Abstract
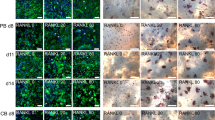
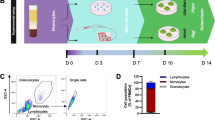
Bone loss occurs insidiously and initially asymptomatically; therefore, osteoporosis is frequently diagnosed only after the first clinical fracture. The aim of this study was to test the hypothesis is that by simply observing the behavior of cultured peripheral monocytes, it might be possible to diagnose altered bone remodeling and, therefore, limit the complications associated with osteoporosis, especially fractures. Monocytes isolated as mononuclear precursors from healthy and ovariectomized rats were cultured both in basal and differentiation medium for up to 3 weeks. Viability and differentiation capability towards the osteoclastic phenotype was checked by light microscopy at early times, whereas differentiation state and synthetic activity (tartrate-resistant acid phosphatase (TRAP) staining; phalloidin, fluorescin isothiocynate (FITC) staining, cathepsin K, metalloproteinase 7 and 9, MMP-7 and MMP-9) were measured at 1, 2, and 3 weeks. Compared to their controls, monocytes isolated from ovariectomized rats proliferate and lean toward the osteoclastic phenotype in the absence of differentiating factors. In both culture conditions, osteoclasts from ovariectomized rats showed significantly higher productions of cathepsin K, MMP-7, and MMP-9 than those of cells isolated from healthy rats, steadily over time. These results obtained in an animal osteoporotic model, if confirmed by clinical studies, open up the possibility to assess the presence of an alteration in bone remodeling with a simple in vitro diagnostic test requiring a small blood sample and less than 48 h. This might allow to early select patients with a spontaneous viability and differentiation of monocytes to osteoclasts for further diagnostic techniques.





Similar content being viewed by others
References
Boyle WJ, Simonet WS, Lacey DL (2003) Osteoclast differentiation and activation. Nature. 423(6937):337-42. doi:10.1038/nature01658
Chen FP, Wang KC, Huang JD (2009) Effect of estrogen on the activity and growth of human osteoclasts in vitro. Taiwan J Obstet Gynecol. 48(4):350-5. doi:10.1016/S10284559(09)60,323-5
Clover J, Dodds RA, Gowen M (1992) Integrin subunit expression by human osteoblasts and osteoclasts in situ and in culture. J Cell Sci. 103 (Pt 1):267-71.
D’Amelio P, Grimaldi A, Pescarmona GP, Tamone C, Roato I, Isaia G (2005) Spontaneous osteoclast formation from peripheral blood mononuclear cells in postmenopausal osteoporosis. FASEB J. 19(3):410-2. doi:10.1096/fj.04-2214fje. Epub 2004 Dec. 20.
Davidge ST, Zhang Y, Stewart KG (2001) A comparison of ovariectomy models for estrogen studies. Am J Physiol Regul Integr Comp Physiol. 280(3):R904-7.
Delaissé JM, Engsig MT, Everts V, del Carmen Ovejero M, Ferreras M, Lund L, Vu TH, Werb Z, Winding B, Lochter A, Karsdal MA, Troen T, Kirkegaard T, Lenhard T, Heegaard AM, Neff L, Baron R, Foged NT (2000) Proteinases in bone resorption: obvious and less obvious roles. Clin Chim Acta. 291(2):223-34. doi:10.1359/jbmr.2002.17.1.77
Eastell RHannon RA (2008) Biomarkers of bone health and osteoporosis risk. Proc Nutr Soc. 67(2):157-62. doi:10.1017/S002966510800699X
Everts V, Delaissé JM, Korper W, Jansen DC, Tigchelaar-Gutter W, Saftig P, Beertsen W (2002) The bone lining cell: its role in cleaning Howship’s lacunae and initiating bone formation. J Bone Miner Res. 17(1):77-90. doi:10.1359/jbmr.2002.17.1.77
Faust J, Lacey DL, Hunt P, Burgess TL, Scully S, Van G, Eli A, Qian Y, Shalhoub V (1999) Osteoclast markers accumulate on cells developing from human peripheral blood mononuclear precursors. J Cell Biochem. 72(1):67-80. doi:10.1002/(SICI)1097-4644(19,990,101)72:1<67::AID-JCB8>3.0.CO;2-A
Fuller K, Lawrence KM, Ross JL, Grabowska UB, Shiroo M, Samuelsson B, Chambers TJ (2008) Cathepsin K inhibitors prevent matrix-derived growth factor degradation by human osteoclasts. Bone. 42(1):200-11. doi:10.1016/j.bone.2007.09.044
Garnero P, Delmas PD (2004) Contribution of bone mineral density and bone turnover markers to the estimation of risk of osteoporotic fracture in postmenopausal women. J Musculoskelet Neuronal Interact. 4(1):50-63.
Garnero P (2014) New developments in biological markers of bone metabolism in osteoporosis. Bone. 66 46-55. doi:10.1016/j.bone.2014.05.016. Epub 2014 Jun. 5
Giavaresi G, De Terlizzi F, Gnudi S, Cadossi R, Aldini NN, Fini M, Rocca M, Ripamonti C, Brandi ML, Giardino R (2000) Discriminant capacity of quantitative ultrasound versus dual X-ray absorptiometry to determine cancellous bone loss in ovariectomized rats. Bone. 26(3):297-303. doi:10.1016/S8756-3282(99)00267-7
Helfrich MH, Thesingh CW, Mieremet RH, van Iperen-van Gent AS (1987) Osteoclast generation from human fetal bone marrow in cocultures with murine fetal long bones. A model for in vitro study of human osteoclast formation and function. Cell Tissue Res. 249(1):125-36.
Hentunen TA, Jackson SH, Chung H, Reddy SV, Lorenzo J, Choi SJ, Roodman GD (1999) Characterization of immortalized osteoclast precursors developed from mice transgenic for both bcl-X(L) and simian virus 40 large T antigen. Endocrinology. 140(7):2954-61. http://dx.doi.org/10.1210/endo.140.7.6867
Inaoka T, Bilbe G, Ishibashi O, Tezuka K, Kumegawa M, Kokubo T (1995) Molecular cloning of human cDNA for cathepsin K: novel cysteine proteinase predominantly expressed in bone. Biochem Biophys Res Commun. 206(1):89-96. doi:10.1006/bbrc.1995.1013
Jevon M, Hirayama T, Brown MA, Wass JA, Sabokbar A, Ostelere S, Athenasou NA (2003) Osteoclast formation from circulating precursors in osteoporosis. Scand J Rheumatol. 32(2):95-100.
Kanis JA, McCloskey EV, Johansson H, Cooper C, Rizzoli R, Reginster J-Y (2013) European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int. 24: 23-57. doi:10.1007/s00198-012-2074
Kiviranta R1, Morko J, Uusitalo H, Aro HT, Vuorio E, Rantakokko J (2001) Accelerated turnover of metaphyseal trabecular bone in mice overexpressing cathepsin K. J Bone Miner Res. 16(8):1444-52. doi:10.1359/jbmr.2001.16.8.1444
Le Gall C, Bellahcène A, Bonnelye E, Gasser JA, Castronovo V, Green J, Zimmermann J, Clézardin P (2007) A cathepsin K inhibitor reduces breast cancer induced osteolysis and skeletal tumor burden. Cancer Res. 67(20):9894-902. doi:10.1158/0008-5472.CAN-06-3940
Lewiecki EM, Cummings SR, Cosman F (2013) Treat-to-target for osteoporosis: is now the time? J Clin Endocrinol Metab. 98(3):946-53. doi:10.1210/jc.2012-3680. Epub 2013 Jan. 21.
Li YF, Zhou CC, Li JH, Luo E, Zhu SS, Feng G, Hu J (2012) The effects of combined human parathyroid hormone (1-34) and zoledronic acid treatment on fracture healing in osteoporotic rats. Osteoporos Int. 23(4):1463-74. doi:10.1007/s00198-011-1751-6. Epub 2011 Sep. 3.
Link TM (2012) Osteoporosis imaging: state of the art and advanced imaging. Radiology. 263(1):3-17 doi:10.1148/radiol.12110462
Littlewood-Evans AJ, Bilbe G, Bowler WB, Farley D, Wlodarski B, Kokubo T, Inaoka T, Sloane J, Evans DB, Gallagher JA (1997) The osteoclast-associated protease cathepsin K is expressed in human breast carcinoma. Cancer Res. 57(23):5386-90.
Massey HM, Flanagan AM (1999) Human osteoclasts derive from CD14-positive monocytes. Br J Haematol. 106(1):167-70. doi:10.1046/j.1365-2141.1999.01491.x
McCormick RK (2007) Osteoporosis: integrating biomarkers and other diagnostic correlates into the management of bone fragility. Altern Med Rev. 12(2):113-45.
Motyckova G, Fisher DE (2002) Pycnodysostosis: role and regulation of cathepsin K in osteoclast function and human disease. Curr Mol Med. 2(5):407-21. doi:10.2174/1,566,524,023,362,401
Pisani P, Renna MD, Conversano F, Casciaro E, Muratore M, Quarta E, Di Paola M, Casciaro S (2013) Screening and early diagnosis of osteoporosis through X-ray and ultrasound based techniques. World J Radiol. 5 (11): 398-410. doi:10.4329/wjr.v5.i11.398
Roodman GD (1999) Cell biology of the osteoclast. Exp Hematol. 27(8):1229-41. doi:10.1016/S0301-472X(99)00061-2
Saftig P, Hunziker E, Wehmeyer O, Jones S, Boyde A, Rommerskirch W, Moritz JD, Schu P, von Figura K (1998) Impaired osteoclastic bone resorption leads to osteopetrosis in cathepsin-K-deficient mice. Proc Natl Acad Sci U S A. 95(23):13,453-8.
Sakakura Y, Shide N, Tsuruga E, Irie K, Yajima T (2001) Effects of running exercise on the mandible and tibia of ovariectomized rats. J Bone Miner Metab. 19(3):159-67.
Schuit SC, van Meurs JB, Bergink AP, van der Klift M, Fang Y, Leusink G, Hofman A, van Leeuwen JP, Uitterlinden AG, Pols HA (2004) Height in pre- and postmenopausal women is influenced by estrogen receptor alpha gene polymorphisms. J Clin Endocrinol Metab. 89(1):303-9. http://dx.doi.org/10.1210/jc.2003-031095
Shalhoub V, Elliott G, Chiu L, Manoukian R, Kelley M, Hawkins N, Davy E, Shimamoto G, Beck J, Kaufman SA, Van G, Scully S, Qi M, Grisanti M, Dunstan C, Boyle WJ, Lacey DL (2000) Characterization of osteoclast precursors in human blood. Br J Haematol. 111(2):501-12. doi:10.1111/j.1365-2141.2000.02379.x
Shih C, Bernard GW (1996) Peripheral blood mononuclear cells develop into multinucleated osteoclasts in tissue culture. Anat Rec. 245(1):41-5.
Troen BR (2004) The role of cathepsin K in normal bone resorption. Drug News Perspect. 17(1):19-28.
U.S. Preventive Services Task Force (2011) Screening for osteoporosis: U.S. preventive services task force recommendation statement. Ann Intern Med. 154(5):356-64. doi:10.7326/0003-4819-154-5-201,103,010-00,307. Epub 2011 Jan. 17.
Wang FQ, So J, Reierstad S, Fishman DA (2005) Matrilysin (MMP-7) promotes invasion of ovarian cancer cells by activation of progelatinase. Int J Cancer. 114(1):19-31. doi:10.1002/ijc.20697.
Yasuda Y, Kaleta J, Brömme D (2005) The role of cathepsins in osteoporosis and arthritis: rationale for the design of new therapeutics. Adv Drug Deliv Rev. 57(7):973-93. doi:10.1016/j.addr.2004.12.013
Zhao H, Cai G, Du J, Xia Z, Wang L, Zhu T (1997) Expression of matrix metalloproteinase-9 mRNA in osteoporotic bone tissues. J Tongji Med Univ. 17(1):28-31.
Acknowledgments
This work was supported by Grants from Rizzoli Orthopedic Institute (Ricerca Corrente), “Cinque x mille 2010,” and to the Operational Programme ERDF 2007-2013 in the region Emilia-Romagna: Activity The 1.1 “Creation of technology centers for Industrial research and technological transfer.”
Conflict of interest
Melania Maglio, Gianluca Giavaresi, and Stefania Pagani declare that they have no competing interests. Francesca Salamanna, Roberto Giardino, and Milena Fini hold a patent on the described method.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Salamanna, F., Maglio, M., Giavaresi, G. et al. In vitro method for the screening and monitoring of estrogen-deficiency osteoporosis by targeting peripheral circulating monocytes. AGE 37, 82 (2015). https://doi.org/10.1007/s11357-015-9819-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11357-015-9819-4