(4):302–11.
Jabbari R, Engstrøm T, Glinge C, Risgaard B, Jabbari J, Winkel BG, et al. Incidence and risk factors of ventricular fibrillation before primary angioplasty in patients with first ST-elevation myocardial infarction: a nationwide study in Denmark. J Am Heart Assoc. 2015;4(1):e001399.
Tofler GH, Stone PH, Muller JE, Rutherford JD, Willich SN, Gustafson NF, et al. Prognosis after cardiac arrest due to ventricular tachycardia or ventricular fibrillation associated with acute myocardial infarction (the MILIS Study). Multicenter Investigation of the Limitation of Infarct Size. Am J Cardiol. 1987;60(10):755–61.
Behar S, Goldbourt U, Reicher-Reiss H, Kaplinsky E, The Principal Investigators of the SPRINT Study. Prognosis of acute myocardial infarction complicated by primary ventricular fibrillation. Principal Investigators of the SPRINT Study. Am J Cardiol. 1990;66(17):1208–11.
Gheeraert PJ, Henriques JPS, de Buyzere ML, Voet J, Calle P, Taeymans Y, et al. Out-of-hospital ventricular fibrillation in patients with acute myocardial infarction: coronary angiographic determinants. J Am Coll Cardiol. 2000;35(1):144–50.
Neumann FJ, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, Benedetto U, et al. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40(2):87–165.
Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, Bueno H, et al. 2017 ESC guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: the Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39(2):119–77.
Al-Khatib SM, et al. Sustained ventricular arrhythmias among patients with acute coronary syndromes with no ST-segment elevation: incidence, predictors, and outcomes. Circulation. 2002;106(3):309–12.
Puymirat E, Simon T, Cayla G, Cottin Y, Elbaz M, Coste P, et al. Acute myocardial infarction: changes in patient characteristics, management, and 6-month outcomes over a period of 20 years in the FAST-MI program (French Registry of Acute ST-Elevation or Non-ST-Elevation Myocardial Infarction) 1995 to 2015. Circulation. 2017;136(20):1908–19.
Gupta S, Pressman GS, Figueredo VM. Incidence of, predictors for, and mortality associated with malignant ventricular arrhythmias in non-ST elevation myocardial infarction patients. Coron Artery Dis. 2010;21(8):460–5.
Scirica BM, Braunwald E, Belardinelli L, Hedgepeth CM, Spinar J, Wang W, et al. Relationship between nonsustained ventricular tachycardia after non-ST-elevation acute coronary syndrome and sudden cardiac death: observations from the metabolic efficiency with ranolazine for less ischemia in non-ST-elevation acute coronary syndrome-thrombolysis in myocardial infarction 36 (MERLIN-TIMI 36) randomized controlled trial. Circulation. 2010;122(5):455–62.
Patel N, Patel NJ, Macon CJ, Thakkar B, Desai M, Rengifo-Moreno P, et al. Trends and outcomes of coronary angiography and percutaneous coronary intervention after out-of-hospital cardiac arrest associated with ventricular fibrillation or pulseless ventricular tachycardia. JAMA Cardiol. 2016;1(8):890–9.
Stanley WC, Marzilli M. Metabolic therapy in the treatment of ischaemic heart disease: the pharmacology of trimetazidine. Fundam Clin Pharmacol. 2003;17(2):133–45.
Marazzi G, Gebara O, Vitale C, Caminiti G, Wajngarten M, Volterrani M, et al. Effect of trimetazidine on quality of life in elderly patients with ischemic dilated cardiomyopathy. Adv Ther. 2009;26(4):455–61.
Vitale C, Wajngaten M, Sposato B, Gebara O, Rossini P, Fini M, et al. Trimetazidine improves left ventricular function and quality of life in elderly patients with coronary artery disease. In: Eur Heart J. England, 2004;1814–21.
Piccini JP, White JA, Mehta RH, Lokhnygina Y, al-Khatib SM, Tricoci P, et al. Sustained ventricular tachycardia and ventricular fibrillation complicating non-ST-segment-elevation acute coronary syndromes. Circulation. 2012;126(1):41–9.
Jabbari R, Risgaard B, Fosbøl EL, Scheike T, Philbert BT, Winkel BG, et al. Factors associated with and outcomes after ventricular fibrillation before and during primary angioplasty in patients with ST-segment elevation myocardial infarction. Am J Cardiol. 2015;116(5):678–85.
Availability of data and material
Availability is needed.
Code availability
MedCalc Statistical Software version 19.4.1 (MedCalc Software Ltd, Ostend, Belgium; https://www.medcalc.org; 2020).
TIBCO Software Inc. (2018). Statistica (data analysis software system), version 13. http://tibco.com.
Funding
Open access funding provided by Semmelweis University. This study was supported by the National Research, Development and Innovation Office of Hungary (NKFIA; NVKP_16-1-2016-0017 National Heart Program). The research was financed by the Thematic Excellence Programme (Tématerületi Kiválósági Program, 2020-4.1.1.-TKP2020) of the Ministry for Innovation and Technology in Hungary, within the framework of the Therapeutic Development and Bioimaging programs of the Semmelweis University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki and was approved in advance by the locally appointed ethics committee (30088-2/2014/EKU).
Consent to participate and consent for publication
All authors are aware of the submission and agree to its publication. The submission is original; it has not been submitted before and is not under consideration for publication elsewhere.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Skoda, R., Bárczi, G., Vágó, H. et al. Prognosis of the non-ST elevation myocardial infarction complicated with early ventricular fibrillation at higher age. GeroScience 43, 2561–2571 (2021). https://doi.org/10.1007/s11357-021-00377-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-021-00377-3