Abstract
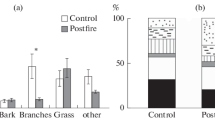
Mass-loss rates during the early phase of decomposition of plant residues were studied for a period of 3 years in Norway spruce forests subjected to air pollution by Cu-Ni smelters on the Kola Peninsula, northwest Russia. Litterbags were deployed in two main patches of forests at the northern tree line, between and below the crowns of spruce trees older than 100 years. The study results demonstrated the dependence of the decomposition rates on the initial concentrations of nutrients and the C/N and lignin/N ratios in plant residues. Lower rates of mass loss in forests subject to air pollution may be related to low quality of plant residues, i.e. high concentrations of heavy metals, low concentrations of nutrients, and high lignin/N and C/N ratios. The increased losses of Ca, Mg, K, and Mn from plant residues in these forests compared to the reference were, probably, related to leaching of their compounds from the residues. The relatively high rates of heavy metal accumulation in the residues were most likely related to uptake of pollutants from the atmosphere, as well as to the lower mass-loss rates. The present study results demonstrate that the forest patchiness should be taken into account in assessment and predictions of decomposition rates in Norway spruce forests. Mass-loss rates of plant residues below the crowns of old spruce trees were significantly lower than those in the patches between the crowns. This was explained by the high C/N and lignin/N ratios in the residues of evergreens which contribute significantly to litterfall below the crowns and by lower soil temperature during winter and spring below the crowns. In addition, a lower amount of precipitation reaching the forest floor below the dense, long crowns of old Norway spruce trees may result in considerably lower washing out of the organic compounds from the residues. Lower mass-loss rates below the crowns of old spruce trees may be part of the evidence that the old-growth spruce forests can continue to accumulate carbon in soil.





Similar content being viewed by others
References
Aamlid D, Tømmervik H, Gytarsky M, Karaban R, Venn K, Michael L, Rindal T, Lukina N, Koptsik G, Løbersli E (1995) Determination of exceedance of critical levels in the border area between Norway and Russia. In: Løbersli E, Venn K, eds Effect of air pollution on terrestrial ecosystems in the border area between Norway and Russia, Vol 10 Proceedings from the second symposium, Svanvik, Norway 3-5101994 DN-Utredning 1995/8, 19–23
ACIA (2005) Arctic climate impact assessment. Cambridge University Press, Cambridge
Aerts R, van Bodegom PM, Cornelissen JHC (2012) Litter stoichiometric traits of plant species of high-latitude ecosystems show high responsiveness to global change without causing strong variation in litter decomposition. New Phytol 196:181–188
Augusto L, Ranger J, Binkley D, Rothe A (2002) Impact of several common tree species of European temperate forests on soil fertility. Ann For Sci 59:233–253
Baath E (1989) Effects of heavy metals in soil on microbial processes and populations (a review). Water Air Soil Pollut 47:335–379
Berg B (2000) Litter decomposition and organic matter turnover in northern forest soils. Forest Ecol Manag 133:13–22
Berg B, Ekbohm G (1991) Litter mass-loss rates and decomposition patterns in some needle and leaf litter types. Long-term decomposition in a Scots pine forest. Can J Bot 69:1449–1456
Binkley D (1994) The influence of tree species on forest soils: processes and patterns. In: Proceedings of the tree and soil workshop, Lincoln University, 28 February–2 March 1994 Eds Mead DJ and Cornforth IS Spec Publ 10. Lincoln University Press, Canterbury, New Zealand, pp 1–33
Binkley D, Giardina C (1998) Why do trees affect soils? The warp and woof of tree-soil interactions. Biogeochemistry 42:89–106
Binkley D, Menyailo O (2005) Gaining insights on the effects of tree species on soils. In: Tree species effects on soils: implications for global change, NATO Science Series IV: Earth and Environmental Sciences, vol 55, pp 1–16
Coq S, Souquet J-M, Meudec E, Cheynier V, Hättenschwiler S (2010) Interspecific variation in leaf litter tannins drives decomposition in a tropical rain forest of French Guiana. Ecology 91:2080–2091
Cornelissen JHC, van Bodegom PM, Aerts R, Callaghan TV, van Logtestijn RSP, Alatalo J, Chapin FS, Gerdo R, Gudmundsson J, Gwynn-Jones D, Hartley AE, Hik DS, Hofgaard A, Jonsdottir IS, Karlsson S, Klein JA, Laundre J, Magnusson B, Michelsen A, Molau U, Onipchenko VG, Quested HV, Sandvik SM, Schmidt IK, Shaver GR, Bjorn Solheim B, Nadejda A, Soudzilovskaia NA, Stenström A, Tolvanen F, Totland O, Wada N, Welker JM, Zhao X, Team MOL (2007) Global negative vegetation feedback to climate warming responses of leaf decomposition rates in cold biomes. Ecol Lett 10:619–627. doi:10.1111/j1461-0248200701051x
Cornwell WK, Cornelissen JHC, Amatangelo K, Dorrepaal E, Eviner VT, Godoy O, Hobbie S, Hoorens B, Kurokawa H, Pe’rez-Harguindeguy N, Quested HM, Santiago LS, Wardle D, Wright I, Aerts R, Allison S, van Bodegom P, Brovkin V, Chatain A, Callaghan T, Diaz S, Garnier E, Gurvich D, Kazakou E, Klein J, Read J, Reich P, Soudzilovskaia N, Vaieretti V, Westoby M (2008) Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol Lett 11:1065–1071
Ershov V, Lukina N, Orlova M, Zukert N (2016) Dynamics of the snow water composition in coniferous forests exposed to airborne industrial pollution. Russian Journal of Ecology 1 47(1):46–52
Freedman B, Hutchinson TC (1980) Effects of smelter pollutants on forest leaf litter decomposition near a nickel-copper smelter at Sudbury, Ontario. Can J Bot 58:1722–1736
Freilich L, Calcote R, Davis M, Pastor J (1993) Patch formation and maintenance in an old-growth hemlock–hardwood forest. Ecology 74:513–527
Fritze H, Pennanen T, Haimi J, Siira-Pietikäinen A, Vanhala P (2000) Effects of heavy metals on soil microflora. In: Malkonen E (ed) Forest condition in a changing environment—the Finnish case, pp 260–265
Gorbacheva T, Kikuchi R (2006) Plant-to-soil pathways in the subarctic—qualitative and quantitative changes of different vegetative fluxes. Environ Biotechnol 2:26–30
Harmens H, Schnyder E, Thöni L, Cooper DM, Mills G, Leblond S, Mohr K, Poikolainen J, Santamaria J, Skudnik M, Zechmeister HG, Lindroos A-J, Hanus-Illnar A (2014) Relationship between site-specific nitrogen concentrations in mosses and measured wet bulk atmospheric nitrogen deposition across. Europe Environ Pollut 14:50–59
Hilli S (2013) Significance of litter production of forest stands and ground vegetation in the formation of organic matter and storage of carbon in boreal coniferous forests. In: Merilä P, Jortikka S (eds) Forest condition monitoring in Finland—national report the Finnish Forest Research Institute Available: http://urnfi/URN:NBN:fi: metla-201305087573
Hilli S, Stark S, Derome J (2010) Litter decomposition rates in relation to litter stocks in boreal coniferous forests along climatic and soil fertility gradients. Appl Soil Ecol 46:200–208
Hobbie SE (1996) Temperature and plant species control over litter decomposition in Alaskan tundra. Ecol Monogr 66:503–522
Jalal M, Read D, Haslam E (1982) Phenolic composition and its seasonal variation in Calluna vulgaris. Phytochemistry 21:1397–1401
Johansson K, Bergback B, Tyler G (2001) Impact of atmospheric long range transport of lead, mercury and cadmium on the Swedish forest environment. Water, Air, Soil Pollut Focus 1:279–297
Keenan R, Prescott C, Kimmens J (1993) Mass and nutrient content of woody debris and forest floor in western red cedar and western hemlock forests on northern forests on northern Vancouver Island. Can J For Res 23:1052–1059
Kitayama K, Suzuki S, Hori M, Takyu M, Aiba SH et al (2004) On the relationships between leaf-litter lignin and net primary productivity in tropical rain forests. Oecologia 140:335–339
Kozlov MV, Barcan V (2000) Environmental contamination in the central part of the Kola Peninsula: history, documentation, and perception. Ambio 29:512–517
Kozlov M, Zvereva E (2015) Decomposition of birch leaves in heavily polluted industrial barrens: relative importance of leaf quality and site of exposure. Environ Sci Pollut Res. doi:10.1007/s11356-015-4165-8 (First Online)
Kurokawa H, Peltzer DA, Wardle DA (2010) Plant traits, leaf palatability and litter decomposability for co-occurring woody species on invasion status and nitrogen fixation ability. Functional Ecology Т 24(3):С. 513–С. 523
Li X, Han S, Zhang Y (2007) Indirect effects of precipitation variation on the decomposition process of Mongolian oak (Quercus mongolica) leaf litter Front Forest China 2:417–423
Ligrone R, Carafa A, Duckett JG, Renzaglia KS, Ruel K (2008) Immunocytochemical detection of lignin-related epitopes in cell walls in bryophytes and the charalean alga. Nitella Plant Syst Evol 270:257–272
Lukina NV, Nikonov VV (1991) Pollution-induced changes in the primary production of spruce ecosystems in the Kola Peninsula. In: Tikkanen E, Varmola M (eds) Research into forest damage connected with air pollution in Finnish Lapland and the Kola Peninsula of the USSR. A seminar held in Kuusamo, Finland, 25–26 May 1990, 373: 120–131
Lukina NV, Nikonov VV (1995) Acidity of podzolic soils subjected to sulphur pollution near a Cu-Ni smelter at the Kola Peninsula. Water, Air, Soil Pollut 85(1995):1057–1062
Lukina NV, Nikonov VV (1998) Content and composition of humus in the northern taiga Al–Fe-humus podzolic soils subject to atmospheric pollution. Eurasian Soil Science 31(6):671–678
Lukina N, Nikonov V (2001) Assessment of environmental impact zones in the Kola Peninsula forest ecosystems. Chemosphere 42(1):19–34
Lukina NV, Nikonov VV, Conroy H (eds) (2003) Degradational succession of forest ecosystems in the surroundings of Cu-Ni smelter in the Kola Peninsula. Laurentian University, Canada http://pdf.library.laurentian.ca/medb/conf/Sudbury03/Forests/93.pdf
Mahabubur Rahman M, Motiur RM (2012) Quantitative chemical defense traits, litter decomposition and Forest ecosystem functioning. In: Blanco JA (ed) Forest ecosystems—more than just trees Available: http://www intechopencom/books/forestecosystems-more-than-just-trees/quantitative-chemical-defense-traits-litter-decomposition-and-forestecosystem-functioning
Makkonen M, Berg MP, Handa IT, Hättenschwiler S, van Ruijven J et al (2012) Highly consistent effects of plant litter identity and functional traits on decomposition across a latitudinal gradient. Ecol Lett 15:1033–1041. doi:10.1111/j.1461-0248.2012.01826.x
Meentemeyer V (1978) Macroclimate and lignin control of litter decomposition rates. Ecology 59:465–472
Moore TR, Trofymow JA, Taylor B, Prescott C, Camiré C et al (1999) Litter decomposition rates in Canadian forests. Glob Change Biol 5:75–82
Newsham KK, Boddy L, Frankland JC, Ineson P (1992a) Effects of dry deposited sulphur dioxide on fungal decomposition of angiosperm tree leaf litter III. Decomposition rates and fungal respiration. New Phytol 122:127–140
Newsham KK, Ineson P, Boddy L, Frankland JC (1992b) Effects of dry-deposited sulphur dioxide on fungal decomposition of angiosperm tree leaf litter II. Chemical content of leaf litters. New Phytol 122:111–125
Nikonov VV, Lukina NV (2000) Influence of spruce and pine on the acidity and composition of atmospheric fallout in north taiga forests of an industrially developed region. Russ J Ecol 31:82–90
Nilsson MC, Wardle DA (2005) Understory vegetation as a forest ecosystem driver: evidence from the northern Swedish boreal forest. Front Ecol Environ 3:421–428
Orlova M, Lukina N, Tutubalina O, Smirnov V, Isaeva L, Hofgaard A (2013) Soil nutrient’s spatial variability in forest–tundra ecotones on the Kola Peninsula, Russia. Biogeochemistry 113(1–3):283–305
Orlova MA, Lukina NV, Smirnov VE (2015) Methodical approaches to forest litter sampling with taking into account biogeocoenosis mosaics. Lesovedenie 3:242–249
Orlova MA, Lukina NV, Smirnov VE, Artemkina NA (2016) The influence of spruce on acidity and nutrient content in soils of Northern Taiga dwarf shrub-green moss spruce forests. Eurasian Soil Science 49(11):1276–1287
Polyanskaya LM, Nikonov VV, Lukina NV, Panikova AN, Zvyagintsev DG (2001) Microorganisms of Al-Fe-humus podzols under lichen pine forests affected by aerotechnogenic pollution. Eurasian Soil Science 34(2):190–200
Prescott C, McDonald M, Weetman G (1993) Availability of N and P in forest floors of adjacent stands of western red cedar–western hemlock and western hemlock–amabilis fir on northern Vancouver Island. Can J for res 23:605–610
Rautio P, Fürst A, Stefan K, Raitio H, Bartels U (2016) Part XII: sampling and analysis of needles and leaves. In: UNECE ICP Forests Programme Co-ordinating Centre (ed) Manual on methods and criteria for harmonized sampling, assessment, monitoring and analysis of the effects of air pollution on forests. Thünen Institute of Forest Ecosystems, Eberswalde, Germany 19 p. http://www.icp-forests.org/Manual.htm
Rief A, Knapp BA, Seeber J (2012) Palatability of selected alpine plant litters for the decomposer Lumbricus rubellus (Lumbricidae). PLoS One 7:e45345
Rigina O (1998) Introduction into the environmental problems in the Kola Peninsula//detection of pollution-induced forest decline in the Kola Peninsula using remote sensing and mathematical modeling. Licentiate Thesis Swedish University of Agricultural Sciences: Raport pp. 8–34.
Rowland AP, Roberts JD (1994) Lignin and cellulose fractions in decomposition studies using acid detergent fibre methods. Commun Soil Sci Plant Anal 25:269–277
Rusek J, Marshall VG (2000) Impacts of airborne pollutants on soil fauna. Ann Rev Ecol Syst 31:395–423
Scott N, Binkley D (1997) Litter quality and annual net N mineralization: comparisons across sites and species. Oecologia 111:151–159
StatSoft Inc (2010) STATISTICA [data analysis software system] Version 91 www.statsoft.com
Steinnes E, Sjøbakk TE (1992) Heavy metals in the soil-plant system in south Varanger. In: Effects of air pollutants on terrestrial ecosystems in the border area between Russia and Norway Oslo: State Pollution Control Authority, pp 86–92
Steinnes E, Lukina N, Nikonov V, Aamlid D, Royset O (2000) A gradient study of 34 elements in the vicinity of a copper-nickel smelter in the Kola Peninsula. Environ Monit Assess 60:71–88
Sterjiades R, Erikson KEL (1993) Biodegradation of lignins. In: Scalbert A (ed) Polyphenolic phenomena. INRA Editions, Paris, pp 115–126
Tuovinen J-P, Laurila T, Lättilä H, Ryaboshapko A, Brukhanov P et al (1993) Impact of the sulphur dioxide sources in the Kola Peninsula on air quality in northernmost. Europe Atmos Environ 27A:1379–1395
Vitousek PM, Turner DR, Parton WJ, Sanford RL (1994) Litter decomposition on the Mauna Loa environmental matrix, Hawaii: patterns, mechanisms, and model. Ecology 75:418–429
Vorob’ova LA (1998) Khimicheskiy analiz pochv [Soil chemical analysis]. Moscow State University Press, Moscow, Russia 272 p (in Russian)
Vorobeichik EL (1991) Changes in cellulose destruction rate under industrial impact. Russ J Ecol 22(6):73–76 (in Russian)
Wardle DA, Nilsson M-C, Zackrisson O, Gallet C (2003) Determinants of litter mixing effects in a Swedish boreal forest. Soil Biol Biochem 35:827–835
Zenkova I (2001) Soil-zoological research of natural and polluted soils at the Kola Peninsula. In: AMAP workshop on trends and effects 18–22 June, McLean. USA AMAP Report 2001(3):B76–B79
Zhang D, Hui D, Luo Y, Zhou G (2008) Rates of litter decomposition in terrestrial ecosystems: global patterns and controlling factors. J Plant Ecol 1:1–9
Acknowledgements
This work was supported by the Russian Science Foundation, project no. 16-17-10284.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Lukina, N.V., Orlova, M.A., Steinnes, E. et al. Mass-loss rates from decomposition of plant residues in spruce forests near the northern tree line subject to strong air pollution. Environ Sci Pollut Res 24, 19874–19887 (2017). https://doi.org/10.1007/s11356-017-9348-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9348-z