Abstract
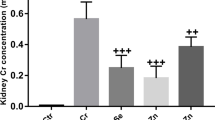
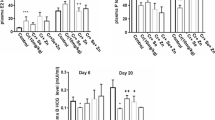
This study evaluated the toxic effects of inorganic mercury (Hg) in pregnant and lactating rats, as well as the possible protective effect of zinc (Zn) and N-acetylcysteine (NAC). Pregnant and lactating rats were pre-treated with ZnCl2 (27 mg/kg) and/or NAC (5 mg/kg) and after 24 h, they were exposed to HgCl2 (10 mg/kg). Animals were sacrificed 24 h after Hg exposure, and biochemical tests and metal determination were performed. Regarding pregnant rats, Hg exposure caused kidney, blood, and placenta δ-aminolevulinic acid dehydratase (δ-ALA-D) activity inhibition, and the pre-treatments showed a tendency of protection. Moreover, all the animals exposed to Hg presented high Hg levels in the kidney, liver, and placenta when compared with control group. Pregnant rats pre-exposed to Zn (Zn-Hg and Zn/NAC-Hg groups) presented an increase in hepatic metallothionein levels. Therefore, lactating rats exposed to Hg presented renal and blood δ-ALA-D inhibition; the pre-treatments showed a tendency to prevent the renal δ-ALA-D inhibition and prevented the blood δ-ALA-D inhibition caused by Hg. Lactating rats exposed to Hg presented high Hg levels in the kidney and liver. These results showed that 10 mg/kg of HgCl2 causes biochemistry alterations in pregnant and lactating rats, and Zn and NAC present promising results against these damages.




Similar content being viewed by others
References
Berlin M, Zalups RK, Fowler BA (2007) Mercury. In: Nordberg GF, Fowler BA, Nordberg M, Friberg L (eds) Handbook on the toxicology of metals, 3rd edn. Elsevier Holland, Amsterdam, pp 675–729. https://doi.org/10.1016/B978-012369413-3/50088-4
Bradford M (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Branco V, Caito S, Farina M, Rocha JBT, Aschner M, Carvalho C (2017) Biomarkers of mercury toxicity: past, present, and future trends. J Toxicol Environ Health B 20:119–154. https://doi.org/10.1080/10937404.2017.1289834
Cardiano P, Foti C, Giuffre O (2016) On the interaction of N-acetylcysteine with Pb2+, Zn2+, Cd2+ and Hg2+. J Mol Liq 223:360–367. https://doi.org/10.1016/j.molliq.2016.08.050
Cherian MG, Jayasurya A, Bay BH (2003) Metallothioneins in human tumors and potential roles in carcinogenesis. Mutat Res 533:201–209. https://doi.org/10.1016/j.mrfmmm.2003.07.013
Deepmala J, Deepak M, Srivastav S, Sangeeta S, Kumar SA, Kumar SS (2013) Protective effect of combined therapy with dithiothreitol, zinc and selenium protects acute mercury induced oxidative injury in rats. J Trace Elem Med Biol 27:249–256. https://doi.org/10.1016/j.jtemb.2012.12.003
Falluel-Morel A, Lin L, Sokolowski K, McCandlish E, Buckley B, DiCicco-Bloom E (2012) N-Acetyl cysteine treatment reduces mercury-induced neurotoxicity in the developing rat hippocampus. J Neurosci Res 90:743–750. https://doi.org/10.1002/jnr.22819
Favero AM, Oliveira CS, Franciscato C, Oliveira VA, Pereira JS, Bertoncheli CM, da Luz SC, Dressler VL, Flores EM, Pereira ME (2014) Lactating and non lactating rats differ to renal toxicity induced by mercuric chloride: the preventive effect of zinc chloride. Cell Biochem Funct 32:420–428. https://doi.org/10.1002/cbf.3032
Franciscato C, Goulart FR, Lovatto NM, Duarte FA, Flores EM, Dressler VL, Peixoto NC, Pereira ME (2009) ZnCl2 exposure protects against behavioral and acetylcholinesterase changes induced by HgCl2. Int J Dev Neurosci 27:459–468. https://doi.org/10.1016/j.ijdevneu.2009.05.002
Franciscato C, Moraes-Silva L, Duarte FA, Oliveira CS, Ineu RP, Flores EM, Dressler VL, Peixoto NC, Pereira ME (2011) Delayed biochemical changes induced by mercury intoxication are prevented by zinc pre-exposure. Ecotoxicol Environ Saf 74:480–486. https://doi.org/10.1016/j.ecoenv.2010.11.011
Girardi G, Elias MM (1991) Effectiveness of N-acetylcysteine in protecting against mercuric chloride-induced nephrotoxicity. Toxicology 67:155–164. https://doi.org/10.1016/0300-483X(91)90139-R
Hanwell A, Linzell JL (1973) The time course of cardiovascular changes in lactation in the rat. J Physiol 233:93–109. https://doi.org/10.1113/jphysiol.1973.sp010299
Herrera E (2002) Implications of dietary fatty acids during pregnancy on placental, fetal and postnatal development-a review. Placenta 16:9–19. https://doi.org/10.1053/plac.2002.0771
Ineu RP, Oliveira CS, Oliveira VA, Moraes-Silva L, Almeida-Luz SC, Pereira ME (2013) Antioxidant effect of zinc chloride against ethanol-induced gastrointestinal lesions in rats. Food Chem Toxicol 58:522–529. https://doi.org/10.1016/j.fct.2013.05.022
Jalilehvand F, Parmar K, Zielke S (2013) Mercury (II) complex formation with N-acetylcysteine. Metallomics 5:1368–1376 https://doi.org/10.1039/C3MT00173C
Joshi D, Mittal DK, Shukla S, Srivastav AK, Srivastav SK (2014) N-acetyl cysteine and selenium protects mercuric chloride-induced oxidative stress and antioxidant defense system in liver and kidney of rats: a histopathological approach. J Trace Elem Med Biol 28:218–226. https://doi.org/10.1016/j.jtemb.2013.12.006
Joshi D, Mittal D, Shrivastav S, Shukla S, Srivastav AK (2011) Combined effect of N-acetyl cysteine, zinc, and selenium against chronic dimethylmercury-induced oxidative stress: a biochemical and histopathological approach. Arch Environ Contam Toxicol 61:558–567. https://doi.org/10.1007/s00244-011-9656-0
Joshi D, Mittal DK, Shukla S, Srivastav AK (2012) Therapeutic potential of N-acetyl cysteine with antioxidants (Zn and Se) supplementation against dimethylmercury toxicity in male albino rats. Exp Toxicol Pathol 64:103–108. https://doi.org/10.1016/j.etp.2010.07.001
Mesquita M, Pedroso TF, Oliveira CS, Oliveira VA, Santos RF, Bizzi CA, Pereira ME (2016) Effects of zinc against mercury toxicity in female rats 12 and 48 hours after HgCl2 exposure. EXCLI J 15:256–267. https://doi.org/10.17179/excli2015-709
Moraes-Silva L, Oliveira CS, Peixoto NC, Pereira ME (2018) Copper attenuates early and late biochemical alterations induced by inorganic mercury in young rats. J Toxicol Environ Health A 81:633–644. https://doi.org/10.1080/15287394.2018.1474153
Oliveira CS, Pereira ME, Oliveira VA, Favero AM, Ineu RP (2018b) Zinc supplementation ameliorates biochemical changes and Hg intestinal deposition caused by inorganic mercury intoxication. World J Pharmacol Toxicol. 1:101
Oliveira CS, Favero AM, Franciscato C, da Luz CS, Pereira ME (2014a) Distinct response of lactating and nonlactating rats exposed to inorganic mercury on hepatic δ-aminolevulinic acid dehydratase activity. Biol Trace Elem Res 158:230–237. https://doi.org/10.1007/s12011-014-9931-9
Oliveira CS, Piccoli BC, Aschner M, Rocha JBT (2017b) Chemical speciation of selenium and mercury as determinant of their neurotoxicity. In: Aschner M, Costa L (eds): Neurotoxicity of Metals. Advances in Neurobiology. Springer. Cham. 18:53–83. https://doi.org/10.1007/978-3-319-60189-2_4
Oliveira CS, Joshee L, George H, Nijhara S, Bridges CC (2017a) Oral exposure of pregnant rats to toxic doses of methylmercury alters fetal accumulation. Reprod Toxicol 69:265–275. https://doi.org/10.1016/j.reprotox.2017.03.008
Oliveira CS, Nogara PA, Ardisson-Araújo DMP, Aschner M, Rocha JBT, Dórea JG (2018a) Neurodevelopmental effects of mercury. In: Aschner M, Costa L (eds): Linking environmental exposure to neurodevelopmental disorders. Advances in Neurotoxicology. Elsevier. 2:27–86. https://doi.org/10.1016/bs.ant.2018.03.005
Oliveira CS, Oliveira VA, Costa LM, Pedroso TF, Fonseca MM, Bernardi JS, Fiuza TL, Pereira ME (2016a) Inorganic mercury exposure in drinking water alters essential metal homeostasis in pregnant rats without altering rat pup behavior. Reprod Toxicol 65:18–23. https://doi.org/10.1016/j.reprotox.2016.06.013
Oliveira CS, Oliveira VA, Ineu RP, Moraes-Silva L, Pereira ME (2012) Biochemical parameters of pregnant rats and their offspring exposed to different doses of inorganic mercury in drinking water. Food Chem Toxicol 50:2382–2387. https://doi.org/10.1016/j.fct.2012.04.046
Oliveira VA, Oliveira CS, Mesquita M, Pedroso TF, Costa LM, Fiuza TL, Pereira ME (2015) Zinc and N-acetylcysteine modify mercury distribution and promote increase in hepatic metallothionein levels. J Trace Elem Med Biol 32:183–188. https://doi.org/10.1016/j.jtemb.2015.06.006
Oliveira VA, Oliveira CS, Ineu RP, Moraes-Silva L, de Siqueira LF, Pereira ME (2014b) Lactating and non-lactating rats differ in sensitivity to HgCl2: Protective effect of ZnCl2. J Trace Elem Med Biol 28:240–246. https://doi.org/10.1016/j.jtemb.2014.01.005
Oliveira VA, Favero G, Stacchiotti A, Giugno L, Buffoli B, Oliveira CS, Lavazza A, Albanese M, Rodella LF, Pereira ME, Rezzani R (2016b) Acute mercury exposition of virgin, pregnant, and lactating rats: Histopathological kidney and liver evaluations. Environ Toxicol 32:1500–1512. https://doi.org/10.1002/tox.22370
Panemangalore M, Banerjee D, Onosaka S, Cherian MG (1983) Changes in the intracellular accumulation and distribution of metallothionein in rat liver and kidney postnatal development. Dev Biol 97:95–102. https://doi.org/10.1016/0012-1606(83)90067-2
Peixoto NC, Pereira ME (2007) Effectiveness of ZnCl2 in protecting against nephrotoxicity induced by HgCl2 in newborn rats. Ecotoxicol Environ Saf 66:441–446. https://doi.org/10.1016/j.ecoenv.2006.02.012
Peixoto NC, Serafim MA, Flores EM, Bebianno MJ, Pereira ME (2007) Metallothionein, zinc, and mercury levels in tissues of young rats exposed to zinc and subsequently to mercury. Life Sci 81:1264–1271. https://doi.org/10.1016/j.lfs.2007.08.038
Peixoto NC, Roza T, Flores EM, Pereira ME (2003) Effects of zinc and cadmium on HgCl2- δ-ALA-D inhibition and Hg levels in tissues of suckling rats. Toxicol Lett 146:17–25. https://doi.org/10.1016/j.toxlet.2003.08.006
Picciano MF (2003) Pregnancy and lactation: Physiological adjustmensts, nutritional requirements and the role of dietary supplements. J Nutr 133:1997–2002. https://doi.org/10.1093/jn/133.6.1997S
Prohaska C, Pomazal K, Steffan I (2000) Determination of Ca, Mg, Fe, Cu, and Zn in blood fractions and whole blood of humans by ICP-OES. Fresenius J Anal Chem 367:479–484. https://doi.org/10.1007/s002160000383
Sandstead HH, Au W (2007) Zinc. In: Nordberg GF, Fowler BA, Nordberg M, Friberg LT (eds): Handbook on the Toxicology of Metals, Academic Press. USA. 47:925–947. https://doi.org/10.1016/B978-012369413-3/50102-6
Sassa S (1982) Delta-aminolevulinic acid dehydratase assay. Enzyme 28:133–145. https://doi.org/10.1159/000459097
Shah D (2011) Magnitude of zinc deficiency and efficacy of zinc. Indian J Pediatr 78:1140–1141. https://doi.org/10.1007/s12098-011-0556-0
Sheats RD, Pankratz VS (2002) Understanding distributions and data types. Semin Orthodon 8:62–66. https://doi.org/10.1053/sodo.2002.32075
Silva-Pereira LC, Rocha CAM, LRCSC Jr, Costa ET, Guimarães APA, Pontes TB, Diniz DL, Leal MF, Moreira-Nunes CA, Burbano RR (2014) Protective effect of prolactin against methylmercury-induced mutagenicity and cytotoxicity on human lymphocytes. Int J Environ Res Public Health 11:9822–9834. https://doi.org/10.3390/ijerph110909822
Solaiman D, Jonah M, Miyazaki W, Ho G, Bhattacharyya MH (2001) Increased metallothionein in mouse liver, kidneys, and duodenum during lactation. Toxicol Sci 60:184–192. https://doi.org/10.1093/toxsci/60.1.184
Stehbens WE (2003) Oxidative stress, toxic hepatitis, and antioxidants with particular emphasis on zinc. Exp Mol Pathol 75:265–276. https://doi.org/10.1016/s0014-4800(03)00097-2
Suzuki K, Hirose H, Hokao R, Takemura N, Motoyoshi S (1993) Changes of plasma osmotic pressure during lactation in rats. J Vet Med Sci 55:561–564. https://doi.org/10.1292/jvms.55.561
Suzuki K, Koizumi N, Hirose H, Hokao R, Takemura N, Motoyoshi S (2000) Changes in plasma arginine vasopressin concentration during lactation in rats. Comp Med 50:277–280
Thomas L (1998) Alanine aminotransferase (ALT), aspartate aminotranferase (AST). In: Thomas L (ed) Clinical Laboratory Diagnostics, First edn. TH-Books Verlangsgesellschaft, Frankfurt, pp 55–65
Trümpler S, Nowak S, Meermann B, Wiesmüller GA, Buscher W, Sperling M, Karst U (2009) Detoxification of mercury species-an in vitro study with antidotes in human whole blood. Anal Bioanal Chem 395:1929–1935. https://doi.org/10.1007/s00216-009-3105-1
Williamson DH (1986) Regulation of metabolism during lactation in the rat. Reprod Nutr Dev 26:597–603. https://doi.org/10.1051/rnd:19860409
Acknowledgments
The authors would like to thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq 311082/2014-9), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/PROEX (23038.005848/2018-31; 0737/2018).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Oliveira, V.A., de Souza da Costa, N., Mesquita, M. et al. Mercury toxicity in pregnant and lactating rats: zinc and N-acetylcysteine as alternative of prevention. Environ Sci Pollut Res 27, 40563–40572 (2020). https://doi.org/10.1007/s11356-020-09836-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09836-4