Abstract
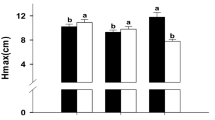
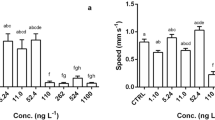
Atrazine, a common chemical pesticide, has toxicity to adult and juvenile amphibians in natural ecosystems; however, it is more common to study its effects on larvae instead of adults. This study assessed the impacts of atrazine in water through short-term exposure (7 days) on male black spotted frog (Pelophylax nigromaculatus) adults fed every day. The jumping ability, including jumping height, distance, time, and speed, was measured by 3D motion analysis software, and the intestinal content microbiota was determined by 16S rRNA amplicon sequencing with QIIME software. The results showed that male P. nigromaculatus exposure to 200 and 500 μg/L atrazine significantly increased jumping distance and jumping time compared to control groups. Conversely, 500 μg/L atrazine treatments significantly decreased the diversity and changed the composition and structure of intestinal content microflora in male P. nigromaculatus compared to control groups. At the phylum level, Chlamydiae was only detected in the control group, and Actinobacteria, Bacteroidetes, Firmicutes, Fusobacteria, and Proteobacteria were the dominant microflora in the atrazine treatment groups. At the genus level, the abundance of Lactobacillus and Weissella significantly increased in atrazine treatment groups compared to control groups. This study can provide a new framework based on movement behavior and intestinal microbiota to evaluate the response of amphibians to short-term exposure to environmental pollution.







Similar content being viewed by others
Data availability
The data used to support the results of the present research work can be obtained from the corresponding author on demand.
References
Bachmann NL, Polkinghorne A, Timms P (2014) Chlamydia genomics: providing novel insights into chlamydial biology. Trends Microbiol 22:464–472. https://doi.org/10.1016/j.tim.2014.04.013
Bastidas RJ, Elwell CA, Engel JN, Valdivia RH (2013) Chlamydial intracellular survival strategies. Cold Spring Harb Perspect Med 3:a010256. https://doi.org/10.1101/cshperspect.a010256
Bäumler AJ, Sperandio V (2016) Interactions between the microbiota and pathogenic bacteria in the gut. Nature. 535:85–93. https://doi.org/10.1038/nature18849
Böhme L, Albrecht M, Riede O, Rudel T (2010) Chlamydia trachomatis-infected host cells resist dsRNA-induced apoptosis. Cell Microbiol 12:1340–1351. https://doi.org/10.1111/j.1462-5822.2010.01473.x
Britson CA, Threlkeld ST (1998) Abundance, metamorphosis, developmental, and behavioral abnormalities in Hyla chrysoscelis tadpoles following exposure to three agrichemicals and methyl mercury in outdoor mesocosms. Bull Environ Contam Toxicol 61:154–161. https://doi.org/10.1007/s001289900742
Brodeur JC, Sassone A, Hermida GN, Codugnello N (2013) Environmentally-relevant concentrations of atrazine induce non-monotonic acceleration of developmental rate and increased size at metamorphosis in Rhinella arenarum tadpoles. Ecotoxicol Environ Saf 92:10–17. https://doi.org/10.1016/j.ecoenv.2013.01.019
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Peña AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336. https://doi.org/10.1038/nmeth.f.303
Chao A (1984) Nonparametric estimation of the number of classes in a population. Scand J Stat 11:265–270. https://doi.org/10.2307/4615964
Chen L, Zhang W, Hua J, Hu C, Lok-Shun, Lai N, Qian PY, Lam PKS, Lam JCW, Zhou B (2018) Dysregulation of intestinal health by environmental pollutants: involvement of the estrogen receptor and aryl hydrocarbon receptor. Environ Sci Technol 52:2323–2330. https://doi.org/10.1021/acs.est.7b06322
Chiu K, Warner G, Nowak RA, Flaws JA, Mei W (2020) The impact of environmental chemicals on the gut microbiome. Toxicol Sci 176(2):253–284. https://doi.org/10.1093/toxsci/kfaa065
Choi JJ, Eum SY, Rampersaud E, Daunert S, Abreu MT, Toborek M (2013) Exercise attenuates PCB-induced changes in the mouse gut microbiome. Environ Health Perspect 121:725–730. https://doi.org/10.1289/ehp.1306534
Chumduri C, Gurumurthy RK, Zadora PK, Mi Y, Meyer TF (2013) Chlamydia infection promotes host DNA damage and proliferation but impairs the DNA damage response. Cell Host Microbe 13:746–758. https://doi.org/10.1016/j.chom.2013.05.010
Das PC, McElroy WK, Cooper RL (2001) Alteration of catecholamines in pheochromocytoma (pc12) cells in vitro by the metabolites of chlorotriazine herbicide. Toxicol Sci 59:127–137. https://doi.org/10.1093/toxsci/59.1.127
Dechartres J, Pawluski JL, Gueguen MM, Jablaoui A, Maguin E, Rhimi M, Charlier TD (2019) Glyphosate and glyphosate-based herbicide exposure during the peripartum period affects maternal brain plasticity, maternal behaviour and microbiome. J Neuroendocrinol 31(9):e12731. https://doi.org/10.1111/jne.12731
Dornelles MF, Oliveira GT (2016) Toxicity of atrazine, glyphosate, and quinclorac in bullfrog tadpoles exposed to concentrations below legal limits. Environ Pollut Res Int 23:1610–1620. https://doi.org/10.1007/s11356-015-5388-4
Fei L, Hu SQ, Ye CY, Huang YZ (2009) Fauna sinica, amphibia Vol. 3, Anura ranidae. Science Press, Beijing
Fouts DE, Szpakowski S, Purushe J, Torralba M, Waterman RC, MacNeil MD, Alexander LJ, Nelson KE (2012) Next generation sequencing to define prokaryotic and fungal diversity in the bovine rumen. PLoS One 7:e48289. https://doi.org/10.1371/journal.pone.0048289
Graymore M, Stagnitti F, Allinson G (2001) Impacts of atrazine in aquatic ecosystems. Environ Int 26:483–495. https://doi.org/10.1016/s0160-4120(01)00031-9
Hawley JA (2020) Microbiota and muscle highway - two way traffic. Nat Rev Endocrinol 16:71–72. https://doi.org/10.1038/s41574-019-0291-6
Hayes TB, Collins A, Lee M, Mendoza M, Noriega N, Stuart AA, Vonk A (2002) Hermaphroditic, demasculinized frogs after exposure to the herbicide atrazine at low ecologically relevant doses. Proc Natl Acad Sci U S A 99:5476–5480. https://doi.org/10.1073/pnas.082121499
Herrel A, Vasilopouloukampitsi M, Bonneaud C (2014) Jumping performance in the highly aquatic frog, Xenopus tropicalis: sex-specific relationships between morphology and performance. PeerJ. 2:e661. https://doi.org/10.7717/peerj.661
Huang MY, Duan RY, Ji X (2014) Chronic effects of environmentally-relevant concentrations of lead in Pelophylax nigromaculata tadpoles: threshold dose and adverse effects. Ecotoxicol Environ Saf 310-316:310–316. https://doi.org/10.1016/j.ecoenv.2014.03.027
Huang WC, Chen YH, Chuang HL, Chiu CC, Huang CC (2019) Investigation of the effects of microbiota on exercise physiological adaption, performance, and energy utilization using a gnotobiotic animal model. Front Microbiol 10:1906. https://doi.org/10.3389/fmicb.2019.01906
Ji Q, Lee J, Lin YH, Jing G, Tsai LJ, Chen A, Hetrick L, Jocoy D, Liu J (2016) Atrazine and malathion shorten the maturation process of Xenopus laevis oocytes and have an adverse effect on early embryo development. Toxicol in Vitro 32:63–69. https://doi.org/10.1016/j.tiv.2015.12.006
Johnson JA, Ihunwo AO, Chimuka L, Mbajiorgu EF (2019) Cardiotoxicity in African clawed frog (Xenopus laevis) sub-chronically exposed to environmentally relevant atrazine concentrations: Implications for species survival. Aquat Toxicol 213:105218. https://doi.org/10.1016/j.aquatox.2019.06.001
Kamdar K, Khakpour S, Chen J, Leone V, Brulc J, Mangatu T, Antonopoulos DA, Chang EB, Kahn SA, Kirschner BS, Young G, DePaolo RW (2016) Genetic and metabolic signals during acute enteric bacterial infection alter the microbiota and drive progression to chronic inflammatory disease. Cell Host Microbe 19:21–31. https://doi.org/10.1016/j.chom.2015.12.006
Knutie SA, Gabor CR, Kohl KD, Rohr JR (2018) Do host-associated gut microbiota mediate the effect of a herbicide on disease risk in frogs? J Anim Ecol 87:489–499. https://doi.org/10.1111/1365-2656.12769
Kohl KD, Cary TL, Karasov WH, Dearing MD (2015) Larval exposure to polychlorinated biphenyl 126 (PCB-126) causes persistent alteration of the amphibian gut microbiota. Environ Toxicol Chem 34:1113–1118. https://doi.org/10.1002/etc.2905
Martin AM, Sun EW, Keating DJ (2019) Mechanisms controlling hormone secretion in human gut and its relevance to metabolism. J Endocrinol 244:R1–R15. https://doi.org/10.1530/JOE-19-0399
Moreno-Navarrete JM, Fernandez-Real JM (2019) The gut microbiota modulates both browning of white adipose tissue and the activity of brown adipose tissue. Rev Endocr Metab Disord 20:387–397. https://doi.org/10.1007/s11154-019-09523-x
Mu D, Meng J, Bo X, Wu M, Xiao H, Wang H (2018) The effect of cadmium exposure on diversity of intestinal microbial community of Rana chensinensis tadpoles. Ecotoxicol Environ Saf 154:6–12. https://doi.org/10.1016/j.ecoenv.2018.02.022
Oberauner L, Zachow C, Lackner S, Högenauer C, Smolle KH, Berg G (2013) The ignored diversity: complex bacterial communities in intensive care units revealed by 16s pyrosequencing. Sci Rep 3:1413. https://doi.org/10.1038/srep01413
Ojekunle O, Banwo K, Sanni AI (2017) In vitro and in vivo evaluation of Weissella cibaria and Lactobacillus plantarum for their protective effect against cadmium and lead toxicities. Lett Appl Microbiol 64:379–385. https://doi.org/10.1111/lam.12731
Papaefthimiou C, Zafeiridou G, Topoglidi A, Chaleplis G, Zografou S, Theophilidis G (2003) Triazines facilitate neurotransmitter release of synaptic terminals located in hearts of frog (Rana ridibunda) and honeybee (Apis mellifera) and in the ventral nerve cord of a beetle (Tenebrio molitor). Comp Biochem Physiol C Toxicol Pharmacol 135:315–330. https://doi.org/10.1016/S1532-0456(03)00119-4
Reilly S, Essner R Jr, Wren S, Easton L, Bishop PJ (2015) Movement patterns in leiopelmatid frogs: insights into the locomotor repertoire of basal anurans. Behav Process 121:43–53. https://doi.org/10.1016/j.beproc.2015.10.001
Reilly SM, Montuelle SJ, Schmidt A, Krause C, Naylor E, Jorgensen ME, Essner RL (2016) Pelvic function in anuran jumping: interspecific differences in the kinematics and motor control of the iliosacral articulation during take-off and landing. J Morphol 277:1539–1558. https://doi.org/10.1002/jmor.20594
Rimayi C, Odusanya D, Weiss JM, de Boer J, Chimuka L, Mbajiorgu F (2018) Effects of environmentally relevant sub-chronic atrazine concentrations on African clawed frog (Xenopus laevis) survival, growth and male gonad development. Aquat Toxicol 199:1–11. https://doi.org/10.1016/j.aquatox.2018.03.028
Rodriguez VM, Thiruchelvam M, Cory-Slechta DA (2005) Sustained exposure to the widely used herbicide atrazine: altered function and loss of neurons in brain monoamine systems. Environ Health Perspect 113:708–715. https://doi.org/10.1289/ehp.7783
Rohr JR, Crumrine PW (2005) Effects of an herbicide and an insecticide on pond community structure and processes. Ecol Appl 15:1135–1114. https://doi.org/10.1890/03-5353
Rohr JR, McCoy KA (2010) A qualitative meta-analysis reveals consistent effects of atrazine on freshwater fish and amphibians. Environ Health Perspect 118:20–32
Rohr JR, Palmer BD (2005) Aquatic herbicide exposure increases salamander desiccation risk eight months later in a terrestrial environment. Environ Toxicol Chem 24:1253–1258. https://doi.org/10.1897/04-448r.1
Rohr JR, Raffel TR, Halstead NT, McMahon TA, Johnson SA, Boughton RK, Martin LB (2013) Early-life exposure to a herbicide has enduring effects on pathogen-induced mortality. Proc Biol Sci 280:20131502. https://doi.org/10.1098/rspb.2013.1502
Scheiman J, Luber JM, Chavkin TA, MacDonald T, Tung A, Pham LD, Wibowo MC, Wurth RC, Punthambaker S, Tierney BT, Yang Z, Hattab MW, Avila-Pacheco J, Clish CB, Lessard S, Church GM, Kostic AD (2019) Meta-omics analysis of elite athletes identifies a performance-enhancing microbe that functions via lactate metabolism. Nat Med 25:1104–1109. https://doi.org/10.1038/s41591-019-0485-4
Schwarzenbach RP, Escher BI, Fenner K, Hofstetter TB, Johnson CA, von, Gunten, U., Wehrli, B. (2006) The challenge of micropollutants in aquatic systems. Science. 313:1072–1077. https://doi.org/10.1126/science.1127291
Simpson EH (1949) Measurement of diversity. Nature 163:688. https://doi.org/10.1136/thx.27.2.261
Solomon KR, Baker DB, Richards RP, Kenneth RD, Klaine SJ, Lapoint TW, Kendall RJ, Weisskopf CP, Giddings JM, Giesy JP, Hall LM Jr, Williams WM (1996) Ecological risk assessment of atrazine in North American surface waters. Environ Toxicol Chem 15:31–76. https://doi.org/10.1002/etc.5620150105
Solomon KR, Carr JA, Du Preez LH, Giesy JP, Kendall RJ, Smith EE, Van Der Kraak GJ (2008) Effects of atrazine on fish, amphibians, and aquatic reptiles: a critical review. Crit Rev Toxicol 38:721–772. https://doi.org/10.1080/10408440802116496
Starr AV, Bargu S, Maiti K, DeLaune RD (2017) The effect of atrazine on louisiana gulf coast estuarine phytoplankton. Arch Environ Contam Toxicol 72:178–188. https://doi.org/10.1007/s00244-016-0335-z
Subbarayal P, Karunakaran K, Winkler AC, Rother M, Gonzalez E, Meyer TF, Rudel T (2015) EphrinA2 receptor (EphA2) is an invasion and intracellular signaling receptor for Chlamydia trachomatis. PLoS Pathog 11:e1004846. https://doi.org/10.1371/journal.ppat.1004846
Sun HS, Sin AT, Poirier MB, Harrison RE (2016) Chlamydia trachomatis inclusion disrupts host cell cytokinesis to enhance its growth in multinuclear cells. J Cell Biochem 117:132–143. https://doi.org/10.1002/jcb.25258
Tan G, Sun W, Xu Y, Wang H, Xu N (2016) Sorption of mercury (II) and atrazine by biochar, modified biochars and biochar based activated carbon in aqueous solution. Bioresour Technol 211:727–735. https://doi.org/10.1016/j.biortech.2016.03.147
Tappe W, Groeneweg J, Jantsch B (2002) Diffuse atrazine pollution in German aquifers. Biodegradation 13:3–10. https://doi.org/10.1023/a:1016325527709
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267. https://doi.org/10.1128/AEM.00062-07
Wang Z, Ji A, Endlein T, Samuel D, Yao N, Wang Z, Dai Z (2014) The role of fore- and hindlimbs during jumping in the Dybowski’s frog (Rana dybowskii). J Exp Zool 321:324–333. https://doi.org/10.1002/jez.1865
Wang X, Bo X, Yao Q, Wu M, Wang H (2019) The effect of fluorine exposure on morphological indicators and intestinal microbial community in Bufo gargarizans tadpoles. Ecol Indic 98:763–771. https://doi.org/10.1016/j.ecolind.2018.11.070
Xu W, Li YY, Lou QQ, Chen XR, Qin ZF, Wie WJ (2015) Low concentrations of dihydrotestosterone induce female-to-male sex reversal in the frog Pelophylax nigromaculatus. Environ Toxicol Chem 34:2370–2377. https://doi.org/10.1002/etc.3072
Yang Y, Song X, Chen A, Wang H, Chai L (2020) Exposure to copper altered the intestinal microbiota in Chinese brown frog (Rana chensinensis). Environ Sci Pollut Res Int 27:13855–13865. https://doi.org/10.1007/s11356-020-07856-8
Yao Q, Yang H, Wang X, Wang H (2019) Effects of hexavalent chromium on intestinal histology and microbiota in Bufo gargarizans tadpoles. Chemosphere. 216:313–323. https://doi.org/10.1016/j.chemosphere.2018.10.147
Zelante T, Iannitti RG, Cunha C, De Luca A, Giovannini G, Pieraccini G, Zecchi R, D’Angelo C, Massi-Benedetti C, Fallarino F, Carvalho A, Puccetti P, Romani L (2013) Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22. Immunity. 39:372–385. https://doi.org/10.1016/j.immuni.2013.08.003
Zhang W, Guo R, Yang Y, Ding J, Zhang Y (2016) Long-term effect of heavy-metal pollution on diversity of gastrointestinal microbial community of Bufo raddei. Toxicol Lett 258:192–197. https://doi.org/10.1016/j.toxlet.2016.07.003
Zheng R, Chen X, Ren C, Teng Y, Shen Y, Wu M, Wang H, Huang M (2020) Comparison of the characteristics of intestinal microbiota response in Bufo gargarizans tadpoles: exposure to the different environmental chemicals (Cu, Cr, Cd and NO3-N). Chemosphere. 247:125925. https://doi.org/10.1016/j.chemosphere.2020.125925
Acknowledgements
We would like to thank the editors and three anonymous reviewers for their helpful comments and suggestions on our paper.
Funding
This work was supported by grants from the National Natural Science Foundation of China (No. 31970494), the Natural Science Foundation of Hunan Province (2019JJ40138), and the Aid program for Science and Technology Innovative Research Team in Higher Educational Institutions of Hunan Province (201937924).
Author information
Authors and Affiliations
Contributions
Qiang Zhao, Minyi Huang, and Renyan Duan conceived the research idea. Minyi Huang, Yang Liu, and Yuyue Wan conducted the field work and experiments. Yang Liu, Yuyue Wan, and Lianfu Wu conducted the statistical analyses. Qiang Zhao, Minyi Huang, and Renyan Duan wrote the first draft. All authors contributed to writing.
Corresponding authors
Ethics declarations
Ethical approval
All the procedures of animal experiments were approved by the Institutional Animal Care and Use Committee at Hunan University of Humanities, Science and Technology.
Consent to participate
We confirm that the manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. We further confirm that the order of authors listed in the manuscript has been approved by all of us.
Consent to publish
We confirm that we have given due consideration to the protection of intellectual property associated with this work and that there are no impediments to publication. We confirm that we have followed the regulations of our institutions concerning intellectual property.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Bruno Nunes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, Q., Huang, M., Liu, Y. et al. Effects of atrazine short-term exposure on jumping ability and intestinal microbiota diversity in male Pelophylax nigromaculatus adults. Environ Sci Pollut Res 28, 36122–36132 (2021). https://doi.org/10.1007/s11356-021-13234-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-13234-9