Abstract
The immune system protects human health from the effects of pathogenic organisms; however, its activity is affected when individuals become infected. These activities require a series of molecules, substrates, and energy sources that are derived from diets. The consumed nutrients from diets help to enhance the immunity of infected individuals as it relates to COVID-19 patients. This study aims to review and highlight requirement and role of macro- and micronutrients of COVID-19 patients in enhancing their immune systems. Series of studies were found to have demonstrated the enhancing potentials of macronutrients (carbohydrates, proteins, and fats) and micronutrients (vitamins, copper, zinc, iron, calcium, magnesium, and selenium) in supporting the immune system’s fight against respiratory infections. Each of these nutrients performs a vital role as an antiviral defense in COVID-19 patients. Appropriate consumption or intake of dietary sources that yield these nutrients will help provide the daily requirement to support the immune system in its fight against pathogenic viruses such as COVID-19.
Similar content being viewed by others
Introduction
Compounds in foods that affect a recognizable change in an organism’s immune system are referred to as immune-enhancing nutrients. Glutamine, arginine, omega-3 fatty acids (FAs), and several others are nutrients that have been proven to express regulatory effects on immune function. Hence, they are assumed as immuno-nutrients or immune regulators (Schloerb 2001). It is well established that diet influences immune levels, and poor diet is the most rampant culprit in cases of weakened immunity worldwide (WHO 2020a, b). Micronutrients such as folic acid, Zn, Mg, Mn, Se, Fe, Cu, B-vitamins, and water-soluble vitamins all function to improve the human body defense mechanism activity against many disease-causing organisms as shown in Table 1 (Rytter et al. 2020). The lack of these essential micronutrients could cause an altered immune response (Gleeson 2013). Therefore, a healthy immune system resulting from a good and balanced consumption of nutrients needed in the body is essential (Agovino et al. 2018).
A healthy immune system is a major defense against disease with no known drugs, an example of such disease being the current COVID-19 pandemic. Several vital nutrients are required for enhancing the body’s innate and adaptive defense mechanisms (Purohit et al. 2020b). Poor micronutrients negatively affect immune function and reduce the body’s ability to resist infections (Carr and Maggini 2017; Gombart et al. 2014). Apart from magnesium and vitamin E, micronutrients have therapeutic effects in the European Union for being a key player in normal immune function (EU 2020). Omega-3 FAs are another nutrient group that helps in the regulation of the immune system, mainly by modulating inflammatory responses (Calder 2020). Nutritional status can greatly impact a person’s total well-being, decrease non-communicable diseases, and reduce vulnerability to growing infections (Agovino et al. 2018). Currently, there is no permanent or proven pill, vaccine or treatment, food, or herb for confirmed prevention of COVID-19 infection based on the information from WHO (WHO 2020a). Poor nutritional status can increase a patient’s susceptibility to contracting lethal COVID-19 (Purohit et al. 2020a). Many nutrients play a vital role in the immune system’s functioning, and an adequately managed balanced diet will enhance the effectiveness of the immune system. Also, for an effective immune response, it is required that zinc, iron, selenium, copper, magnesium, and vitamins be present in consumed diets (Calder 2013; Smith et al. 2018). Nutrients like vitamins and minerals influence health and upregulation responses of the immune system to harmful agents as well as laboratory antigens (Jose et al. 2017; Pan et al. 2018; El-Senousey et al. 2018) . Prior to admission, COVID-19 patients should be checked for nutrient deficiencies using standard screening tools to determine effective treatment and dietary regimens (Reber et al. 2019; Lomax and Calder 2009; Yaqoob 2017).
In review aims to discuss the immunoboosting potential of macro and micronutrients in COVID support therapy. This review intends to provide basic insight and understanding of synergetic mechanisms of vitamin A, B, C, D, E, zinc, iron, and selenium on the immune system. Later, we elaborate the immune functions of micro- and macronutrients in COVID support therapy with recently published data. Various micro- and macronutrients are highlighted in this review for the management of COVID-19.
Immune fuctions of nutrients
Macronutrients (carbohydrates, fats, and proteins)
Carbohydrates also known as sugars are the primary energy source that should be obtained frequently from food intake. Research has also shown that certain protein monomers such as arginine, glutamine, taurine, methionine, and cysteine possess immunomodulatory activities (Li et al. 2007). Meat is a highly digestible (about 94% digested) protein source as compared to beans (78%) and whole wheat (86%) (Bhutta 1999). Meat is a good origin of all the eight essential amino acids and lacks in non-essential amino acids. The use of a scoring method known as protein digestibility–corrected amino acid score to rate how viable a protein is uses a maximum score of 1.0. Most plant foods have values of 0.5–0.7 while animal proteins like beef have a value of 0.9 (Schaafsma 2000). Glutamic acid/glutamine is the highest amino acid in meats (16.5%), followed by aspartate, alanine, and arginine. Taurine is a free amino acid that is primarily present in all tissues, with its presence higher in blood, heart, and retina (Wójcik et al. 2010). Taurine mainly helps in bile acid conjugation and other biological functions. Also, taurine is implicated in several physiological processes including osmotic regulation, modulation of the immune responses, and membrane integrity. It plays a vital role in nervous system coordination and the eyes (HuxTable 1992). Due to the limited nature of the endogenous synthesis of taurine, it is often considered a conditional essential nutrient, and it is majorly obtained from foods. Aquatic animals are mostly a preferred taurine source than their terrestrial animal counterparts (Bouckenooghe et al. 2006; Dragnes et al. 2009; Spitze et al. 2003). High taurine levels have been reported in some marine invertebrates, compared to terrestrial plants with a low taurine content (Kataoka and Ohnishi 1986). Taurine is also present in considerable amounts in meats (77 mg/100 g in beef and 110 mg/100 g in lamb) (Purchas et al. 2004).
Furthermore, some polyunsaturated fatty acids (PUFA), as well as their metabolic products, modulate cell functions, particularly omega (ω)-3 fatty acids (DHA and EPA) that affect immune cell activities (Gleeson 2013). However, most positive effects are caused by n-3 PUFA, particularly DHA and EPA. These FAs have been shown to possess pleiotropic effects, consequently influencing the production of the inflammatory components via in vivo, membrane functionality, and blood flow properties and studies have shown the preventive properties of n-3 PUFA in several ailments (Riediger et al. 2009). Beef from animals fed with pasture is a preferred source of ω-3 fats than those fed with grains, and this clarifies why Australian meats have a preferred fatty acid ratio compared with that in the USA, where grains are the major source of food for the animals (Sinclair and O’Dea 1987; Marmer et al. 1984) . Studies have shown that meats from fowls and pigs possess lower ω-3 PUFA than beef and lambs though fish remain the most preferred source of omega-3 PUFA. Moreover, ω-3 and ω-6 PUFAs mostly modulate inflammatory effects, being a starting material for leukotrienes or prostaglandins and resolvins or protectins (both are derivatives of omega-3 FA that helps inflamed tissues to return to normal once the inflammatory response is over) respectively. Protectin D is an omega-3 serving as a newly discovered antiviral drug that shows a promising role in interventions of the novel virus COVID-19 (Purohit et al. 2020a). Recently, 160 mg and 90 mg daily consumption of omega-3 fats (EPA and DHA) was recommended for males and females, respectively, as the Nutrient Reference Values for Australians, with an upper consumption range of 610 mg for control and 430 mg for prevention of lingering chronic ailments (National Health and Medical Research Council 2006). Australian beef is a good source of omega-3 FA with 135 g of red meat yielding more significant than 30 mg (Food Standards Australia New Zealand 2002). Also, mutton muscle meat yields a good level of long-chain omega-3 PUFA. Among Australians, fish and red meat serve as the first and second-largest sources of omega-3 long-chain PUFA (Howe et al. 2006).
Endogenous negative-feedback mechanisms help resolve inflammation quickly after an immune response. In achieving this, EPA and DHA localized at the inflammatory region are transformed into cell signaling molecules known as specialized pro-resolving lipid mediators (SPMs), namely maresins, protectins, and resolvins. They work together to quench inflammation and ensure the process of healing in tissues with respiratory system inclusive (Calder 2012; Basil and Levy 2016) . Sea animals are rich in protein with lower calories, and high levels of omega-3 long-chain PUFAs compared to terrestrial animals (Tacon and Metian 2013) . It has been found that the absence of PUFAs can cause a slow removal of inflammation (Mehta et al. 2020) . This will be crucial to the so-known cytokine storm observed amid severe COVID-19 with the presentation of unsuppressed inflammation (Pedersen and Ho 2020; Gao et al. 2017). SPMs derived from DHA and EPA protect against acute lung injury and acute respiratory distress syndrome (ARDS) (Zhang et al. 2019).
Antioxidants rich in DHA and EPA have been employed in several trials involving ARDS patients. A recent review on these trials noted an undeniable betterment in oxygen flow in the circulatory system and a considerable amount of decrease in the need for ventilators, new organ breakdown, time spent in the intensive care unit (ICU), and mortality (Dushianthan et al. 2019). Eicosanoids derived from n-3 PUFA possess the ability to downregulate immune response (Calder 2001). Eicosanoids derived from n-6 PUFA possess pro-inflammatory properties and enhanced immune responses such as pyrexia and agony. In the past four decades, research has shown the importance of EPA and DHA for protection against several diseases (Calder 2006). α-linolenic acid, an omega-3 FA usually found in seeds and oils, can be used as a substrate for DHA and EPA biosynthesis. Although, its synthesis may be inadequate in old people and babies (Brenna 2002) . Altering the fatty acid (FA) content in the diet helps to enrich meat from chicken with n-3 PUFA (Ribeiro et al. 2013; Konieczka et al. 2017). Meat from rabbits contains 60% unsaturated FA and 32.5% PUFA, this is higher than what is obtained from other meats including farm birds, and it may serve as an important diet in humans (Wood et al. 2008). DHA and EPA are the most bioactive n-3 PUFA; lower activity observed in α-linolenic acid is due to its biotransformation to the bioactive EPA which is usually lacking in man (17:1); enhancement of DHA and EPA in rabbits can be done by animal feeding (Decker and Park 2010) .
Micronutrients (vitamins and minerals)
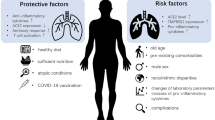
Deficiencies of trace elements result in immune disorder. A nutritious and balanced meal has the potential to boost and regulate immune cells against COVID as demonstrated in Fig. 1. A viable immunity is the most potent tool to combat a disease like COVID-19, with no established treatment or drug. The increased risk of death due to poor nutrition is due to increased severity of infection coupled with slowed recuperation.
In addition, an increase in more nutrient demands results from infection (WHO 2020a, b). A recent study reported that optimum nutritional status helps protect the body against infections with viruses (Maggini et al. 2018; Calder 2020). Also, another study provided nutritional advice to help curtail lung damages caused by infections with coronavirus and other lung issues (Wu and Zha 2020) . Studies have reported that deficiency in individual nutrient or combo of nutrients affects the immune system by activating immune cells, modifying the production of signaling molecules, and expression of genes (Valdés-Ramos et al. 2010). Also, depression of immune function and higher vulnerability to infection may occur. Proper intake of the required amount of nutrients is crucial in the strong maintenance of immune response (Gleeson et al. 2004; Fernández-Quintela et al. 2020).
Vitamin A
Studies have shown that consumption of vitamin A-deficient diets hampers the potency of attenuated vaccines in bovine coronavirus and thereby increases vulnerability of calves to infections. Vitamin A also demonstrates a prospective headway towards discovering a coronavirus treatment and preventing lung problems (Jee et al. 2013) . Vitamin A and folate are abundant in the liver, but the levels in lean meat are low (Table 1). Vitamin levels are often higher in older animals; hence, beef has more vitamins than mutton meat or veal (Williams et al. 2007). Chicken meat is the only meat with a useful amount of β-carotenes which can serve as a precursor for vitamin A which can be enhanced by food fortification (Decker and Park 2010) .
Vitamin A and folic acid are often largely present in offal meats (Biesalski 2005). Vitamin A is important for adequate development. A study recommended the liver as a good vitamin A source with 100 g yielding more than the recommended dietary value (USDA 2011). Mucin production in the respiratory tract is sustained by vitamin A. Mucin serves as a barrier that protects the respiratory tract from pathogenic infections (Fan et al. 2015). Vitamin A is very crucial in the production of antibodies. It helps in the movement of T lymphocytes to infection or inflammation site, enabling cells of the mucous membrane involved in IgA production to develop an appropriate immune response (Mora and Andrian 2006) .
A randomized, single-blinded, and two-arm clinical trial of 7 days was executed to evaluate severity and mortality rate in ICU patients suffering from COVID-19 that were supplemented with vitamin A, B, C, D, and E (Beigmohammadi et al. 2020).
Vitamin B
Another wise approach could be to supplement the COVID-19 patient with vitamin B. Over two-thirds of the daily required vitamin B can be obtained from 100 g serving of red meat as they serve as the most bioavailable source of vitamin B (Sinclair et al. 1999; Williams et al. 2007). A 100 g intake of red meat can yield 25% of the everyday required amount of other B vitamins such as vitamin B2, B3, pyridoxine, and pantothenic acid (Williams 2007a, b). Seafood such as herring, oysters, clams, anchovies, pilchard, and sardines contain high levels of vitamin B12. Mussels, clam, and oysters have a B12 content of 15.71, 87.0, and 46.3 mg per 100 g respectively. B12 content in sardine, salmon, and tuna ranges from 3.8 to 8.9 mg per 100 g. Clams and mollusks have B12 content of 98 mg per 100 g (Watanabe et al. 2001).
The combination of C and E vitamins serves as a potential antioxidant treatment for COVID-19 cardiac complications (Wang et al. 2020). More so, there is no adequate fact on vitamin E use as an agent for prevention or treatment against COVID-19. It is suitable to maintain nutritional habits, alongside a healthy nutritious diet containing enough minerals, antioxidants, and vitamins. Bio-available vitamin B can be obtained from meat. However, its concentrations vary considerably in meats from different animal species. High temperatures during meat processing lower the levels of vitamin B (Lombardi-Boccia et al. 2005). The daily recommended amount of vitamin B12 for an adult is 22 µg, and this can be obtained through the consumption of 100 g of beef (LARN 1996). Studies have revealed that 100 g consumption of rabbit meat can yield triple the vitamin B12 daily requirement (DR), comprising about 8% vitamin B2, 21% vitamin B6, 12% vitamin B5, and 77% vitamin B3 (Hernàndez and Dalle Zotte 2010) . However, pork yields 37% of the RDI for vitamins B1, B2, B6, and B12 (Esteve et al. 2002).
For niacin, a consumption of 100 g of chicken breast will yield 56% of the DR and 27% DR of B6 vitamin, while a consumption of 100 g of turkey breast will provide 31% DR of niacin and 29% DR of vitamin B6 (USDA 2011). Regulatory T cells (Treg) are known for their high expression of vitamin B9 receptors; vitamin B9 (folic acid) is a survival factor, and it helps in preventing excessive immune response (Hernàndez and Dalle Zotte 2010; Sakaguchi et al. 2009). Inadequate vitamin B9 may result in a low population of Treg cells, thereby resulting in upregulation in the susceptibility of the organism to sudden inflammation outburst, as observed during the fatal stage in patients with COVID-19 (Kinoshita et al. 2012).
Vitamin C
It is a water soluble micronutrient that acts as an antioxidant. Vitamin C cannot synthesize in the body because of the absence of the enzyme that is a key factor for its de novo biosynthesis. It blocks oxidative stresses via reduction or prevention of generation of reactive oxygen and nitrogen species (Xu et al. 2021; Aldwihi et al. 2021). Vitamin C provides support to the immune system, protecting it against the coronavirus. Vitamin C can be used as an alternative treatment in COVID-19 cases. Reports from some controlled trials showed that sometimes, patients administered with vitamin C showed a reduced occurrence of pneumonia. However, high-dose administration in patients is yet to be approved (Hemila 1997).
Vitamin C influences various aspects of immunity such as development and function of adaptive and innate immune cells, aiding activities of the epithelial barrier, translocation of white blood cells to infection sites, phagocytosis, killing of microbes, and producing antibodies. The deficiency of vitamin C results in susceptibility to worse respiratory infections (Hemila 2017).
A study reported a reduced risk of pneumonia due to intake of vitamin C supplements (Hemilä and Chalker 2013) . The mortality risk is reduced in aged people by adding vitamin C to foods (Hemila and Louhiala 2013). Vitamin C intake decreases the virulence period of infections associated with the upper region of the respiratory tract and decreased significantly the risk of infection when administered as a preventative measure to people who undergo enhanced physical stress (Hemilä and Chalker 2013). The intake of vitamin C-deficient foods by a healthy young adult (humans) results in reducing vitamin C content in mononuclear cells by half and reduced ability of the T lymphocyte-mediated immune responses in recalling antigens (Jacob et al. 1991).
Recently, investigations to validate its effect in severe COVID-19 treatment have begun (Carr 2020). Vitamin C may possess effects on viral infections of the respiratory tract especially when specific therapy for COVID-19 is absent. It is noteworthy that vitamin C increases the resistance to coronavirus and under certain conditions reduce the risk of infections of the lower respiratory tract (Hemilä 2003; Hemilä and Douglas 1999).
For severe conditions of COVID-19 which require ICU, its high-dose intravenous administration is highly valuable for such patients. Numerous trials of its high-dose i/v administration have demonstrated varied outcomes regarding laboratory and clinical results for acute lung injury and acute respiratory suffering disease (Hemilä and Chalker 2020) .
Its combination with glycyrrhizic acid was proposed for COVID-19 treatment by bioinformatical network pharmacology. In another case, its co-administration with quercetin was also suggested. But, further investigations did not provide a strong indication for the utility of its high dose or its combination with zinc for the treatment. At present, pilot-scale studies are ongoing such as in Canada (LOVIT-COVID) and Italy (NCT04323514) for proving its utility for improving the condition of COVID-19 patients (Clemente-Suárez et al. 2021).
In a single case report, a 74-year-old female suffering from COVID-19 ARDS was supplemented with vitamin C (1 g bis in die upto six days orally) and after that, 11 g/day for 10 days as a continuous infusion. Her clinical status was improved within 5 days of treatment and she was capable of halting mechanical ventilation (Waqas Khan et al. 2020) .
Seventeen patients (age 64 ± 14 yrs, M:F = 10:7) of the USA suffering from SARS-CoV-2 were administered intravenously with vitamin C (1 g ter in die for 3 days). It was observed that there was a significant decline in ferritin and D-dimer intensities and a fraction of FiO2 that helps in inspiration of oxygen (Hiedra et al. 2020).
In China, 34-year-old subject (male) with COVID-19-associated symptoms (dry cough, fatigue, reduced hunger, and subjective fever) was given vitamin C (3 g per day) in combination with antiviral and antimicrobial cures. After 14 days of this ailment, it improved her SpO2 levels that assured her improved clinical condition (Chen et al. 2021) .
A female patient (66 years old) from China suffering from COVID-19-associated shortness of inhalation and continuous fever received orally (200 mg ter in die) of vitamin C with diammonium glycyrrhizinate. After 7 days of the following treatment, her condition was improved and she was completely healthy (Ding et al. 2020).
For twelve Chinese patients with severe (n = 6, 56 years old) or critical (n = 6; 63 years old) COVID-19 pneumonia, a high dose of vitamin C (162.7 and 178.6 mg/kg/day) was administered intravenously. There was a significant decline in counts of C-reactive protein, lymphocyte, and CD4+ T cells, as well as an enhancement of organ failure score, which was improved in the severe group in contrast to the critical group (Zhao et al. 2020).
Twelve trials with 1766 subjects of ICU conditions showed that its administration shortened the stay in ICU by 8%. Another 8 trials demonstrated that its administration declined the period of mechanical ventilation in patients who needed the longest ventilation (Hemilä and Chalker 2019).
Vitamin D
Vitamin D fights against acute infections of the respiratory tract. Recent studies reported a reduced risk of infections with COVID-19 on increased intake of vitamin D as shown in Fig. 2. It plays a critical role by affecting the maturation of immune cells (Grant et al. 2020). Studies reported a reduced level of vitamin D in healthy individuals particularly at the end of winter season, this coincides with COVID-19 discovery in winter of 2019 (Arabi et al. 2020). Vulnerable patients are advised to increase their vitamin D intake. Due to the stay-at-home order, there is reduced exposure of people to sunlight. Therefore, it is very important to increase the consumption of dietary sources rich in vitamin D (Zabetakis et al. 2020). Vitamin D-rich foods include oily fish and cod liver oil, among others (Larsen et al. 2011). The lack of vitamin D in cold weather periods is connected to viral outbreaks. The proper vitamin D level prevents the risk of chronic conditions like high blood pressure, cancer, heart diseases, stroke, diabetes, and hypertension in people with respiratory infections (Muscogiuri et al. 2017).
Vitamin D performs a series of function in the body, such as protection of the respiratory tract, tight junction preservation, and elimination of encapsulated viruses via cathelicidin and defensin activation, and decreases pro-inflammatory cytokine production by the innate immune system; thus, the risk of cytokine storm development is reduced, which will consequently lead to pneumonia. Considering that going outside is not very feasible and the exposure to sunlight is limited, foods now serve as the best alternative source of vitamin D. Fish, egg yolk, liver, yogurt, and milk are known foods that contain vitamin D. Several body defense cells possess vitamin D receptors which enhance efficacy after bonding to ligands, and therefore, vitamin D greatly impacts on immunity. Vitamin D promotes monocyte differentiation into macrophages, thereby increasing their killing capacity, affecting cytokines release, and promoting antigen presentation. Additionally, metabolites of vitamin D help in regulating the release of proteins with antimicrobial properties that destroy pathogens and therefore reduce infections of the lungs (Gombart 2009; Greiller and Martineau 2015). Research has shown that vitamin D is present in meat (Ovesen et al. 2003). Cholecalciferol (vitamin D3), which is obtained through skin exposure to sunlight, has greater potency than ergocalciferol (vitamin D2) found in mushrooms (Holick 2008; Norman 2008). Vulnerability to respiratory tract infection has been reported in some studies in people with low vitamin D in the blood (Cannell et al. 2006; Jollie et al. 2013 Also, several meta-analyses reported that the addition of vitamin D to food decreases the risk of respiratory tract infections in humans (Charan et al. 2012; Autier et al. 2017). Its lack impedes the functions of the immune system due to its immunomodulatory role, thereby increasing inborn immunity by antiviral peptide production, which helps in the improvement of mucosal defenses (Gombart et al. 2005; Wang et al. 2010).
Recently, some reviews propounded that low vitamin D in the blood results in compromised immune functions in the respiratory tracts; thus, severity as well as mortality increases in patients with COVID-19. Also, its antiviral effects inhibit directly viral replication, have been reported in recent data, and are also effective in immunomodulatory and anti-inflammatory away (Teymoori-Rad et al. 2019). Poultry products are good sources of riboflavin, pyridoxine, and vitamin B3 (Borenstein 1981). Unlike the riboflavin content, the thiamine content of muscles is drastically reduced when muscles are cooked (Al-Khalifa and Dawood 1993) . For patients with COVID-19, an Italian research group suggested a dietary protocol that is involving vitamin D supplementation for those who are deficient (Caccialanza et al. 2020) .
Various data based on clinical trials revealed its varying results on respiratory tract infections. Thirty-nine types of research (14 clinical trials, 8 case–control, 4 cross-sectional, and 13 cohort studies) indicated a significant impact of low level of vitamin D and more threat of both upper and lower respiratory tract infections from observational investigations. However, inconsistent outcomes were reported from RCTs (Jollie et al. 2013).
Twenty-five randomized controlled investigations (meta-analysis) revealed that its supplementation interconnected with low threat of acute respiratory infections (OR = 0.88 and p < 0.001). Its positive effect was even more pronounced in subjects with vitamin D deficiency at the start of intervention (Martineau et al. 2017).
Some research suggests a link between vitamin D insufficiency and liability to COVID infection and disease severity. Ilie et al. conducted a study in 20 European countries and observed a negative correlation between its mean serum levels and COVID-19 belongings and mortality rate. Rigorously, its lower level has been recognized in geriatric patients, particularly in Spain, Italy, and Switzerland (Ilie et al. 2020).
Seven retrospective investigations (1368 subjects with COVID-19) found its mean serum level of 22.9 nmol/l. Its lower serum levels were linked with subjects with poor disease forecasting in contrast to those with good results, demonstrating a standardized mean difference of − 5.12 (p = 0.012). It was summarized that its deficiency shows an independent fundamental role in COVID-19 severity (Munshi et al. 2020).
A consequent study with an Israeli cohort of 7807 patients recognized its low level among those who confirmed positive for COVID-19 contrasted to negative. Its lower level was measured as an independent threat factor for COVID-19 (p < 0.001) and hospitalization (p < 0.05) (Merzon et al. 2020).
Vitamin E
Tocopherol (vitamin E) is an effective immune-enhancing nutrient. It protects PUFA which usually enriches immune cells from oxidation. Marine fish are known for their expression of vitamin E. Salmon and shellfish can yield about 15% of vitamin E DR Coquette et al. 1986) . Chicken meat also contains vitamin E. Twelve milligrams is a DR of vitamin E according to EFSA NDA Panel (Holland et al. 1993). Fortified meat and dietary supplements with α-tocopheryl acetate can also serve as vitamin E sources; this possesses high anti-inflammatory properties. Vitamin E in muscle cell membranes helps to reduce lipid oxidation and could help prevent protein oxidation. Meats supplemented with vitamin E have a long shelf life and an improved color, flavor, and texture (EFSA NDA Panel 2015). It also plays a significant role in enhancing of immune reactions by inactivating and inhibiting free radicals (Zhang et al. 2010). Oral intake of vitamin E helps in the improvement of response by T cell and macrophage activities against infective agents and it reduces vulnerability to upper respiratory tract infections in older patients (Maggini et al. 2007; Meydani et al. 2005, 2004; Pae et al. 2012).
Vitamins C and E had been considerably authenticated to diminish lung infection in animals, and similar outcomes were obtained in geriatric patients. While, they had no advantage in children suffering from pneumonia (Murni et al. 2021).
Minerals
Selenium supplementation displays potential for COVID-19 treatment. Red meat supplies more than 20% RDI per 100 g served, although selenium levels in meat are likely to be greatly influenced by the location where animals were fed as well as the season (Williams 2007a, b). Zinc supplementation helps lower symptoms of COVID-19 such as stooling and reduces infections of the respiratory tract. The increasing risk in the development of acute infections of the respiratory tracts results due to low iron levels in the body (Guillin et al. 2019; Wu et al. 2019). Zinc, a trace element, plays a significant role in immune cell development and is known for its crucial role as a cofactor for many enzymes (Prasad 2008). An inadequate amount of zinc can lead to defective immunity resulting in increased vulnerability to pneumonia (Walker and Black 2004; Hess et al. 2009). Increasing zinc intakes help against infections with COVID-19 due to reduced replication of the virus; it also reduces gastrointestinal effects and decreases respiratory symptoms (Zheng et al. 2020).
Studies have shown that an average intake of 40 mg of zinc per day may help in controlling RNA-viruses such as coronaviruses and influenza (McCarty and DiNicolantonio 2020). Research has shown that a man’s zinc status is among the vital factors determining immunity to infections with virus, stating that low intake of zinc in a population increases the risk of contracting infections such as HIV (Read et al. 2019). Best sources of iron and zinc include beef and lamb meats, with 100 g of meat providing a portion of the daily adult requirements. Heme iron, which is mainly found in meat, is well absorbed; meat protein promotes iron absorption from meat. The rate of absorption of iron from meat is higher than that from plants (heme iron); an example is the absorption of folic acid which is tenfold higher compared to vegetables (Biesalski 2005). In addition, the absorption of zinc from animal protein diet is higher than plant foods; therefore, vegetarians should increase their zinc intake by 50% higher (National Health and Medical Research Council 2006). Studies on human subjects reported that people on a low-copper diet have reduced lymphocyte proliferation and decreased IL-2 production, and copper administration reverses these effects (Hopkins and Failla 1997). Raw lean cuts from beef and veal have a copper content ranging from 0.055 to 0.190 mg per 100 g, while the copper content from 100 g of lamb and mutton ranges from 0.090 to 0.140 mg and 0.190 to 0.240 mg respectively. The consumption of 100 g meat or liver daily can yield half of the daily required micronutrients such as iron, selenium, zinc, and vitamins (Chan et al. 1995). Nutritional status maintenance is very crucial in combating COVID-19 (Williams 2007a, b).
Iron
Iron is basically found in a series of foods as heme and non-heme irons. Heme iron can be found only in the flesh of animals with its origin from hemoglobin and myoglobin, readily bioavailable and easily absorbed as an intact molecule by the intestinal lumen (Hallberg and Hulthen 2000; Simpson and McKie 2009). Heme iron can be obtained by consuming foods such as lamb, beef, and mutton. The benefit of heme iron in meat is that it is more biologically available with red meat having 72 to 87% and rabbit and pork meat having 56 to 62% (Lombardi-Boccia et al. 2002). Non-heme iron can be obtained from animal flesh when animals eat plant foods containing non-heme iron and also from fortified foods. Non-heme iron can also be obtained from the consumption of spinach, beans, breakfast fortified with cereals and lentils, and enriched breads.
Iron is the most abundant transitional metal that is present in the body. It is a crucial constituent for all alive cells since it plays a key role in metabolic processes such as energy production, transportation, and storage of oxygen, drug detoxification and synthesis, and repairing and transcription of DNA (Habib et al. 2021). It mainly bounds to macromolecules such as hemoglobin, transferrin, ferritin, and other iron-containing proteins. Unbounded iron (labile or catalytic iron) is considered a transitional pool of extracellular and cellular iron. Moreover, iron status and nutrition are modifiable modulators of the immunologic response to SARS-Cov-2 mRNA vaccines (Gozzi-Silva et al. 2021).
It is proposed that SARS-CoV-2 may need iron for replication and other roles, offering a prospective mechanism for greater pathogenicity in the existence of high SCI. Chakurkar et al. 2021 reported the role of ferrotoxicity in COVID-19 and the concept that SCI is a sign of poor results in COVID-19. Tissue injury either from a direct cytopathic effect can result in rise in their level or inflammation or ischemia leads to a burst release of intracellular iron stores (Chakurkar et al. 2021).
Iron chelation therapy is beneficial in COVID-19 because it is well known that SARS-CoV-2 requires iron for viral replication (Liu et al. 2020). So, this therapy can be a novel approach to COVID-19 treatment. This therapy signifies a pillar in the treatment of iron overload due to a wide spectrum of diseases and multiple chelating agents are currently registered and usually employed in clinical practice (Perricone et al. 2020). Bipyridyl and desferoxamine (iron chelators) are under clinical trial for COVID-19 patients (NCT04333550) (Romeo et al. 2001).
Banchini et al. suggested that iron helps in exploring both endogenous (insulin, heparin, and erythropoietin) and exogenous options (vitamins D and C, toclizumab, carvedilol), relating to hepcidin control in the setting of COVID-19 (Banchini et al. 2020). Abbas et al. also suggested the use of iron chelators to reduce disease severity in COVID-19 infection (Abbas and Mostafa 2020) .
Zinc
Zinc is an essential nutrient that must continuously be added to diets since it cannot be synthesized and stored in the body (Maggini et al. 2010) . The lack of zinc, even in a low grade, leads to acquired and innate immune system disorder (Shankar and Prasad 1998). Zinc supplementation is effective and cost-effective intervention method used to treat and prevent respiratory tract infections and diarrhea, especially in children living in poor areas (Baqui et al. 2003; Brooks et al. 2005). Apart from iron, beef, and lamb meat, pork meat can also be considered a rich dietary zinc source (Wyness et al. 2011).A total of 100 g of beef meat can yield 26% of zinc.
Zinc is considered to fortify the body’s antiviral machinery and its ions (Zn2+) play significant roles in the growth, replication, differentiation, and immune cell functions. Physical processes such as virus binding, infection, and uncoating, as well as inhibition of polymerase enzymatic processes and viral protease, connote the antiviral properties of Zn against different viruses (Overbeck et al. 2008; Haddad et al. 1999). Phytates make zinc from plant sources less bioavailable than animal sources like meat (Mocchegiani et al. 2000). Zinc deficiency in old people is associated with age-related immune response dysregulation (Prasad et al. 1993). Its supplementation in diets has been reported to improve immunity in old people (Mocchegiani et al. 1999). Its deficiency in the elderly is considered a risk factor resulting in susceptibility to infection. Zinc supplementation plays a crucial role in the prevention of infectious diseases (Kajanachumpol et al. 1995).
At lower concentrations, a combination of zinc ions (Zn2+) and zinc ionophores prevents replication of SARS-CoV, via inhibiting elongation of RdRP and reducing binding of RNA template. Resemblances among RdRP of SARS-CoV and SARS-CoV-2 may be responsible for inhibition of replication of SARS-CoV-2 in cell culture. According to one hypothesize, oral or i/v administration of zinc ascorbate could be beneficial in prevention and ailment of COVID-19, because of the higher intracellular Zn2+ concentration that inhibits SARS-CoV-2 replication (Lee et al. 2009).
Various clinical trials are presently ongoing for analyzing the effect of zinc in different forms for COVID-19 (Cingolani 2021) .
Forty-eight subjects (randomized double-blind study) suffering from cold got zinc acetate lozenges (80 mg/day) or placebo within 24 h once the symptoms begin. In contrast to placebo, there was a significant short in cold symptoms and total severity score of all symptoms (p value < 0.002) in zinc supplementation (Prasad et al. 2000).
Four patients suffering from COVID-19 received a high dose of zinc (207 mg/day) orally that resulted in better oxygenation and fast resolution of quickness of inhalation later after 1 day of ailment (Yao et al. 2020).
Sometimes, zinc has been recommended for improving common cold symptoms. A total of 100 subjects (randomized, double-blind, placebo-controlled study) with common cold received 13.3 mg zinc. In contrast to placebo, it considerably declined period of symptoms of common cold, from 7.6 to 4.4 days. In the case study, it was observed that the combination of azithromycin (500) + naproxen (500) + vitamin C (1000) + zinc + vitamin D3 (1000) resulted in significant outcomes in course of COVID-19 treatment (Khodavirdipour 2021) .
Selenium
Selenium plays a role in the regulation of several physiological functions and is also an integral part of selenoproteins; they form part of the body’s antioxidant defense system (e.g., glutathione peroxidase). The recommended intake of selenium for females and male adults is 60 and 70 μg per day in the UK and 55 and 70 μg per day in the USA respectively (Reilly 1998). This requirement often cannot be achieved because the selenium concentration in foods is different for different regions and countries.
Among red meats, lamb and beef meats have high selenium levels, followed by mutton and veal. Among white meats, reasonable selenium level is found in chicken (Bou et al. 2005). Low selenium level in the body leads to decrease in immune system function, cognitive decline, and mortality. Antiviral effects were observed under the higher intake of selenium or supplementation (Rayman 2012) . Studies have shown an increase in selenium levels of beef from animals given dietary supplementation of selenium (Juniper et al. 2008; Dokoupilová et al. 2007). Seafood is also a good dietary source of selenium; they ranked 17th according to selenium level by the USDA National Nutrient Database (Ralston 2008). Selenium obtained from fish is bioavailable and higher in amount than yeast (Fox et al. 2004).
Copper
Copper (Cu) is crucial at ensuring strong immunity; it plays a vital role in the normal functioning of helper T cells, neutrophils, natural killer cells, macrophages, and B cells that help in the destruction of infectious microorganisms and production of specific antibodies and cell-mediated immunity. The lack of copper in humans causes white blood cells to decrease, immune reactions, and abnormalities in bone and connective tissue (Percival 1998). Cu2+ also plays a crucial role by its active participation in immune cell growth and differentiation (Li et al. 2019). Cu2+ is found in foods such as crustaceans, mollusks, fish, and land meats; its deficiency affects innate and adaptive immunity (Munoz et al. 2007). Its supplementation helps in restoring the secretion and activity of Inter-leukin-2, which is very helpful for cytotoxicity of NK cell and T helper cell proliferation. A recent study reported the sensitivity of new coronavirus strains (SARS-CoV-2) to a copper surface (Van Doremalen et al. 2020). Other studies have revealed blockage of papain-like protease-2 by Cu2+, a protein required for replication by SARS-CoV-1; therefore, the need for high demand of copper supplementation is essential (Baez-Santos et al. 2015; Han et al. 2005). Due to the high competition in the absorption of Cu and Zn in the jejunum through metallothionein, high doses of zinc (greater than 150 mg per day) can lead to deficiency of Cu in healthy individuals. Therefore, it is possible that people taking Zn supplement regularly could be susceptible to contacting SARS-CoV-2.
Low Cu2+ levels can lead to stress responses by pathogens; hence, an optimal Cu2+ level is required. Presently, there is limited understanding of the effect of medicinal use of Cu2+ as related to COVID-19. The intake of Cu affects the immune function of a host as well as micronutrients metabolism which helps prevent virulence. Hence, Cu2+ intake is beneficial in patients with COVID-19 (Rahaa et al. 2020).
Magensium
Magnesium plays a pivotal role in immune system function regulation by strongly affecting immunoglobulin production, immunoglobulin M (IgM) lymphocyte binding, immune cell adhesion, T helper-B cell adherence, antibody-dependent cytolysis, and response of macrophages to lymphokines (Liang et al. 2012). Its role in an immune response against viral infections has been reported (Chaigne-Delalande et al. 2013). Magnesium plays a key role both in physiology and pathology. Newly, it has been hypothesized that its low level may favor the transition from mild to critical clinical manifestations of COVID-19. Decreased NK and T-cell cytotoxicity because of magnesium deficiency may illuminate the vulnerability of elder, hypertensive, obese, and diabetic patients to SARS-CoV-2 infection (Faa et al. 2021). Furthermore, its deficiency upregulates pro-inflammatory cytokine creation in monocytes and raises NFkB expression (Fanni et al. 2020).
Despite the nonexistence of controlled trials, magnesium supplementation for supportive treatment in COVID-19-suffering patients should be encouraged. This may be valuable in all phases of the COVID-19. It is very well known that magnesium is involved in over 600 enzymatic reactions in human cells. Its level may explain an increased risk of severe COVID-19 (Fanni et al. 2020).
A combined oral treatment of older COVID-19 patients with magnesium, vitamin D, and vitamin B12 reduced the percentage of patients needing oxygen and ICU support (Tan et al. 2020a, b).
An observational cohort study was executed for evaluating the combined effect of vitamin D, magnesium, and vitamin B12 in older subjects (≥ 50 years) with COVID-19. Eighteen subjects received DMB already onset of primary outcome and 26 subjects did not. Fewer treated subjects than controls needed beginning of oxygen therapy amid hospitalization (17.6 vs 61.5%, p = 0.006) (Tan et al. 2020a, b).
Conclusion
It is well established that diet influences immune levels, and poor diet is the most rampart culprit in cases of weakened immunity worldwide. Nutritional status can greatly impact a person’s total well-being, decrease non-communicable diseases, and reduce vulnerability to growing infections. The immune system protects the body from pathogenic organisms such as SARS-COV-2 by its surveillance activities. Its activity is affected when individuals become infected with microorganisms that cause diseases, leading to an increased rate of metabolism that demands substrates, molecules, and energy sources such as micronutrients (folic acid, Zn, Mg, Mn, Se, Fe, Cu, B-vitamins, vitamin E, vitamin D, vitamin C) and macronutrients (carbohydrates, fats, and proteins) that can only be derived from dietary sources by the consumption of foods. The lack of any of these important micronutrients could cause an altered immune response. These nutrients provide the immune system with vital supporting roles in its fight against pathogens and thereby reducing the risk associated with infections. Prior to admission, COVID-19 patients should be checked for nutrient deficiencies using standard screening tools to determine effective treatment and dietary regimens.
A series of studies was found to have demonstrated the enhancing potentials of macronutrients (carbohydrates, proteins, and fats) and micronutrients (vitamins, copper, zinc, iron, calcium, magnesium, and selenium) in supporting the immune system’s fight against respiratory infections. Each of these nutrients performs a vital role as an antiviral defense in COVID-19 patients. As the exploration for the effective cure for COVID-19 continues, particular micro- and macro nutrients may effect the severity of infection, symptoms, and outcomes related to the diseases.
Data availability
The data sets in this study are available from the corresponding author on reasonable request.
Abbreviations
- ARDS:
-
Acute respiratory distress syndrome
- COVID-19:
-
Coronavirus disease 2019
- Cu:
-
Copper
- DR:
-
Daily requirement
- Fe:
-
Iron
- ICU:
-
Intensive care unit
- Mg:
-
Magnesium
- Mn:
-
Manganese
- PUFA:
-
Polyunsaturated fatty acids
- Se:
-
Selenium
- SPMs:
-
Specialized pro-resolving mediators
- Treg:
-
Regulatory T cells
- Zn:
-
Zinc
References
Abbas AM, Mostafa AS (2020) Use of iron chelators to reduce the severity of COVID-19. Thromb Haemost 4:1042
Agovino M, Cerciello M, Gatto A (2018) Policy efficiency in the field of food sustainability. The adjusted food agriculture and nutrition index. J Environ Manage 15(218):220–233. https://doi.org/10.1016/j.jenvman.2018.04.058
Aldwihi LA, Khan SI, Alamri FF (2021) Patients’ behavior regarding dietary or herbal supplements before and during COVID-19 in Saudi Arabia. Int J Environ Res Public Health 18(10):5086. https://doi.org/10.3390/ijerph18105086
Al-Khalifa AS, Dawood AA (1993) Effects of cooking methods on thiamin and riboflavin contents of chicken meat. Food Chem 48(1):69–74
Arabi YM, Fowler R, Hayden FG (2020) Critical care management of adults with community-acquired severe respiratory viral infection. Intensive Care Med 46(2):315–328
Autier P, Mullie P, Macacu A, Dragomir M, Boniol M, Coppens K et al (2017) Effect of vitamin D supplementation on non-skeletal disorders: a systematic review of meta-analyses and randomized trials. Lancet Diabetes Endocrinol 5:986–1004
Baez-Santos YM, St John SE, Mesecar AD (2015) The SARS-coronavirus papain-like protease: structure, function and inhibition by designed antiviral compounds. Antiviral Res 115:21–38
Banchini F, Vallisa D, Maniscalco P, Capelli P (2020) Iron overload and hepcidin overexpression could play a key role in COVID infection, and may explain vulnerability in elderly, diabetics, and obese. Acta Biomed 91. https://doi.org/10.23750/abm.v91i3.9826
Baqui AH, Zaman K, Persson LA, El Arifeen S, Yunus M, Begum N et al (2003) Simultaneous weekly supplementation of iron and zinc is associated with lower morbidity due to diarrhea andacute lower respiratory infection in Bangladeshi infants. J Nutr 133(12):4150–4157
Basil MC, Levy BD (2016) Specialized pro-resolving mediators: endogenous regulators of infection and inflammation. Nat Rev Immunol 16:51–67
Beigmohammadi MT, Bitarafan S, Hoseindokht A, Abdollahi A, Amoozadeh L, Mahmoodi Ali Abadi M, Foroumandi M (2020) Impact of vitamins A, B, C, D, and E supplementation on improvement and mortality rate in ICU patients with coronavirus-19: a structured summary of a study protocol for a randomized controlled trial. Trials 21(1):614. https://doi.org/10.1186/s13063-020-04547-0
Bhutta Z (1999) Protein: digestibility and availability. In: Sadler M, Strain J, Caballero B (eds) Encyclopedia of Human Nutrition. Academic Press, San Diego, pp 1646–1656
Biesalski HK, Nohr D (2009) The nutritional quality of meat. In: Kerry JP, Ledward D (eds) Improving the sensory and nutritional quality of fresh meat, 1st edn. Woodhead Publishing Ltd, England, Cambridge
Biesalski HK (2005) Meat as a component of a healthy diet – are there any risks or benefits if meat is avoided in the diet? Meat Sci 70:509–524
Borenstein B (1981) Vitamins and amino acids. In: Furia T (ed) Handbook of Food Additives, vol 1. CRC Press, Boca Raton, pp 85–114
Bou R, Guardiola F, Barroeta AC, Codony R (2005) Effect of dietary fat sources and zinc and selenium supplements on the composition and consumer acceptability of chicken meat. Poult Sci 84:1129–1140
Bouckenooghe T, Remacle C, Reusens B (2006) Is taurine a functional nutrient? Curr Opin Clin Nutr Metab Care 9:728–733
Brenna JT (2002) Efficiency of conversion of alpha-linolenic acid to long chain n-3 fatty acids in man. Curr Opin Clin Nutr Metab Care 5:127–132
Brooks WA, Santosham M, Naheed A, Goswami D, Wahed MA, Diener-West M et al (2005) Effect of weekly zinc supplements on incidence of pneumonia and diarrhoea in children younger than 2 years in an urban, low-income population in Bangladesh: randomized controlled trial. Lancet 366:999–1004
Caccialanza R, Laviano A, Lobascio F, Montagna E, Bruno R, Ludovisi S et al (2020) Early nutritional supplementation in non-critically ill patients hospitalized for the 2019 novel coronavirus disease (COVID-19): Rationale and feasibility of a shared pragmatic protocol. Nutrition 74:110835
Calder PC (2001) Polyunsaturated fatty acids, inflammation, and immunity. Lipids 36:1007–1024
Calder PC (2006) N-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am J Clinical Nutr 83:1505S-1519S
Calder PC (2012) Omega-3 polyunsaturated fatty acids and inflammatory processes: nutrition or pharmacology?: omega-3 fatty acids and inflammation. Br J Clin Pharmacol 75:645–662
Calder PC (2013) Feeding the immune system. Proc Nutr Soc 72:299–309
Calder PC (2020) Nutrition, immunity and COVID-19. BMJ Nutr Prev Health 3:74–92
Calder PCC, Gombart AF, Eggersdorfer M (2020) Optimal nutritional status for a well-functioning immune system is an important factor to protect against viral infections. Nutrients 12(4):1181
Cannell JJ, Vieth R, Umhau JC, Holick MF, Grant WB, Madronich S, Garland CF et al (2006) Epidemic influenza and vitamin D. Epidemiol Infect 134:1129–1140
Carr AC (2020) A new clinical trial to test high-dose vitamin C in patients with COVID-19. Crit Care 24:133
Carr AC, Maggini S (2017) Vitamin C and immune function. Nutrients 9:1211
Chaigne-Delalande B, Li F-Y, O’Connor GM, Lukacs MJ, Jiang P, Zheng L et al (2013) Mg2+ regulates cytotoxic functions of NK and CD8 T cells in chronic EBV infection through NKG2D. Science 341(6142):186e91
Chakurkar V, Rajapurkar M, Lele S, Mukhopadhyay B, Lobo V, Injarapu R, Sheikh M, Dholu B, Ghosh A, Jha V (2021) Increased serum catalytic iron may mediate tissue injury and death in patients with COVID-19. Sci Rep 11(1):19618. https://doi.org/10.1038/s41598-021-99142-x
Chan W, Brown J, Lee S, Buss DH (1995) Meat, poultry and game. Fifth supplement to Mccance & Widdowson’s the composition of foods. London: The Royal Society of Chemistry and the Ministry of Agriculture Fisheries and Food 40:161.
Charan J, Goyal JP, Saxena D, Yadav P (2012) Vitamin D for prevention of respiratory tract infections: a systematic review and meta-analysis. J Pharmacol Pharmacother 3:300–303
Chen Y, Chen W, Zhou J, Sun C, Lei Y (2021) Large pulmonary cavity in COVID-19 cured patient case report. Ann Palliat Med 9:5786–5791. https://doi.org/10.21037/apm-20-452
Cingolani V (2021) Hypothesis of zinc ascorbate as best zinc ionophore for raising antiviral resistance against Covid-19. J Med Virol 93(9):5205–5208. https://doi.org/10.1002/jmv.26989
Clemente-Suárez VJ, Ramos-Campo DJ, Mielgo-Ayuso J, Dalamitros AA, Nikolaidis PA, Hormeño-Holgado A, Tornero-Aguilera JF (2021) Nutrition in the actual COVID-19 pandemic. A narrative review. Nutrients 13(6):1924. https://doi.org/10.3390/nu13061924
Coquette A, Vray B, Vanderpas J (1986) Role of vitamin E in the protection of the resident macrophage membrane against oxidative damage. Arch Int Physiol Biochem 94:S29–S34
Decker EA, Park Y (2010) Healthier meat products as functional foods. Meat Sci 86:49–55
Ding H, Deng W, Ding L, Ye X, Yin S, Huang W (2020) Glycyrrhetinic acid and its derivatives as potential alternative medicine to relieve symptoms in nonhospitalized COVID-19 patients. J Med Virol 92:2200–2204. https://doi.org/10.1002/jmv.26064
Dokoupilová A, Marounek M, Skřivanová V, Březina P (2007) Selenium content in tissues and meat quality in rabbits fed selenium yeast. Czech J Animal Sci 52:165–169
Dragnes BT, Larsen R, Ernstsen MH, Maehre H, Elvevoll EO (2009) Impact of processing on the taurine content in processed seafood and their corresponding unprocessed raw materials. Int J Food Sci Nutr 60:143–152
Dushianthan A, Cusack R, Burgess VA, Grocott MP, Calder PC (2019) Immunonutrition for acute respiratory distress syndrome (ARDS) in adults. Cochrane Database Syst Rev 1(1):CD012041
EFSA Nda Panel (EFSA Panel on dietetic products, nutrition and allergies) (2015) Scientific opinion on dietary reference values for vitamin E as α-tocopherol. EFSA J 13:4149
El-Senousey HK, Chen B, Wang JY, Atta AM, Mohamed FR, Nie QH (2018) Effects of dietary vitamin C, vitamin E, and alpha-lipoic acid supplementation on the antioxidant defense system and immune-related gene expression in broilers exposed to oxidative stress by dexamethasone. Poult Sci 97(1):30–38
Esteve MJ, Farré R, Frìgola A, Pilamunga C (2002) Contents of vitamins B1, B2, B6, and B12 in pork and meat products. Meat Sci 62:73–78
EU 2020. EU Register on Nutrition and Health Claims. 2020. https://ec.europa.eu/food/safety/labelling_nutrition/claims/register/public/?event=search. Accessed 5th March, 2020).
Faa G, Saba L, Fanni D, Kalcev G, Carta M (2021) Association between hypomagnesemia, covid-19, respiratory tract and lung disease. Open Respir Med J 15:43–45. https://doi.org/10.2174/1874306402115010043
Fan X, Liu S, Liu G, Zhao J, Jiao H, Wang X et al (2015) Vitamin A deficiency impairs mucin expression and suppresses the mucosal immune function of the respiratory tract in chicks. PLoS One 10(9):e0139131
Fanni D, Gerosa C, Nurchi VM, Manchia M, Saba L, Coghe F, Crisponi G, Gibo Y, Van Eyken P, Fanos V, Faa G (2020) The role of magnesium in pregnancy and in fetal programming of adult diseases. Biological Trace Element Res. https://doi.org/10.1007/s12011-020-02513-0
Fernández-Quintela A, Milton-Laskibar I, Trepiana J, Gomez-Zorita S, Kajarabille N, Leniz A, Gonzalez M, Portillo MP (2020) Key aspects in nutritional management of COVID-19 patients. J Clinc Medicine 9:2589
Food Standards Australia New Zealand (2002) Food Standards Code, vol 2. Information Australia, Canberra
Fox TE, Van den Heuvel E, Atherton CA, Dainty JR, Lewis DJ, Langford NJ et al (2004) Bioavailability of selenium from fish, yeast and selenate: a comparative study in humans using stable isotopes. Eur J Clin Nutr 58:343–349
Gao Y, Zhang H, Luo L, Lin J, Li D, Zheng S et al (2017) Resolvin D1 improves the resolution of inflammation via activating NF-B p50/p50–mediated cyclooxygenase-2 expression in acute respiratory distress syndrome. J Immunol 199:2043–2054
Gleeson M (2013) Exercise, nutrition and immunity. In: Calder PC, Yaqoob P (eds) Diet, Immunity and Inflammation. Woodhead Publishing, Cambridge, pp 652–685
Gleeson M, Nieman DC, Pedersen BK (2004) Exercise, nutrition and immune function. J Sports Sci 22:115–125
Gombart AF (2009) The vitamin D–antimicrobial peptide pathway and its role in protection against infection. Future Microbiol 4:1151–1165
Gombart AF, Borregaard N, Koeffler HP (2005) Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1, 25-dihydroxyvitamin D3. FASEB J 19:1067–1077
Gombart AF, Pierre A, Maggini S (2014) The immune system in children with malnutrition—a systematic review. PLoS One 9(8):e105017
Gozzi-Silva SC, Teixeira FME, Duarte AJDS, Sato MN, Oliveira LM (2021) Immunomodulatory role of nutrients: how can pulmonary dysfunctions improve? Front Nutr 8:674258. https://doi.org/10.3389/fnut.2021.674258
Grant WB, Lahore H, McDonnell SL, Baggerly CA, French CB, Aliano JL, Bhattoa HP (2020) Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients 12:988
Greiller C, Martineau A (2015) Modulation of the immune response to respiratory viruses by vitamin D. Nutrients 7:4240–4270
Guillin OM, Vindry C, Ohlmann T, Chavatte L (2019) Selenium, selenoproteins and viral infection. Nutrients 11:2101
Habib HM, Ibrahim S, Zaim A, Ibrahim WH (2021) The role of iron in the pathogenesis of COVID-19 and possible treatment with lactoferrin and other iron chelators. Biomed Pharmacother 136:111228. https://doi.org/10.1016/j.biopha.2021.111228
Haddad EH, Berk LS, Kettering JD, Hubbard RW, Peters WR (1999) Dietary intake and biochemical, hematologic, and immune status of vegans compared with nonvegetarians. Am J Clin Nutr 70(1):S586–S593
Hallberg L, Hulthen L (2000) Prediction of dietary iron absorption: An algorithm for calculating absorption and bioavailability of dietary iron. Am J Clin Nutr 71:1147–1160
Han YS, Chang GG, Juo CG, Lee HJ, Yeh SH, Hsu JT et al (2005) Papain-like protease 2 (PLP2) from severe acute respiratory syndrome coronavirus (SARS-CoV): expression, purification, characterization, and inhibition. Biochem 44:10349–10359
Hemila H (1997) Vitamin C intake and susceptibility to pneumonia. Pediatr Infect Dis J 16:836–847
Hemilä H (2003) Vitamin C and SARS coronavirus. J Antimicrob Chemother 52:1049–1050
Hemila H (2017) Vitamin C and infections. Nutrients 9:339
Hemilä H, Chalker E (2013) Vitamin C for preventing and treating the common cold. Cochrane Database Syst Rev 1:CD000980
Hemilä H, Chalker E (2019) Vitamin C can shorten the length of stay in the ICU: a meta-analysis. Nutrients 11:E708
Hemilä H, Chalker E (2020) Vitamin C may reduce the duration of mechanical ventilation in critically ill patients: a meta-regression analysis. J Intensive Care 8:15
Hemilä H, Douglas RM (1999) Vitamin C and acute respiratory infections. Int J Tuberc Lung Dis 3:756–761
Hemila H, Louhiala P (2013) Vitamin C for preventing and treating pneumonia. Cochrane Database Syst Rev 8:CD005532
Hernàndez P, Dalle Zotte A (2010) Influence of diet on rabbit meat quality. pp 163– 178. In: de Blas C (ed) Nutrition of the rabbit. Univesidad Poletenica, Madrid, J. Wiseman, University of Nottingham, UK
Hess SY, Lönnerdal B, Hotz C, Rivera JA, Brown KH (2009) Recent advances in knowledge of zinc nutrition and human health. Food Nutr Bull 30:S5–S11
Hiedra R, Lo KB, Elbashabsheh M, Gul F, Wright RM, Albano J, Azmaiparashvili Z, Patarroyo Aponte G (2020) The use of i/v vitamin C for patients with COVID-19: a single center observational study. Expert Rev Anti-Infect Ther 18:1259–1261. https://doi.org/10.1080/14787210.2020.1794819
Holick MF (2008) The vitamin D deficiency pandemic and consequences for nonskeletal health: Mechanisms of action. Mol Aspects Med 29:361–368
Holland B, Brown J, Bush DH (1993) Fish and fish products. In: The fifth supplement of McCance and Widdowson’s, the composition of foods. Royal Society of Chemistry, Cambridge/London
Hopkins RG, Failla ML (1997) Copper deficiency reduces interleukin-2 (IL-2) production and IL-2 mRNA in human T-lymphocytes. J Nutr 127:257–262
Howe P, Meyer B, Record S, Baghurst K (2006) Dietary intakes of long-chain omega-3 polyunsaturated fatty acids: Contribution of meat sources. Nutr 22:47–53
Huxtable RJ (1992) Physiological actions of taurine. Physiol Rev 72:101–163
Ilie PC, Stefanescu S, Smith L (2020) The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin Exp Res 32:1195–1208. https://doi.org/10.1007/s40520-020-01570-8
Jacob RA, Kelley DS, Pianalto FS, Swendseid ME, Henning SM, Zhang JZ, Ames BN, Fraga CG, Peters JH (1991) Immunocompetence and oxidant defense during ascorbate depletion of healthy men. Am J Clin Nutr 54:1302S-1309S
Jee J, Hoet AE, Azevedo MP, Vlasova AN, Loerch SC, Pickworth CL, Hanson J, Saif LJ (2013) Effects of dietary vitamin A content on antibody responses of feedlot calves inoculated intramuscularly with an inactivated bovine coronavirus vaccine. Am J Vet Res 74(10):1353–1062
Jollie DA, Griths CJ, Martineau AR (2013) Vitamin D in the prevention of acute respiratory infection: systematic review of clinical studies. J Steroid Biochem Mol Biol 136:321–329
Jose N, Elangovan AV, Awachat VB, Shet D, Ghosh J, David CG (2017) Response of in ovo administration of zinc on egg hatchability and immune response of commercial broiler chicken. J Anim Physiol Anim Nutr (berl) 102(2):591–595
Juniper DT, Phipps RH, Ramos-Morales E, Bertin G (2008) Effect of dietary supplementation with selenium-enriched yeast or sodium selenite on selenium tissue distribution and meat quality in beef cattle. J Animal Sci 86(11):3100–3109
Kajanachumpol S, Srisurapanon S, Supanit I, Roongpisuthipong C, Apibal S (1995) Effect of zinc supplementation on zinc status, copper status and cellular immunity in elderly patients with diabetes mellitus. J Med Assoc Thai 78:344–349
Kataoka H, Ohnishi N (1986) Occurrence of taurine in plants. Agric Biol Chem Tokyo 50:1887–1898
Khodavirdipour A (2021) Inclusion of Cephalexin in COVID-19 treatment combinations may prevent lung involvement in mild infections: a case report with pharmacological genomics perspective. Glob Med Genet 8(2):78–81. https://doi.org/10.1055/s-0041-1726461
Khodour Y, Kaguni LS, Stiban J (2019) Iron-sulfur clusters in nucleic acid metabolism: varying roles of ancient cofactors. Enzymes 45:225–256. https://doi.org/10.1016/bs.enz.2019.08.003
Kinoshita M, Kayama H, Kusu T, Yamaguchi T, Kunisawa J, Kiyono H et al (2012) Dietary folic acid promotes survival of Foxp3+ regulatory T cells in the colon. J Immunol 189:2869–2878
Konieczka P, Czauderna M, Smulikowska S (2017) The enrichment of chicken meat with omega-3 fatty acids by dietary fish oil or its mixture with rapeseed or flaxseed – effect of feeding duration. Animal Feed Sci Technol 223:42–52
Kunisawa J, Kiyono H (2013) Vitamin-mediated regulation of intestinal immunity. Front Immunol 4:189
LARN (1996) Livelli di Assunzione Raccomandati di Energia e Nutrienti per la Popolazione Italiana. SINU, Rome, pp 1–185
Larsen L, Eilertsen K, Elvevoll EO (2011) Health benefits of marine foods and ingredients. Biotechnol Adv 29:508–518
Lee C, Lee JM, Lee NR, Jin BS, Jang KJ, Kim DE, Jeong YJ, Chong Y (2009) Aryl diketoacids (ADK) selectively inhibit duplex DNA-unwinding activity of SARS coronavirus NTPase/helicase. Bioorg Med Chem Lett 19(6):1636–1638. https://doi.org/10.1016/j.bmcl.2009.02.010
Li C, Li Y, Ding C (2019) The role of copper homeostasis at the host-pathogen Axis: from bacteria to fungi. Int J Mol Sci 20(1):175
Li P, Yin YL, Li D, Kim SW, Wu G (2007) Amino acids and immune function. Br J Nutr 98(2):237–252
Liang RY, Wu W, Huang J, Jiang SP, Lin Y (2012) Magnesium affects the cytokine secretion of CD4(+) T lymphocytes in acute asthma. J Asthma 49(10):1012–1015
Liu W, Zhang S, Nekhai S, Liu S (2020) Depriving Iron supply to the virus represents a promising adjuvant therapeutic against viral survival. Curr Clin Microbiol Rep 20:1–7. https://doi.org/10.1007/s40588-020-00140-w
Lomax AR, Calder PC (2009) Prebiotics, immune function, infection and inflammation: a review of the evidence. Br J Nutr 101:633–658
Lombardi-Boccia G, Lanzi S, Aguzzi A (2005) Aspects of meat quality: trace elements and B vitamins in raw and cooked meats. J Food Compost Anal 18(1):39–46
Lombardi-Boccia G, Martinez-Dominguez B, Aguzzi A (2002) Total heme and non heme iron in raw and cooked meats. J Food Sci 67(5):1738–1741
Maggini S, Pierre A, Calder PC (2018) Immune function and micronutrient requirements change over the life course. Nutrients 10(10):1–10
Maggini S, Wenzlaff S, Hornig D (2010) Essential role of vitamin C and zinc in child immunity and health. J Int Med Res 38:386–414
Maggini S, Wintergerst ES, Beveridge S, Hornig DH (2007) Selected vitamins and trace elements support immune function by strengthening epithelial barriers and cellular and humoral immune responses. Br J Nutr 98(1):S29–S35
Marmer W, Maxwell R, Williams J (1984) Effects of dietary regimen and tissue site on bovine fatty acid profiles. J Anim Sci 59(1):109–121
Martineau AR, Jolliffe DA, Hooper RL, Greenberg L, Aloia JF et al (2017) Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data. BMJ 356:i6583. https://doi.org/10.1136/bmj.i6583
McCarty MF, DiNicolantonio JJ (2020) Nutraceuticals have potential for boosting the type 1 interferon response to RNA viruses including influenza and coronavirus. Prog Cardiovasc Dis 63(3):383–385
Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ (2020) HLH Across Speciality Collaboration, UK. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet 395:1033–1034
Merzon E, Tworowski D, Gorohovski A, Vinker S, Golan Cohen A, Green I, Frenkel-Morgenstern M (2020) Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: an Israeli population-based study. FEBS J 287:3693–3602. https://doi.org/10.1111/febs.15495
Meydani SN, Han SN, Wu D (2005) Vitamin E and immune response in the aged: molecular mechanisms and clinical implications. Immunol Rev 205:269–284
Meydani SN, Leka LS, Fine BC, Dallal GE, Keusch GT, Singh F et al (2004) Vitamin E and respiratory tract infections in elderly nursing home residents: a randomized controlled trial. JAMA 292:828–836
Mocchegiani E, Giacconi R, Muzzioli M, Cipriano C et al (2000) Zinc, infections and immunosenescence. Mech Ageing Dev 121:21–35
Mocchegiani E, Muzzioli M, Gaetti R, Veccia S, Viticchi C, Scalise G (1999) Contribution of zinc to reduce CD4+ risk factor for “severe” infection relapse in aging: Parallelism with HIV. Int J Immunopharmacol 21:271–281
Mora JR, von Andrian UH (2006) T-cell homing specificity and plasticity: new concepts and future challenges. Trends Immunol 27:235–243
Munoz C, Rios E, Olivos J, Brunser O, Olivares M (2007) Iron, copper and immunocompetence. Br J Nutr 98(1):S24–S38
Munshi R, Hussein MH, Toraih EA, Elshazli RM, Jardak C, Sultana N et al (2020) Vitamin D insufficiency as a potential culprit in critical COVID-19 patients. J Med Virol 93(2):733–740. https://doi.org/10.1002/jmv.26360
Murni IK, Prawirohartono EP, Triasih R (2021) Potential role of vitamins and zinc on acute respiratory infections including COVID-19. Glob Pediatr Health 8:2333794X211021739. https://doi.org/10.1177/2333794X211021739
Muscogiuri G, Altieri B, Annweiler C, Balercia G, Pal B, Boucher J et al (2017) Vitamin D and chronic diseases: the current state of the art. Arch Toxicol 91:97–107
National Health and Medical Research Council (2006) Nutrient reference values for Australia and New Zealand including recommended dietary intakes. Commonwealth Department of Health and Ageing, Canberra
Norman AW (2008) From vitamin D to hormone D: fundamentals of the vitamin D endocrine system essential for good health. Am J Clin Nutr 88:491S-499S
Overbeck S, Rink L, Haase H (2008) Modulating the immune response by oral zinc supplementation: a single approach for multiple diseases. Arch Immunol Ther Exp (warsz) 56:15–30
Ovesen L, Brot C, Jakobsen J (2003) Food contents and biological activity of 25- hydroxyvitamin D: a vitamin D metabolite to be reckoned with? Ann Nutr Metab 47:107–113
Pae M, Meydani SN, Wu D (2012) The role of nutrition in enhancing immunity in aging. Aging Dis 3:91–129
Pan S, Zhang K, Ding X, Wang J, Peng H, Zeng Q, Xuan Y, Su Z, Wu B, Bai S (2018) Effect of high dietary manganese on the immune responses of broilers following oral Salmonella typhimurium inoculation. Biol Trace Elem Res 181(2):347–360
Pedersen SF, Ho YC (2020) SARS-CoV-2: a storm is raging. J Clin Investig 130(5):2202–2205
Percival SS (1998) Copper and immunity. Am J Clin Nutr 67:1064S-1078S
Perricone C, Bartoloni E, Bursi R, Cafaro G, Guidelli GM, Shoenfeld Y, Gerli R (2020) COVID-19 as part of the hyperferritinemic syndromes: the role of iron depletion therapy. Immunol Res 68(4):213–224. https://doi.org/10.1007/s12026-020-09145-5
Prasad A, Fitzgerald J T, Hess JW, Kaplan J, Pelen F, Dardenne M (1993) Zinc deficiency in elderly patients. Nutrition:218–224
Prasad AS (2008) Zinc in human health: effect of zinc on immune cells. Mol Med 14:353–357
Prasad AS, Fitzgerald JT, Bao B, Beck FW, Chandrasekar PH (2000) Duration of symptoms and plasma cytokine levels in patients with the common cold treated with zinc acetate. A randomized, double-blind, placebo-controlled trial. Ann Intern Med 133:245–252. https://doi.org/10.7326/0003-4819-133-4-200008150-00006
Purchas R, Rutherfurd S, Pearce P, Vather R, Wilkinson BHP (2004) Concentrations in beef and lamb of taurine, carnosine, coenzyme Q10, and creatine. Meat Sci 66:629–637
Purohit D, Pandey P, Makhija M, Manchanda D, Rathi J, Kumar D, Verma R, Jalwal P, Mittal V, Kaushik D (2020a) Correlation of risk perception with the COVID-19 related knowledge and preventive measures: a study on Indian pharmacy students. Int J Curr Res Rev 13(3):113–119
Purohit D, Saini M, Pathak N, Verma R, Kaushik D, Katiyar P, Jalwal P, Pandey P (2020b) COVID-19 ‘The pandemic’: an update on the present status of the outbreak and possible treatment options. Biomed Pharmacol J 13(4):1791–1807
Rahaa S, Mallickb R, Basakc S, Duttaroy KA (2020) Is copper beneficial for COVID-19 patients? Med Hypotheses 142:109814
Ralston NVC (2008) Selenium health benefit values as seafood safety criteria. EcoHealth 5:442–455
Rayman MP (2012) Selenium and human health. Lancet 379(9822):1256–1268
Read SA, Obeid S, Ahlenstiel C, Ahlenstiel G (2019) The role of zinc in antiviral immunity. Adv Nutr 10(4):696–710
Reber E, Gomes F, Vasiloglou MF, Schuetz P, Stanga Z (2019) Nutritional risk screening and assessment. J Clin Med 8(7):1065
Reilly C (1998) Selenium: a new entrant into the functional food arena. Trends Food Sci Technol 9:114–118
Ribeiro T, Lordelo MM, Alves SP, Bessa RJ, Costa P, Lemos JP, Ferreira LM, Fontes CM, Prates JA (2013) Direct supplementation of diet is the most efficient way of enriching broiler meat with n-3 long-chain polyunsaturated fatty acids. Br Poult Sci 54:753–765
Riediger ND, Othman RA, Suh M, Moghadasian MH (2009) A systemic review of the roles of n- 3 fatty acids in health and disease. J Am Diet Assoc 109:668–679
Romeo AM, Christen L, Niles EG, Kosman DJ (2001) Intracellular chelation of iron by bipyridyl inhibits DNA virus replication: ribonucleotide reductase maturation as a probe of intracellular iron pools. J Biol Chem 276:24301–24308. https://doi.org/10.1074/jbc.M010806200
Rytter MJ, Kolte L, Briend A, Friis H, Christensen VB (2020) A review of micronutrients and the immune system-working in harmony to reduce the risk of infection. Nutrients 12(1):236. https://doi.org/10.3390/nu12010236
Sakaguchi S, Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T (2009) Regulatory T cells: how do they suppress immune responses? Int Immunol 21:1105–1111
Schaafsma G (2000) The protein digestibility-corrected amino acid score. J Nutr 130:1865S-1867S
Schloerb PR (2001) Immune-enhancing diets: products, components, and their rationales. JPEN J Parenter Enteral Nutr 25(2):S3–S7. https://doi.org/10.1177/014860710102500202
Shankar AH, Prasad AS (1998) Zinc and immune function: the biological basis of altered resistance to infection. Am J Clin Nutr 6:447S-463S
Simpson RJ, McKie AT (2009) Regulation of intestinal iron absorption: the mucosa takes control? Cell Metab 10(2):84–87
Sinclair A, Mann N, O’Connell S (1999) The nutrient composition of Australian beef and lamb. RMIT, Melbourne
Sinclair A, O’Dea K (1987) The lipid levels and fatty acid compositions of the lean portions of Australian beef and lamb. Food Technol Aust 39:228–231
Smith AD, Panickar KS, Urban JF, Dawson HD (2018) Impact of micronutrients on the immune response of animals. Annu Rev Anim Biosci 6:227–254
Spitze AR, Wong DL, Rogers QR, Fascetti AJ (2003) Taurine concentrations in animal feed ingredients; cooking influences taurine content. J Anim Physiol Anim Nutr 87:251–262
Tacon AGJ, Metian M (2013) Fish matters: importance of aquatic foods in human nutrition and global food supply. Rev Fisher Sci 21:22–38
Tan CW, Ho LP, Kalimuddin S, Cherng BPZ (2020) A cohort study to evaluate the effect of combination vitamin D, magnesium and vitamin B12 (DMB) on progression to severe outcome in older COVIDS-19 patients. medRxiv. https://doi.org/10.1101/2020.06.01.20112334
Tan CW, Ho LP, Kalimuddin S, Cherng BPZ, Teh YE, Thien SY, Wong HM, Tern PJW, Chandran M, Chay JWM, Nagarajan C, Sultana R, Low JGH, Ng HJ (2020b) Cohort study to evaluate the effect of vitamin D, magnesium, and vitamin B12 in combination on progression to severe outcomes in older patients with coronavirus (COVID-19). Nutrition 79–80:111017. https://doi.org/10.1016/j.nut.2020.111017
Teymoori-Rad M, Shokri F, Salimi V, Marashi SM (2019) The interplay between vitamin D and viral infections. Rev Med Virol 29:e2032
USDA (2011) USDA National Nutrient Database for Standard Reference. http://www.ars.usda.gov/nutrientdata. Accessed 22nd December, 2020
Valdés-Ramos R, Martínez-Carrillo BE, Aranda-González II, Guadarrama AL, Pardo-Morales RV, Tlatempa P et al (2010) Diet, exercise and gut mucosal immunity. Proc Nutr Soc 69:644–650
Van Doremalen N, Bushmaker T, Morris DH, Holbrook MG, Gamble A, Williamson BN et al (2020) Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV- 1. N Engl J Med 382(16):1564–1656
Walker CF, Black RE (2004) Zinc and the risk for infectious disease. Annu Rev Nutr 24:255–275
Wang JZ, Zhang RY, Bai J (2020) An anti-oxidative therapy for ameliorating cardiac injuries of critically ill COVID-19-infected patients. Int J Cardiol 312:137–138
Wang TT, Dabbas B, Laperriere D, Bitton AJ, Soualhine H, Tavera-Mendoza LE et al (2010) Direct and indirect induction by 1, 25-dihydroxyvitamin D3 of the NOD2/CARD15-defensin β2m innate immune pathway defective in Crohn disease. J Biol Chem 285:2227–2231
Waqas Khan HM, Parikh N, Megala SM, Predeteanu GS (2020) Unusual early recovery of a critical COVID-19 patient after administration of intravenous vitamin C. Am J Case Rep 21:1–6
Watanabe F, Katsura H, Takenaka S, Enomoto T, Miyamoto E, Nakatsuka T, Nakano Y (2001) Characterization of vitamin B12 compounds from edible shellfish, clam, oyster, and mussel. Int J Food Sci Nutr 52:263–268
WHO 2020a. World Health Organization. Off-label use of medicines for COVID-19 (2020a). https://www.who.int/newsroom/commentaries/detail/o-label-use-of-medicines-for-covid-19. Accessed 22nd December, 2020a
WHO 2020a. World Health Organization. Coronavirus. 2020b. https://www.who.int/healthtopics/coronavirus. Accessed 22nd December, 2020b
Williams P (2007a) Nutritional composition of red meat. Nutr Dietetics 64:S113–S119
Williams P (2007b) Section 2: key nutrients delivered by red meat in the diet. Nutr Dietetics 64(4):S113–S119
Williams P, Droulez V, Levy G, Stobaus T (2007) Composition of Australian red meat 2002. 3. Nutrient profile. Food Aust 59:331–341
Wójcik OP, Koenig KL, Zeleniuch-Jacquotte A, Costa M, Chen Y (2010) The potential protective effects of taurine on coronary heart disease. Atherosclerosis 208:19–25
Wood JD, Enser M, Fisher AV, Nute GR, Sheard PR, Richardson RI et al (2008) Fat deposition, fatty acid composition and meat quality: a review. Meat Sci 78:343–358
Wu D, Lewis ED, Pae M, Meydani SN (2019) Nutritional modulation of immune function: analysis of evidence, mechanisms, and clinical relevance. Front Immunol 15(9):3160
Wu JZ, Zha P (2020) Treatment strategies for reducing damages to lungs in patients with coronavirus and other infections. Preprints 2020020116
Wyness L, Weichselbaum E, O’Connor A, Williams EB, Benelam B, Riley H, Stanner S (2011) Red meat in the diet: an update. Nutr Bulletin 36:34–77
Xu F, Wen Y, Hu X, Wang T, Chen G (2021) The potential use of vitamin C to prevent kidney injury in patients with COVID-19. Diseases 9(3):46. https://doi.org/10.3390/diseases9030046
Yao JS, Paguio JA, Dee EC, Tan HC, Moulick A, Milazzo C, Jurado J, Della Penna N, Celi LA (2020) The minimal effect of zinc on the survival of hospitalized patients with COVID-19: an observational study. Chest 59(1):108–111. https://doi.org/10.1016/j.chest.2020.06.082
Yaqoob P (2017) Ageing alters the impact of nutrition on immune function. Proc Nutr Soc 76:347–351
Zabetakis I, Lordan R, Norton C, Tsoupras A (2020) COVID-19: the inflammation link and the role of nutrition in potential mitigation a review. Nutrients 12:1466
Zhang HW, Wang Q, Mei HX, Zheng SX, Ali AM, Wu QX et al (2019) RvD1 ameliorates LPS-induced acute lung injury via the suppression of neutrophil infiltration by reducing CXCL2 expression and release from resident alveolar macrophages. Int Immunopharmacol 76:105877
Zhang W, Xiao S, Samaraweera H, Lee EJ, Ahn DU (2010) Improving functional value of meat products. A review. Meat Sci 86:15–31
Zhao B, Ling Y, Li J, Peng Y, Huang J, Wang Y, Qu H, Gao Y et al (2020) Beneficial aspects of high dose intravenous vitamin C on patients with covid-19 pneumonia in severe condition: a retrospective case series study. Ann Palliat Med 9:1–11. https://doi.org/10.21037/apm-20-1387
Zheng YY, Ma YT, Zhang JY, Xie X (2020) COVID-19 and the cardiovascular system. Nature Rev Cardiol 17:259–260
Author information
Authors and Affiliations
Contributions
Gaber El-Saber Batiha: writing — original draft preparation, conceptualization; Ali I. Al-Gareeb: writing — review and editing; Safaa Qusti: writing — review and editing; Eida M. Alshammari: writing — review and editing; Deepak Kaushik: conceptualization, supervision; Ravinder Verma: writing — original draft preparation; Hayder M. Al- kuraishy: writing — review and editing.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Resposible Editor: Lotfi Aleya
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Batiha, G.ES., Al-Gareeb, A.I., Qusti, S. et al. Deciphering the immunoboosting potential of macro and micronutrients in COVID support therapy. Environ Sci Pollut Res 29, 43516–43531 (2022). https://doi.org/10.1007/s11356-022-20075-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-20075-7