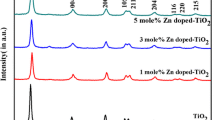
Abstract
We report a facile method to tune TiO2 nanoparticles’ morphology by modifying and an acid-catalyzed sol-gel synthesis with Pluronic P123. Synthesized particles were characterized by transmission electron microscopy, BET analysis, and X-ray diffraction spectroscopy. XRD analysis revealed a high anatase content while BET measurements showed that porous volume strongly depends on the amount of P123. We demonstrate that high amounts of P123 increase particle’s aspect-ratio from spherical to rod-shape morphology. We evaluated the photocatalytic performances for the removal of methyl viologen (paraquat) and found that best performances are obtained for the following weight ratio P123/TiO2 = 7.5. Furthermore, P25 is less active than synthesized nanoparticles.






Similar content being viewed by others
References
Andreozzi R (1999) Advanced oxidation processes (AOP) for water purification and recovery. Catal Today 53:51–59. doi:10.1016/S0920-5861(99)00102-9
Bu S, Jin Z, Liu X et al (2004) Fabrication of TiO2 porous thin films using peg templates and chemistry of the process. Mater Chem Phys 88:273–279. doi:10.1016/j.matchemphys.2004.03.033
Bu SJ, Jin ZG, Liu XX et al (2005) Synthesis of TiO2 porous thin films by polyethylene glycol templating and chemistry of the process. J Eur Ceram Soc 25:673–679. doi:10.1016/j.jeurceramsoc.2003.12.025
Calleja G, Serrano D, Sanz R (2004) Study on the synthesis of high-surface-area mesoporous TiO2 in the presence of nonionic surfactants. Ind Eng Chem Res 2485–2492. doi:10.1021/ie030646a
Carra I, Sanchez Perez JA, Malato S et al (2014) Performance of different advanced oxidation processes for tertiary wastewater treatment to remove the pesticide acetamiprid. J Chem Technol Biotechnol 91:72–81. doi:10.1002/jctb.4577
Christoforidis KC, Sengele A, Keller V, Keller N (2015) Single-step synthesis of SnS < inf > 2</inf > Nanosheet-decorated TiO < inf > 2</inf > Anatase nanofibers as efficient photocatalysts for the degradation of gas-phase diethylsulfide. ACS Appl Mater Interfaces 7:19324–19334. doi:10.1021/acsami.5b05370
Dinis-Oliveira RJ, Duarte JA, Sánchez-Navarro A et al (2008) Paraquat poisonings: mechanisms of lung toxicity, clinical features, and treatment. Crit Rev Toxicol 38:13–71. doi:10.1080/10408440701669959
Gomes A, Fernandes E, Lima JLFC (2005) Fluorescence probes used for detection of reactive oxygen species. J Biochem Biophys Methods 65:45–80. doi:10.1016/j.jbbm.2005.10.003
Hashimoto K, Irie H, Fujishima A (2007) A historical overview and future prospects. AAPPS Bull 17:12–28. doi:10.1007/BF00290457
He W, Liu Y, Wamer WG, Yin J-J (2014) Electron spin resonance spectroscopy for the study of nanomaterial-mediated generation of reactive oxygen species. J Food Drug Anal 22:49–63. doi:10.1016/j.jfda.2014.01.004
Ishibashi KI, Fujishima A, Watanabe T, Hashimoto K (2000) Detection of active oxidative species in TiO2 photocatalysis using the fluorescence technique. Electrochem Commun 2:207–210. doi:10.1016/S1388-2481(00)00006-0
Jahromi HS, Taghdisian H, Afshar S, Tasharrofi S (2009) Effects of pH and polyethylene glycol on surface morphology of TiO2 thin film. Surf Coatings Technol 203:1991–1996. doi:10.1016/j.surfcoat.2009.01.034
Jitputti J, Suzuki Y, Yoshikawa S (2008) Synthesis of TiO2 nanowires and their photocatalytic activity for hydrogen evolution. Catal Commun 9:1265–1271. doi:10.1016/j.catcom.2007.11.016
Lee J, Han S, Hyeon T (2004) Synthesis of new nanoporous carbon materials using nanostructured silica materials as templates. 478–486
Livage J, Henry M, Sanchez C (1988) Sol-gel chemistry of transition metal oxides. Prog Solid State Chem 18:259–341. doi:10.1016/0079-6786(88)90005-2
Livage J, Sanchez C, Henry MDS (1989) The chemistry of the sol gel process. Solid State Ionics 33:633–638
Louit G, Foley S, Cabillic J et al (2005) The reaction of coumarin with the OH radical revisited: hydroxylation product analysis determined by fluorescence and chromatography. Radiat Phys Chem 72:119–124. doi:10.1016/j.radphyschem.2004.09.007
Marien CBD, Cottineau T, Robert D, Drogui P (2016) TiO2 nanotube arrays: influence of tube length on the photocatalytic degradation of paraquat. Appl Catal B Environ 194:1–6. doi:10.1016/j.apcatb.2016.04.040
Moctezuma E, Leyva E, Monreal E et al (1999) Photocatalytic degradation of the herbicide “Paraquat.”. Chemosphere 39:511–517
Myilsamy M, Mahalakshmi M, Murugesan V, Subha N (2015) Enhanced photocatalytic activity of nitrogen and indium co-doped mesoporous TiO < inf > 2</inf > nanocomposites for the degradation of 2,4-dinitrophenol under visible light. Appl Surf Sci 342:1–10. doi:10.1016/j.apsusc.2015.03.017
Newton GL, Milligan JR (2006) Fluorescence detection of hydroxyl radicals. Radiat Phys Chem 75:473–478. doi:10.1016/j.radphyschem.2005.10.011
Nosaka Y, Komori S, Yawata K et al (2003) Photocatalytic •OH radical formation in TiO2 aqueous suspension studied by several detection methods. Phys Chem Chem Phys 5:4731–4735. doi:10.1039/b307433a
Organization WH (1984) Paraquat and diquat. International Programme on Chemical Safety
Peng T, Zhao D, Dai K et al (2005) Synthesis of titanium dioxide nanoparticles with mesoporous anatase wall and high photocatalytic activity. J Phys Chem B 109:4947–4952. doi:10.1021/jp044771r
Tian B, Yang H, Liu X et al (2002) Fast preparation of highly ordered nonsiliceous mesoporous materials via mixed inorganic precursors. Chem Commun (Camb):1824–1825. doi:10.1039/b205006d
Wang Z, Ma W, Chen C et al (2011) Probing paramagnetic species in titania-based heterogeneous photocatalysis by electron spin resonance (ESR) spectroscopy—a mini review. Chem Eng J 170:353–362. doi:10.1016/j.cej.2010.12.002
Wen J, Li X, Liu W et al (2015) Photocatalysis fundamentals and surface modification of TiO2 nanomaterials. Cuihua Xuebao/Chinese J Catal 36:2049–2070. doi:10.1016/S1872-2067(15)60999-8
Xiang Q, Yu J, Wong PK (2011) Quantitative characterization of hydroxyl radicals produced by various photocatalysts. J Colloid Interface Sci 357:163–167. doi:10.1016/j.jcis.2011.01.093
Yang G, Ding H, Chen D et al (2016) A simple route to synthesize mesoporous titania from TiOSO4: influence of the synthesis conditions on the structural, pigments and photocatalytic properties. Appl Surf Sci 376:227–235. doi:10.1016/j.apsusc.2016.03.156
Zhang J, Zhou P, Liu J, Yu J (2014) New understanding of the difference of photocatalytic activity among anatase, rutile and brookite TiO2. Phys Chem Chem Phys 16:20382–20386. doi:10.1039/C4CP02201G
Zhao XS, Su F, Yan Q et al (2006) Templating methods for preparation of porous structures. J Mater Chem 16:637. doi:10.1039/b513060c
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
ESM 1
(DOCX 59 kb)
Rights and permissions
About this article
Cite this article
Marien, C.B.D., Marchal, C., Koch, A. et al. Sol-gel synthesis of TiO2 nanoparticles: effect of Pluronic P123 on particle’s morphology and photocatalytic degradation of paraquat. Environ Sci Pollut Res 24, 12582–12588 (2017). https://doi.org/10.1007/s11356-016-7681-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-7681-2