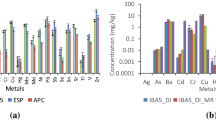
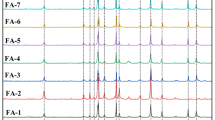
Abstract
Municipal solid waste incinerator (MSWI) fly ashes are characterized by high calcium oxide (CaO) content. Carbon dioxide (CO2) adsorption by MSWI fly ash was discussed based on thermogravimetry (TG)/differential thermal analysis (DTA), minerology analysis, and adapting the Stenoir equation. TG/DTA analysis showed that the weight gain of the fly ash below 440 °C was as high as 5.70 %. An adapted Stenoir equation for MSWI fly ash was discussed. The chloride in MSWI fly ash has a major impact on CO2 adsorption by MSWI fly ash or air pollution control (APC) residues. Geochemical modeling of the critical trace elements copper (Cu), cadmium (Cd), zinc (Zn), lead (Pb), and antimony (Sb) before and after carbonation was performed using a thermodynamic equilibrium model for solubility and a surface complexation model for metal sorption. Leaching of critical trace elements was generally found to be strongly dependent on the degree of carbonation attained, and their solubility appeared to be controlled by several minerals. Adsorption on ferrum (Fe) and aluminum (Al) colloids was also responsible for removal of the trace elements Cd, Pb, and Sb. We used Hakanson’s potential ecological risk index (HPERI) to evaluate the risk of trace element leaching in general. The results demonstrate that the ecological risk showed a V-shaped dependency on pH; the optimum pH of the carbonated fly ash was found to be 10.3–11, resulting from the optimum carbonation (liquid-to-solid (L/S) ratio = 0.25, carbonation duration = ∼30–48 h). The dataset and modeling results presented here provide a contribution to assessing the leaching behavior of MSWI fly ash under a wide range of conditions.






Similar content being viewed by others
References
Astrup T, Dijkstra LJ, Comans RNJ, Van Der Sloot HA, Christensen TH (2006a) Geochemical modeling of leaching from MSWI air-pollution-control residues. Environ Sci Technol 40:3551–3557
Astrup T, Mosbœk H, Christensen TH (2006b) Assessment of long-term leaching from waste incineration air-pollution-control residues. Waste Manag 26:803–814
Baciocchi R, Polettini A, Pomi R, Prigiobbe V, Zedwitz VNV, Steinfeld A (2006) CO2 sequestration by direct gas-solid carbonation of Air Pollution Control (APC) residues. Energy Fuel 20:1933–1940
Baciocchi R, Costa G, Bartolomeo ED, Polettini A, Pomi R (2009) The effects of accelerated carbonation on CO2 uptake and metal release from incineration APC residues. Waste Manag 29:2994–3003
Bayuseno AP, Schmahl WW (2011) Characterization of MSWI fly ash through mineralogy and water extraction. Resour Conserv Recycl 55:524–534
Boom AD, Degrez M (2012) Belgian MSWI fly ashes and APC residues: a characterisation study. Waste Manag 32:1163–1170
Cappai G, Cara S, Muntoni A, Piredda M (2012) Application of accelerated carbonation on MSW combustion APC residues for metal immobilization and CO2 sequestration. J Hazard Mater 207–208:159–164
Chandler AJ, Eighmy TT, Hartlem J, Hjelmar O, Kosson DS, Sawell SE, Van der Sloot HA, Verlow J (1997) Municipal solid waste incinerator residues. Elsevier Science, Amsterdam
Cho JS, Kim SM, Chun HD, Han GW, Lee CH (2011) Carbon dioxide capture with accelerated carbonation of industrial combustion waste. Int J Chem Eng Appl 2:60–65
Colangelo F, Messina F, Cioffi R (2015) Recycling of MSWI fly ash by means of cementitious double step cold bonding pelletization: technological assessment for the production of lightweight artificial aggregates. J Hazard Mater 299:181–191
Cornelis G, Van Gerven T, Vandecasteele C (2006) Antimony leaching from uncarbonated and carbonated MSWI bottom ash. J Hazard Mater 137:1284–1292
Cornelis G, Van Gerven T, Snellings R, Verbinnen B, Elsen J, Vandecasteele C (2011) Stability of pyrochlores in alkaline matrices: solubility of calcium antimonate. Appl Geochem 26:809–817
Cornelis G, Gerven TV, Vandecasteele C (2012) Antimony leaching from MSWI bottom ash: modelling of the effect of pH and carbonation. Waste Manag 32:278–286
Costa G, Baciocchi R, Polettini A, Pomi R, Hills CD, Carey PJ (2007) Current status and perspectives of accelerated carbonation processes on municipal waste combustion residues. Environ Monit Assess 135:55–75
De Boom A, Aubert JE, Degrez M (2014) Carbonation of municipal solid wasteincineration electrostatic precipitatorfly ashes in solution. Waste Manag Res 32:406–413
Dijkstra JJ, Van Der Sloot HA, Comans RNJ (2002) Process identification and model development of contaminant transport in MSWI bottom ash. Waste Manag 22:531–541
Dijkstra JJ, Van Der Sloot HA, Comans RNJ (2006) The leaching of major and trace elements from MSWI bottom ash as a function of pH and time. Appl Geochem 21:335–351
Dijkstra JJ, Meeussen JCL, Van der Sloot HA, Comans RNJ (2008) A consistent geochemical modelling approach for the leaching and reactive transport of major and trace elements in MSWI bottom ash. Appl Geochem 23:1544–1562
Dzombak DA, Morel FMM (1990) Surface complexation modeling: hydrous ferric oxide. Wiley, New York
Ecke H (2003) Sequestration of metals in carbonated municipal solid waste incineration (MSWI) fly ash. Waste Manag 23:631–640
Ecke H, Menad N, Lagerkvist A (2002) Treatment-oriented characterization of dry scrubber residue from municipal solid waste incineration. J Mater Cycles Waste Manag 4:117–126
Fernández Bertos M, Li X, Simons SJR, Hills CD, Carey PJ (2004) Investigation of accelerated carbonation for the stabilisation of MSW incinerator ashes and the sequestration of CO2. Green Chem 6:428–436
Freyssinet P, Piantone P, Azaroual M, Itard Y, Clozel-Leloup B, Guyonnet D, Baubron JC (2002) Chemical changes and leachate mass balance of municipal solid waste bottom ash submitted to weathering. Waste Manag 22:159–172
Gori M, Bergfeldt B, Pfrang-Stotz G, Reichelt J, Sirini P (2011) Effect of short-term natural weathering on MSWI and wood waste bottom ash leaching behaviour. J Hazard Mater 189:435–443
Hakanson L (1980) An ecological risk index for aquatic pollution control. A sedimentological approach. Water Res 14:975–1001
Halim CE, Short SA, Scott JA, Amal R, Low G (2005) Modelling the leaching of Pb, Cd, As, and Cr from cementitious waste using PHREEQC. J Hazard Mater 125:45–61
He PJ, Zhang H, Shao LM, Lee DJ (2006a) Leaching of carbonated air pollution control residues using compliance leaching tests. J Environ Qual 35:442–449
He PJ, Cao QK, Shao LM, Lee DJ (2006b) Aging of air pollution control residues from municipal solid waste incinerator: role of water content on metal carbonation. Sci Total Environ 359:26–37
Huntzinger DN, Gierke JS, Sutter LL, Kawatra KS, Eisele TC (2009) Mineral carbonation for carbon sequestration in cement kiln dust from waste. J Hazard Mater 168: 31–37
Hyks J, Astrup T, Christensen TH (2007) Influence of test conditions on solubility controlled leaching predictions from air-pollution-control residues. Waste Manag Res 25:457–466
Hyks J, Astrup T, Christensen TH (2009) Long-term leaching from MSWI air-pollution-control residues: Leaching characterization and modeling. J Hazard Mater 162:80--91
Hyks J, Nesterov I, Mogensen E, Jensen PA, Astrup T (2011) Leaching from waste incineration bottom ashes treated in a rotary kiln. Waste Manag Res 29:995–1007
Jiang JG, Chen MZ, Zhang Y, Xu X (2009) Pb stabilization in fresh fly ash from municipal solid waste incinerator using accelerated carbonation technology. J Hazard Mater 161:1046–1051
Keppert M, Pavlík Z, Tydlitát V, Volfová P, Švarcová S, Šyc M, Černý R (2012) Properties of municipal solid waste incineration ashes with respect to their separation temperature. Waste Manag Res 30(10):1041–1048
Kim SY, Tanaka N, Matsuto T, Tojo Y (2005) Leaching behaviour of elements and evaluation of pre-treatment methods for municipal solid waste incinerator residues in column leaching tests. Waste Manag Res 22:220–229
Kirby CS, Rimstidt JD (1994) Interaction of municipal solid waste ash with water. Environ Sci Technol 28:443–451
Kosson DS, van Der Sloot HA, Sanchez F (2002) An integrated framework for evaluating leaching in waste management and utilization of secondary materials. Environ Eng Sci 19:159–204
Lam CHK, Ip AWM, Barford JP, McKay G (2010) Use of incineration MSW ash: a review. Sustainability 2:1943–1968
Lee G, Bigham JM, Faure G (2002) Removal of trace metals by coprecipitation with Fe, Al and Mn from natural waters contaminated with acid mine drainage in the Ducktown Mining District, Tennessee. Appl Geochem 17:569–581
Leuz AK, Hug SI, Wehrli B, Johnson CA (2006) Iron-mediated oxidation of antimony (III) by oxygen and hydrogen peroxide compared to arsenic (III) oxidation. Environ Sci Technol 40:2565–2571
Li X, Fernández Bertos M, Hills CD, Carey PJ and Simon S (2007) Accelerated carbonation of municipal solid waste incineration fly ashes. Waste Management 27:1200–1206
Lindberg D, Molin C, Hupa M (2015) Thermal treatment of solid residues from WtE units: a review. Waste Manag 37:82–94
Liu JG, Liu F, Zheng P, Nie YF (2006) Geochemical modeling of leaching behavior of heavy metals in municipal solid waste incinerator fly ashes. Chin J Geochem 25:83
Margallo M, Taddei MBM, Hernández-Pellón A, Aldaco R, Irabien Á (2015) Environmental sustainability assessment of the management of municipal solid waste incineration residues: a review of the current situation. Clean Techn Environ Policy 17:1333–1353
Meima JA, Comans RNJ (1997) Geochemical modeling of weathering reactions in municipal solid waste incinerator bottom ash. Environ Sci Technol 31:1269–1276
Meima JA, Comans RNJ (1998a) Reducing Sb-leaching from municipal solid waste incinerator bottom ash by addition of sorbent minerals. J Geochem Explor 62:299–304
Meima JA, Comans RNJ (1998b) Application of surface complexation/precipitation modeling to contaminant leaching from weathered municipal solid waste incinerator bottom ash. Environ Sci Technol 32:688–693
Meima JA, Comans RNJ (1999) The leaching of trace elements from municipal solid waste incinerator bottom ash at different stages of weathering. Appl Geochem 14:159–171
Meima JA, Van Der Weijden RD, Eighmy TT, Comans RNJ (2002) Carbonation processes in municipal solid waste incinerator bottom ash and their effect on the leaching of copper and molybdenum. Appl Geochem 17:1503–1513
Ministry of Environmental Protection, the People’s Republic of China (2010) Solid waste–extraction procedure for leaching toxicity–horizontal vibration method (HJ 557 2010)
National Bureau of Statistics of the People’s Republic of China (2014) China statistical yearbook. http://www.stats.gov.cn/tjsj/ndsj/2014/zk/html/Z0820e.htm
Parkhurst DL, Appelo CAJ (2005) User’s guide to PHREEQC (version 3)—a computer program for speciation batch reaction, one dimensional transport and inverse geochemical calculations. Water Resour Inv Report 99–4259, Denver CO: U.S. Geol. Surv, pp 688–693
Pedersen AJ, Frandsen FJ, Riber C, Astrup T, Thomsen SN, Lundtorp K, Mortensen LF (2009) A full-scale study on the partitioning of trace elements in municipal solid waste incineration—effects of firing different waste types. Energy Fuel 23:3475–3489
Pedersen AJ, Lith SC, Frandsen FJ, Steinsen SD, Holgersen LB (2010) Release to the gas phase of metals, S and Cl during combustion of dedicated waste fractions. Fuel Process Technol 91:1062–1072
Polettini A, Pomi R (2004) The leaching behavior of incinerator bottom ash as affected by accelerated ageing. J Hazard Mater B113:209–215
Qu M, Li W, Zhang C (2014) Spatial distribution and uncertainty assessment of potential ecological risks of heavy metals in soil using sequential Gaussian simulation. Hum Ecol Risk Assess 20:764–778
Quina MJ, Bordado JC, Quinta-Ferreira RM (2008) Treatment and use of air pollution control residues from MSW incineration: an overview. Waste Manag 28:2097–2121
Quina MJ, Bordado JCM, Quinta-Ferreira RM (2009) The influence of pH on the leaching behaviour of inorganic components from municipal solid waste APC residues. Waste Manag 29:2483–2493
Quina MJ, Bordado JCM, Quinta-Ferreira RM (2011) Chemical stabilization of air pollution control residues from municipal solid waste incineration. J Hazard Mater 179:382–392
Rani DA, Boccaccini AR, Deegan D, Cheeseman CR (2008) Air pollution control residues from waste incineration: current UK situation and assessment of alternative technologies. Waste Manag 28:2279–2292
Riber C, Fredriksen GS, Christensen TH (2005) Heavy metal content of combustible municipal solid waste in Denmark. Waste Manag Res 23:126–132
Struis RPWJ, Ludwig C, Lutz H, Scheidegger AM (2004) Speciation of Zinc in municipal solid waste incineration fly ash after heat treatment: an X-ray absorption spectroscopy study. Environ Sci Technol 38:3760–3767
Sun YF, Watanabe N, Qiao W, Gao X, Wang W, Zhu T (2010) Polysulfide as a novel chemical agent to solidify/stabilize lead in fly ash from municipal solid waste incineration. Chemosphere 81:120–126
Tian SC, Jiang JG, Zhang C (2011) Influence of flue gas SO2 on the toxicity of heavy metals in municipal solid waste incinerator fly ash after accelerated carbonation stabilization. J Hazard Mater 192:1609–1615
Todorovic J, Ecke H (2006a) Treatment of MSWI residues for utilization as secondary construction minerals: a review of methods. Minerals & Energy 3–4:45–59
Todorovic J, Ecke H (2006b) Demobilisation of critical contaminants in four typical waste-to-energy ashes by carbonation. Waste Manag 26:430–441
Todorovic J, Svensson M, Herrmann I, Ecke H (2006) Artificial carbonation for controlling the mobility of critical elements in bottom ash. J Mater Cycles Waste Manag 8:145–153
Van der Bruggen B, Vogels G, Van Herck P, Vandecasteele C (1998) Simulation of acid washing of municipal solid waste incineration fly ashes in order to remove heavy metals. J Hazard Mater 57:127–144
Van Gerven T, Van Keer E, Arickx S, Jaspers M, Wauters G, Vandecasteele C (2005) Carbonation of MSWI bottom ash to decrease heavy metal leaching, in view of recycling. Waste Manag 25:291–300
Van Herck P, Van der Bruggen B, Vogels G, Vandecasteele C (2000) Application of computer modelling to predict the leaching behaviour of heavy metals from MSWI fly ash and comparison with a sequential extraction method. Waste Manag 20:203–210
Wang L, Jin Y, Nie Y (2010) Investigation of accelerated and natural carbonation of MSWI fly ash with a high content of Ca. J Hazard Mater 174:334–343
Wilcox J (2012) Carbon Capture. Springer, New York
Wu SM, Xu YF, Sun J, Cao ZB, Zhou JZ, Pan Y, Qian GR (2015) Inhibiting evaporation of heavy metal by controlling its chemical speciation in MSWI fly ash. Fuel 158:764–769
Yao J, Li WB, Tang ML, Fang CR, Feng HJ, Shen DS (2010) Effect of weathering treatment on the fractionation and leaching behavior of copper in municipal solid waste incinerator bottom ash. Chemosphere 81:571–576
Ye H, Zang S, Xiao H, Zhang L (2015) Speciation and ecological risk of heavy metals and metalloid in the sediments of Zhalong Wetland in China. Int J Environ Sci Technol 12:115–124
Zacco A, Borgese L, Gianoncelli A, Struis RPWJ, Depero LE, Bontempi E (2014) Review of fly ash inertisation treatments and recycling. Environ Chem Lett 12:153–175
Zhang H, He PJ, Shao LM, Feng JH, Cao QK (2006) Leaching behavior of Pb and Zn in air pollution control residues and their modeling prediction. J Environ Sci (China) 18:583–586
Zhang H, He PJ, Shao LM, Lee DJ (2008a) Temporary stabilization of air pollution control residues using carbonation. Waste Manag 28:509–517
Zhang Y, Jiang JG, Chen MZ (2008b) MINTEQ modeling for evaluating the leaching behavior of heavy metals in MSWI fly ash. J Environ Sci 20:1398–1402
Zhang HY, Zhao YC, Qi JY (2010) Thermal characterization of fly ash from one municipal solid waste incinerator (MSWI) in Shanghai. Process Saf Environ Prot 88:269–275
Zhu F, Masaki T, Kenji S, Kazuyuki O, Yoshinori K (2008) Chloride chemical form in various types of fly ash. Environ Sci Technol 42:3932–3937
Acknowledgments
Financial support from the National Science Foundation (No. 51108276) in China is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Wang, L., Chen, Q., Jamro, I.A. et al. Geochemical modeling and assessment of leaching from carbonated municipal solid waste incinerator (MSWI) fly ash. Environ Sci Pollut Res 23, 12107–12119 (2016). https://doi.org/10.1007/s11356-016-6320-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-6320-2