Abstract
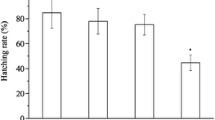
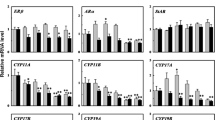
Bisphenol F (BPF) has been frequently detected in various environmental compartments, and previous studies found that BPF exhibits similar estrogenic and anti-androgenic effects on the mammalian endocrine system to those of bisphenol A (BPA). However, the potential disrupting effects of BPF on aquatic organisms and the underling disrupting mechanisms have not been investigated. In this study, the potential disrupting mechanisms of BPF on the hypothalamic–pituitary–gonadal (HPG) axis and liver were probed by employing the OECD 21-day short-term fecundity assay in zebrafish. The results show that BPF exposure (1 mg/L) impaired the reproductive function of zebrafish, as exemplified by alterations to testicular and ovarian histology of the treated zebrafish. Homogenate testosterone (T) levels in male zebrafish decreased in a concentration-dependent manner, and 17β-estradiol (E2) levels increased significantly when fish were exposed to 0.1 and 1 mg/L BPF. The real-time polymerase chain reaction was performed to examine gene expression in the HPG axis and liver. Hepatic vitellogenin expression was significantly upregulated in males, suggesting that BPF possesses estrogenic activity. The disturbed hormone balance was enhanced by the significant changes in gene expression along the HPG axis. These alterations suggest that BPF leads to adverse effects on the endocrine system of teleost fish, and that these effects were more prominent in males than in females.







Similar content being viewed by others
References
Andersson E, Nijenhuis W, Male R, Swanson P, Bogerd J, Taranger GL, Schulz RW (2009) Pharmacological characterization, localization and quantification of expression of gonadotropin receptors in Atlantic salmon (Salmo salar L.) ovaries. Gen Comp Endocrinol 163(3):329–339
Ankley GT, Villeneuve DL (2006) The fathead minnow in aquatic toxicology: past, present and future. Aquat Toxicol 78(1):91–102
Joel CN, Colette V, Elisabeth P, Charlier TD, Olivier K, Pascal C (2016) Estrogenic effects of several BPA analogs in the developing zebrafish brain. Front Neurosci 10:112
Chen D, Kannan K, Tan H, Zheng Z, Feng YL, Wu Y, Widelka M (2016) Bisphenol analogues other than BPA: environmental occurrence, human exposure, and toxicity—a review. Environ Sci Technol 50(11):5438–5453
Clelland E, Peng C (2009) Endocrine/paracrine control of zebrafish ovarian development. Mol Cell Endocrinol 312(1):42–52
Christianson-Heiska IL, Haavisto T, Paranko J, Bergelin E, Isomaa B (2008) Effects of the wood extractives dehydroabietic acid and betulinol on reproductive physiology of zebrafish (Danio rerio)—a two-generation study. Aquat Toxicol 86(3):388–396
Daouk T, Larcher T, Roupsard F, Lyphout L, Rigaud C, Ledevin M, Loizeau V, Cousin X (2011) Long-term food-exposure of zebrafish to PCB mixtures mimicking some environmental situations induces ovary pathology and impairs reproduction ability. Aquat Toxicol 105(3):270–278
European Commission, 2011. Bisphenol a: EU ban on baby bottles to enter into force tomorrow
Filby AL, van Aerle R, Duitman J, Tyler CR (2008) The kisspeptin/gonadotropin-releasing hormone pathway and molecular signaling of puberty in fish. Biol Reprod 78(2):278–289
FDA (2008) Draft assessment of Bisphenol A for use in food contact applications. Food and Drug Administration, U.S.
Goldinger DM, Demierre AL, Zoller O, Rupp H, Reinhard H, Magnin R, Becker TW, Bourqui-Pittet M (2015) Endocrine activity of alternatives to BPA found in thermal paper in Switzerland. Reg Toxicol Pharmacol 71(3):453–462
Han XB, Yuen KW, Wu RS (2013) Polybrominated diphenyl ethers affect thereproduction and development, and alter the sex ratio of zebrafish (Daniorerio). Environ Pollut 182:120–126
Ji K, Hong S, Kho Y, Choi K (2013) Effects of bisphenol S exposure on endocrine functions and reproduction of zebrafish. Environ Sci Technol 47(15):8793–8800
Keiter S, Baumann L, Färber H, Holbech H, Skutlarek D, Engwall M et al (2012) Long-term effects of a binary mixture of perfluorooctane sulfonate (PFOS) and bisphenol A (BPA) in zebrafish (Danio rerio). Aquat Toxicol 118–119:116–129
Kinnberg, K., Korsgaard, B., Bjerregaard, P. 2003. Effects of octylphenol and 17 beta-estradiol on the gonads of guppies (poecilia reticulata) exposed as adults via the water or as embryos via the mother. Comp Biochem Physiol Part C Toxicol Pharmacol 134(1), 45–55
Kwok HF, So WK, Wang Y, Ge W (2005) Zebrafish gonadotropins and their receptors: I. Cloning and characterization of zebrafish follicle-stimulating hormone and luteinizing hormone receptors—evidence for their distinct functions in follicle development. Biol Reprod 72(6):1370–1381
Lee S, Liao C, Song GJ, Ra K, Kannan K, Moon HB (2015) Emission of bisphenol analogues including bisphenol A and bisphenol F from wastewater treatment plants in Korea. Chemosphere 119:1000–1006
Le Fol V, Aïtaïssa S, Sonavane M, Porcher JM, Balaguer P, Cravedi JP, Zalko D, Brion F (2017) In vitro and in vivo estrogenic activity of BPA, BPF and BPS in zebrafish-specific assays. Ecotoxicol Environ Saf 142:150–156
Liao C, Liu F, Guo Y, Moon HB, Nakata H, Wu Q, Kannan K (2012a) Occurrence of eight bisphenol analogues in indoor dust from the United States and several Asian countries: implications for human exposure. Environ Sci Technol 46(16):9138–9145
Liao C, Liu F, Moon HB, Yamashita N, Yun S, Kannan K (2012b) Bisphenol analogues in sediments from industrialized areas in the United States, Japan, and Korea: spatial and temporal distributions. Environ Scie Technol 46(21):11558–11565
Liao C, Kannan K (2013) Concentrations and profiles of bisphenol A and other bisphenol analogues in foodstuffs from the United States and their implications for human exposure. J Agric Food Chem 61(19):4655–4662
Liu C, Yu L, Deng J, Lam PK, Wu RS, Zhou B (2009) Waterborne exposure to fluorotelomer alcohol 6: 2 FTOH alters plasma sex hormone and gene transcription in the hypothalamic–pituitary–gonadal (HPG) axis of zebrafish. Aquat Toxicol 93(2):131–137
Liu X, Ji K, Jo A, Moon HB, Choi K (2013) Effects of TDCPP or TPP on gene transcriptions and hormones of HPG axis, and their consequences on reproduction in adult zebrafish (Danio rerio). Aquat Toxicol 134:104–111
Mandich A, Bottero S, Benfenati E, Cevasco A, Erratico C, Maggioni S, Massari A, Pedemonte F, Vigano L (2007) In vivo exposure of carp to graded concentrations of bisphenol A. Gen Comp Endocrinol 153(1):15–24
Molinamolina JM, Amaya E, Grimaldi M, Sáenz JM, Real M, Fernández MF, Balaguer P, Olea N (2013) In vitro study on the agonistic and antagonistic activities of bisphenol-S and other bisphenol-A congeners and derivatives via nuclear receptors. Toxicol Appl Pharmacol 272(1):127–136
Naderi M, Wong MY, Gholami F (2014) Developmental exposure of zebrafish (Danio rerio) to bisphenol-S impairs subsequent reproduction potential and hormonal balance in adults. Aquat Toxicol 148(2):195–203
Nash JP, Kime DE, Van der Ven LT, Wester PW, Brion F, Maack G, Allner PS, Tyler CR (2004) Long-term exposure to environmental concentrations of the pharmaceutical ethynylestradiol causes reproductive failure in fish. Environ Health Perspect:1725–1733
Organization for Economic Co-operation and Development (2009a) Fish short term reproduction assay. OECD guideline 229. Paris, France
Organization for Economic Co-operation and Development (2009b) 21-day Fish Assay. OECD guideline 230. Paris, France
Pivnenko K, Pedersen GA, Eriksson E, Astrup TF (2015) Bisphenol a and its structural analogues in household waste paper. Waste Manag 44:39–47
Rochester JR (2013) Bisphenol A and human health: a review of the literature. Reprod Toxicol 42:132–155
Rochester JR, Bolden AL (2015) Bisphenol S and F: a systematic review and comparison of the hormonal activity of bisphenol A substitutes. Environ Health Perspect 123(7):643–650
Segner H (2009) Zebrafish (Danio rerio) as a model organism for investigating endocrine disruption. Comp Biochem Physiol C 149(2):187–195
Shang EHH, Yu RMK, Wu RSS (2006) Hypoxia affects sex differentiation and development, leading to a male-dominated population in zebrafish (Danio rerio). Environ Sci Technol 40:3118–3122
Shi J, Jiao Z, Zheng S, Li M, Zhang J, Feng Y, Yin J, Shao B (2015) Long-term effects of bisphenol AF (BPAF) on hormonal balance and genes of hypothalamus-pituitary-gonad axis and liver of zebrafish (Danio rerio), and the impact on offspring. Chemosphere 128:252–257
Sohoni PCRT, Tyler CR, Hurd K, Caunter J, Hetheridge M, Williams T, Woods C, Sumpter JP (2001) Reproductive effects of long-term exposure to bisphenol A in the fathead minnow (Pimephales promelas). Environ Sci Technol 35(14):2917–2925
Song M, Liang D, Liang Y, Chen M, Wang F, Wang H, Jiang G (2014a) Assessing developmental toxicity and estrogenic activity of halogenated bisphenol A on zebrafish (Danio rerio). Chemosphere 112:275–281
Song S, Song M, Zeng L, Wang T, Liu R, Ruan T, Jiang G (2014b) Occurrence and profiles of bisphenol analogues in municipal sewage sludge in china. Environ Pollut 186(1):14–19
Stroheker T, Chagnon MC, Pinnert MF, Berges R, Canivenc-Lavier MC (2003) Estrogenic effects of food wrap packaging xenoestrogens and flavonoids in female Wistar rats: a comparative study. Reprod Toxicol 17:421–432
Uren Webster TM, Laing LV, Florance H, Santos EM (2014) Effects of glyphosate and its formulation, roundup, on reproduction in zebrafish (Danio rerio). Environ Sci Technol 48(2):1271–1279
Van den Belt K, Verheyen R, Witters H (2001) Reproductive effects of ethynylestradiol and 4t-octylphenol on the zebrafish (Danio rerio). Arch Environ Contam Toxicol 41(4):458–467
Viganò L, Benfenati E, Bottero S, Cevasco A, Monteverde M, Mandich A (2010) Endocrine modulation, inhibition of ovarian development and hepatic alterations in rainbow trout exposed to polluted river water. Environ Pollut 158(12):3675–3683
Yamaguchi A, Ishibashi H, Arizono K, Tominaga N (2015) In vivo and in silico analyses of estrogenic potential of bisphenol analogs in medaka (oryzias latipes) and common carp (cyprinus carpio). Ecotoxicol Environ Saf 120:198–205
Yamasaki K, Noda S, Imatanaka N, Yakabe Y (2004) Comparative study of the uterotrophic potency of 14 chemicals in a uterotrophic assay and their receptor-binding affinity. Toxicol Lett 146:111–120
Yamasaki K, Takeyoshi Y, Yakabe Y, Sawaki M, Imatanaka N, Takatsuki M (2002) Comparison of gene reporter assay and immature rat uterotrophic assay of twenty-three chemicals. Toxicology 170:21–30
Yamazaki E, Yamashita N, Taniyasu S, Lam J, Lam PK, Moon HB, Jeong Y, Kannan P, Achyuthan H, Munuswamy N, Kannan K (2015) Bisphenol A and other bisphenol analogues including BPS and BPF in surface water samples from Japan, China, Korea and India. Ecotoxicol Environ Saf 122:565–572
Yang Q, Yang X, Liu J, Ren W, Chen Y, Shen S (2017) Exposure to bisphenol B disrupts steroid hormone homeostasis and gene expression in the hypothalamic–pituitary–gonadal axis of zebrafish. Water Air Soil Pollut 228(3):112
Yang X, Liu Y, Li J, Chen M, Peng D, Liang Y, Zhang J, Jiang G (2014b) Exposure to Bisphenol AF disrupts sex hormone levels and vitellogenin expression in zebrafish. Environ Toxicol 31(3):285–294
Yang Y, Guan J, Yin J, Shao B, Li H (2014a) Urinary levels of bisphenol analogues in residents living near a manufacturing plant in south China. Chemosphere 112:481–486
Yön ND, Akbulut C (2014) Histological changes in zebrafish (Danio rerio) ovaries following administration of bisphenol A. Pakistan J Zool 46(4):1153–1159
Zhou X, Kramer JP, Calafat AM, Ye X (2014) Automated on-line column-switching high performance liquid chromatography isotope dilution tandem mass spectrometry method for the quantification of bisphenol a, bisphenol F, bisphenol S, and 11 other phenols in urine. J Chromatogr B Analytic Technologi Biomedical Life Sci 944:152–156
Zoller O, Brüschweiler BJ, Magnin R, Reinhard H, Rhyn P, Rupp H, Zeltnenr S, Felleisen R (2015) Natural occurrence of bisphenol F in mustard. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 33(1):137–146
Acknowledgments
This work was supported by the Natural Science Foundation of Jiangsu Province (General Program) (No. BK20151100), National Natural Science Foundation of China (No. 21507038), and Graduate Research and Innovation Projects of Jiangsu Province (No. KYLX15_0813).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Cinta Porte
Electronic supplementary material
ESM 1
(DOCX 24 kb)
Rights and permissions
About this article
Cite this article
Yang, Q., Yang, X., Liu, J. et al. Effects of BPF on steroid hormone homeostasis and gene expression in the hypothalamic–pituitary–gonadal axis of zebrafish. Environ Sci Pollut Res 24, 21311–21322 (2017). https://doi.org/10.1007/s11356-017-9773-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9773-z