Abstract
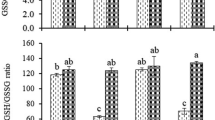
The foliar applied silicon (Si) has the potential to ameliorate heavy metals, especially cadmium (Cd) toxicity; however, Si dose optimization is strategically important for boosting the growth of soil microbes and Cd stress mitigation. Thus, the current research was performed to assess the Si-induced physiochemical and antioxidant trait alterations along with Vesicular Arbuscular Mycorrhiza (VAM) status in maize roots under Cd stress. The trial included foliar Si application at the rate of 0, 5, 10, 15, and 20 ppm while Cd stress (at the rate of 20 ppm) was induced after full germination of maize seed. The response variables included various physiochemical traits such as leaf pigments, protein, and sugar contents along with VAM alterations under induced Cd stress. The results revealed that exogenous application of Si in higher doses remained effective in improving the leaf pigments, proline, soluble sugar, total proteins, and all free amino acids. Additionally, the same treatment remained unmatched in terms of antioxidant activity compared to lower doses of foliar-applied Si. Moreover, VAM was recorded to be at peak under 20 ppm Si treatment. Thus, these encouraging findings may serve as a baseline to develop Si foliar application as a biologically viable mitigation strategy for maize grown in Cd toxicity soils. Overall, the exogenous application of Si helpful for reducing the uptake of Cd in maize and also improving the mycorrhizal association as well as the philological mechanism and antioxidant activities in plant under cadmium stress conditions. Also, future studies must test more doses concerning to varying Cd stress levels along with determining the most responsive crop stage for Si foliar application.













Similar content being viewed by others
Data availability
The raw data are available and will be provided on demand.
References
Abbasi A, Sufyan M, Arif MJ, Sahi ST (2020) Effect of silicon on tritrophic interaction of cotton, Gossypium hirsutum (Linnaeus), Bemisia tabaci (Gennadius) (Homoptera: Aleyrodidae) and the predator, Chrysoperla carnea (Stephens) (Neuroptera: Chrysopidae). Arthropod Plant Interact 14:717–725. https://doi.org/10.1007/s11829-020-09786-1
Abd El-Mageed TA, Shaaban A, Abd El-Mageed SA et al (2021) Silicon defensive role in maize (Zea mays L.) against drought stress and metals-contaminated irrigation water. Silicon 13:2165–2176. https://doi.org/10.1007/s12633-020-00690-0
Ahmad Z, Barutçular C, Zia Ur Rehman M et al (2022) Pod shattering in canola reduced by mitigating drought stress through silicon application and molecular approaches-a review. J Plant Nutr 46:101–128
Ahmad Z, Waraich EA, Akhtar S et al (2018) Physiological responses of wheat to drought stress and its mitigation approaches. Acta Physiol Plant 40:80. https://doi.org/10.1007/s11738-018-2651-6
Ahmad Z, Waraich EA, Iqbal MA et al (2021) Foliage applied silicon ameliorates drought stress through physio-morphological traits, osmoprotectants and antioxidant metabolism of camelina (Camelina sativa L.) genotypes. Acta Sci Pol Hortorum Cultus 20:43–57. https://doi.org/10.24326/ASPHC.2021.4.4
Ahmed S, Iqbal M, Ahmad Z et al (2023) Foliar application of silicon-based nanoparticles improve the adaptability of maize (Zea mays L.) in cadmium contaminated soils. Environ Sci Pollut Res 30:41002–41013. https://doi.org/10.1007/s11356-023-25189-0
Alayafi AH, Al-Solaimani SGM, Abd El-Wahed MH et al (2022) Silicon supplementation enhances productivity, water use efficiency and salinity tolerance in maize. Front Plant Sci 13:1–12. https://doi.org/10.3389/fpls.2022.953451
Alhousari F, Greger M (2018) Silicon and mechanisms of plant resistance to insect pests. Plants 7:1–11. https://doi.org/10.3390/plants7020033
Anwar S, Khan S, Ashraf MY et al (2017) Impact of chelator-induced phytoextraction of cadmium on yield and ionic uptake of maize. Int J Phytoremediation 19:505–513. https://doi.org/10.1080/15226514.2016.1254153
Aqeel M, Khalid N, Tufail A et al (2021) Elucidating the distinct interactive impact of cadmium and nickel on growth, photosynthesis, metal-homeostasis, and yield responses of mung bean (Vigna radiata L.) varieties. Environ Sci Pollut Res 28:27376–27390. https://doi.org/10.1007/s11356-021-12579-5
Arnon DI (1949) Copper enzymes in isolated chloroplasts. polyphenoloxidase in beta vulgaris. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1.1
Begum N, Qin C, Ahanger MA et al (2019) Role of arbuscular mycorrhizal fungi in plant growth regulation: implications in abiotic stress tolerance. Front Plant Sci 10:1–15. https://doi.org/10.3389/fpls.2019.01068
Bharwana SA, Ali S, Farooq MA et al (2013) Alleviation of lead toxicity by silicon is related to elevated photosynthesis, antioxidant enzymes suppressed lead uptake and oxidative stress in cotton. J. Bioremed. Biodeg 4(4):10–4172. https://doi.org/10.4172/2155-6199.1000187
Bhat JA, Shivaraj SM, Singh P et al (2019) Role of silicon in mitigation of heavy metal stresses in crop plants. Plants 8:1–20. https://doi.org/10.3390/plants8030071
Cakmak I, Hengeler C, Marschner H (1994) Changes in phloem export of sucrose in leaves in response to phosphorus, potassium and magnesium deficiency in bean plants. J Exp Bot 45:1251–1257. https://doi.org/10.1093/jxb/45.9.1251
Chance B, Maehly AC (1955) [136] Assay of catalases and peroxidases. {black small square}. Methods Enzymol 2:764–775. https://doi.org/10.1016/S0076-6879(55)02300-8
Currie HA, Perry CC (2007) Silica in plants: biological, biochemical and chemical studies. Ann Bot 100:1383–1389. https://doi.org/10.1093/aob/mcm247
Emamverdian A, Ding Y, Xie Y, Sangari S (2018) Silicon mechanisms to ameliorate heavy metal stress in plants. Biomed Res Int 2018. https://doi.org/10.1155/2018/8492898
Farooq H, Asghar HN, Khan MY et al (2015) Auxin-mediated growth of rice in cadmium-contaminated soil. Turkish J Agric for 39:272–276. https://doi.org/10.3906/tar-1405-54
Farooq MA, Saqib ZA, Akhtar J (2015) Silicon-mediated oxidative stress tolerance and genetic variability in rice (Oryza sativa L.) grown under combined stress of salinity and boron toxicity. Turkish J Agric for 39:718–729. https://doi.org/10.3906/tar-1410-26
Ferrol N, Tamayo E, Vargas P (2016) The heavy metal paradox in arbuscular mycorrhizas: from mechanisms to biotechnological applications. J Exp Bot 67:6253–6565. https://doi.org/10.1093/jxb/erw403
Giovannetti M, Mosse B (1966) An evaluation of techniques for measuring air-fuel ratio. New Phytol 84:489–500. https://doi.org/10.4271/660118
Gomes MP, Marques TCLLSEM, Soares AM (2013) Cadmium effects on mineral nutrition of the Cd-hyperaccumulator Pfaffia glomerata. Biol 68:223–230. https://doi.org/10.2478/s11756-013-0005-9
Greger M, Kabir AH, Landberg T et al (2016) Silicate reduces cadmium uptake into cells of wheat. Environ Pollut 211:90–97. https://doi.org/10.1016/j.envpol.2015.12.027
Hildebrandt U, Regvar M, Bothe H (2007) Arbuscular mycorrhiza and heavy metal tolerance. Phytochemistry 68:139–146. https://doi.org/10.1016/j.phytochem.2006.09.023
Howladar SM, Al-Robai SA, Al-Zahrani FS et al (2018) Silicon and its application method effects on modulation of cadmium stress responses in Triticum aestivum (L.) through improving the antioxidative defense system and polyamine gene expression. Ecotoxicol Environ Saf 159:143–152. https://doi.org/10.1016/j.ecoenv.2018.05.004
Hussain I, Ashraf MA, Rasheed R et al (2015) Exogenous application of silicon at the boot stage decreases accumulation of cadmium in wheat (Triticum aestivum L.) grains. Rev Bras Bot 38:223–234. https://doi.org/10.1007/s40415-014-0126-6
Ismael MA, Elyamine AM, Moussa MG et al (2019) Cadmium in plants: uptake, toxicity, and its interactions with selenium fertilizers. Metallomics 11:255–277. https://doi.org/10.1039/c8mt00247a
Jaishankar M, Tseten T, Anbalagan N et al (2014) Toxicity, mechanism and health effects of some heavy metals. Interdiscip Toxicol 7:60–72. https://doi.org/10.2478/intox-2014-0009
Joner EJ, Briones R, Leyval C (2000) Metal-binding capacity of arbuscular mycorrhizal mycelium. Plant Soil 226:227–234. https://doi.org/10.1023/A:1026565701391
Keller C, Rizwan M, Davidian JC et al (2015) Effect of silicon on wheat seedlings (Triticum turgidum L.) grown in hydroponics and exposed to 0 to 30 µM Cu. Planta 241:847–860. https://doi.org/10.1007/s00425-014-2220-1
Khalid N, Aqeel M, Noman A et al (2021) Interactions and effects of microplastics with heavy metals in aquatic and terrestrial environments. Environ Pollut 290:118104. https://doi.org/10.1016/j.envpol.2021.118104
Li H, Luo N, Li YW et al (2017) Cadmium in rice: transport mechanisms, influencing factors, and minimizing measures. Environ Pollut 224:622–630. https://doi.org/10.1016/j.envpol.2017.01.087
Liang Y, Sun W, Zhu YG, Christie P (2007) Mechanisms of silicon-mediated alleviation of abiotic stresses in higher plants: a review. Environ Pollut 147:422–428. https://doi.org/10.1016/j.envpol.2006.06.008
Liao JP, Lin XG, Cao ZH et al (2003) Interactions between arbuscular mycorrhizae and heavy metals under sand culture experiment. Chemosphere 50:847–853. https://doi.org/10.1016/S0045-6535(02)00229-1
Liu EK, He WQ, Yan CR (2014) White revolution to white pollution -Agricultural plastic film mulch in China. Environ Res Lett 9(9):091001. https://doi.org/10.1088/1748-9326/9/9/091001
Liu J, Zhang H, Zhang Y, Chai T (2013) Silicon attenuates cadmium toxicity in Solanum nigrum L. by reducing cadmium uptake and oxidative stress. Plant Physiol Biochem 68:1–7. https://doi.org/10.1016/j.plaphy.2013.03.018
Lowry OH, Randall RJ (1951) Protein measurement byt the folin reagent. J Biol Chem 193:265–275
Luyckx M, Hausman JF, Lutts S, Guerriero G (2017) Silicon and plants: current knowledge and technological perspectives. Front Plant Sci 8:1–8. https://doi.org/10.3389/fpls.2017.00411
Ma JF (2004) Role of silicon in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci Plant Nutr 50:11–18. https://doi.org/10.1080/00380768.2004.10408447
Ma X, Zhang Q, Zhu Q et al (2015) A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mol Plant 8:1274–1284. https://doi.org/10.1016/j.molp.2015.04.007
MajeedZargar S, Nazir M, Kumar Agrawal G et al (2010) Silicon in plant tolerance against environmental stressors: towards crop improvement using omics approaches. Curr Proteomics 7:135–143. https://doi.org/10.2174/157016410791330507
Meharg C, Meharg AA (2015) Silicon, the silver bullet for mitigating biotic and abiotic stress, and improving grain quality, in rice? Environ Exp Bot 120:8–17. https://doi.org/10.1016/j.envexpbot.2015.07.001
Mitani N, Chiba Y, Yamaji N, Ma JF (2009) Identification and characterization of maize and barley Lsi2-like silicon efflux transporters reveals a distinct silicon uptake system from that in rice. Plant Cell 21:2133–2142. https://doi.org/10.1105/tpc.109.067884
Naeem A, Saifullah Z-u-R et al (2018) Silicon nutrition lowers cadmium content of wheat cultivars by regulating transpiration rate and activity of antioxidant enzymes. Environ Pollut 242:126–135. https://doi.org/10.1016/j.envpol.2018.06.069
Nagajyoti PC, Lee KD, Sreekanth TVM (2010) Heavy metals, occurrence and toxicity for plants: a review. Environ Chem Lett 8:199–216. https://doi.org/10.1007/s10311-010-0297-8
Nawaz M, Wang X, Saleem MH et al (2021) Deciphering plantago ovata forsk leaf extract mediated distinct germination, growth and physio-biochemical improvements under water stress in maize (Zea mays l.) at early growth stage. Agronomy 11(7):1404. https://doi.org/10.3390/agronomy11071404
Nisar S, Iqbal M, Ashraf J et al (2022) Enhancing salt tolerance in cotton by improving its morpho-physiological and antioxidant potential through foliar applied silicon. SILICON. https://doi.org/10.1007/s12633-022-01849-7
Hamilton PB (1965) Amino-acids on hands. Nature 205:284–285
Parmar P, Kumari N, Sharma V (2013) Structural and functional alterations in photosynthetic apparatus of plants under cadmium stress. Bot Stud 54:1–6. https://doi.org/10.1186/1999-3110-54-45
Phillips JM, Hayman DS (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 55:158-IN18. https://doi.org/10.1016/s0007-1536(70)80110-3
ur Rehman MZ, Rizwan M, Rauf A et al (2019) Split application of silicon in cadmium (Cd) spiked alkaline soil plays a vital role in decreasing Cd accumulation in rice (Oryza sativa L.) grains. Chemosphere 226:454–462. https://doi.org/10.1016/j.chemosphere.2019.03.182
Saleem MH, Parveen A, Khan SU et al (2022) Silicon fertigation regimes attenuates cadmium toxicity and phytoremediation potential in two maize (Zea mays L.) cultivars by minimizing its uptake and oxidative stress. Sustainability 14(3):1462. https://doi.org/10.3390/su14031462
Shafeeq-ur-Rahman XQ, Yatao X et al (2020) Silicon and its application methods improve physiological traits and antioxidants in Triticum aestivum (L.) under cadmium stress. J Soil Sci Plant Nutr 20:1110–1121. https://doi.org/10.1007/s42729-020-00197-y
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 2012:1–26. https://doi.org/10.1155/2012/217037
Shi M, Lam TTY, Hon CC et al (2010) Molecular epidemiology of PRRSV: a phylogenetic perspective. Virus Res 154:7–17. https://doi.org/10.1016/j.virusres.2010.08.014
Smith SE, Smith FA (2011) Roles of arbuscular mycorrhizas in plant nutrition and growth: new paradigms from cellular to ecosystem scales. Annu Rev Plant Biol 62:227–250. https://doi.org/10.1146/annurev-arplant-042110-103846
Sohail MI, ur Rehman MZ, Rizwan M et al (2020a) Effect of biochars, biogenic, and inorganic amendments on dissolution and kinetic release of phytoavailable silicon in texturally different soils under submerged conditions. Arab J Geosci 13:1–12. https://doi.org/10.1007/s12517-020-05399-3
Sohail MI, Zia ur Rehman M, Rizwan M et al (2020b) Efficiency of various silicon rich amendments on growth and cadmium accumulation in field grown cereals and health risk assessment. Chemosphere 244:125481. https://doi.org/10.1016/j.chemosphere.2019.125481
Steel RGD, Torrie JH (1962) Principles and procedures of statistics with special reference to the biological sciences. Inc Stat 11:170. https://doi.org/10.1002/bimj.19620040313
Szollosi R, Varga IS, Erdei L, Mihalik E (2009) Cadmium-induced oxidative stress and antioxidative mechanisms in germinating Indian mustard (Brassica juncea L.) seeds. Ecotoxicol Environ Saf 72:1337–1342. https://doi.org/10.1016/j.ecoenv.2009.04.005
Tului V, Janmohammadi M, Abbasi A et al (2021) Influence of iron, zinc and bimetallic zn-fe nanoparticles on growth and biochemical characteristics in chickpea (Cicer arietinum) cultivars. Agric For 67:179–193. https://doi.org/10.17707/AgricultForest.67.2.13
Usman M, Zia-ur-Rehman M, Rizwan M et al (2023) Effect of soil texture and zinc oxide nanoparticles on growth and accumulation of cadmium by wheat: a life cycle study. Environ Res 216:114397
Viciedo DO, de Mello PR, Lizcano Toledo R et al (2019) Silicon supplementation alleviates ammonium toxicity in sugar beet (Beta vulgaris L.). J Soil Sci Plant Nutr 19:413–419. https://doi.org/10.1007/s42729-019-00043-w
Yemm EW, Willis AJ (1954) The estimation of carbohydrates in plant extracts by anthrone. Int J Pharm Pract 7:197. https://doi.org/10.1111/j.2042-7174.1999.tb00969.x
Yu R, Ji J, Yuan X et al (2012) Accumulation and translocation of heavy metals in the canola (Brassica napus L.)-soil system in Yangtze River Delta. China Plant Soil 353:33–45. https://doi.org/10.1007/s11104-011-1006-5
Zhang C, Wang L, Nie Q et al (2008) Long-term effects of exogenous silicon on cadmium translocation and toxicity in rice (Oryza sativa L.). Environ Exp Bot 62:300–307. https://doi.org/10.1016/j.envexpbot.2007.10.024
Acknowledgements
This research work was funded by Institutional Fund Projects under grant No. (IFPIP: 439-130-1443). The authors gratefully acknowledge technical and financial support provided by the Ministry of Education and King Abdulaziz University, DSR, Jeddah, Saudi Arabia. We are thankful to University of Central Punjab Constituent College Yazman Road Bahawalpur for sharing lab facilities and providing space for research experiments.
Funding
This work was funded by Institutional Fund Projects under grant No. (IFPIP: 439-130-1443), Ministry of Education in Saudi Arabia.
Author information
Authors and Affiliations
Contributions
ZA and ARN planned and supervised the research, UUZ conducted the research work and wrote the introduction part, and AA and SA wrote the manuscript; UUZ, ZA, and SH did the static analysis and graphical representation; EAW and HFA read the manuscript as proofreading and arranged it according to the journal style; ZA provided reagents, assisted in the analytical work and MAI improved the English language quality of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
All authors approve this manuscript for submission to Environmental Science and Pollution Research.
Consent to participate
All authors participate in the preparation of the manuscript.
Consent for publication
All authors consent to the manuscript published in Environmental Science and Pollution Research.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Gangrong Shi
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zia, U.U., Niazi, A.R., Ahmad, Z. et al. Dose optimization of silicon for boosting arbuscular mycorrhizal fungi colonization and cadmium stress mitigation in maize (Zea mays L.). Environ Sci Pollut Res 30, 67071–67086 (2023). https://doi.org/10.1007/s11356-023-26902-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-26902-9