Abstract
Purpose
Follicle-stimulating hormone receptor (FSHR) is overexpressed in primary and metastatic tumor. Molecular imaging of FSHR is beneficial for prognosis and therapy of cancer. FSHβ(33–53) (YTRDLVYKDPARPKIQKTCTF), denoted as FSH1, is a FSHR antagonist. In the present study, maleimide-NOTA conjugate of FSH1 (NOTA-MAL-FSH1) was designed and labeled with [18F] aluminum fluoride. The resulting tracer, 18F-Al-NOTA-MAL-FSH1, was preliminarily evaluated in PET imaging of FSHR-positive tumor.
Procedures
NOTA-MAL-FSH1 was synthesized and radiolabeled with Al18F complex. The tumor-targeting potential and pharmacokinetic profile of the 18F-labeled compound were evaluated in vitro and in vivo using a PC3 human prostate tumor model.
Results
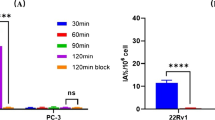
18F-Al-NOTA-MAL-FSH1 can be efficiently produced within 30 min with a non-decay-corrected yield of 48.6 ± 2.1 % and a radiochemical purity of more than 95 %. The specific activity was at least 30 GBq/μmol. The radiotracer was stable in phosphate-buffered saline and human serum for at least 2 h. The IC50 values of displacement 18F-Al-NOTA-MAL-FSH1 with FSH1 were 252 ± 1.12 nM. The PC3 human prostate tumor xenografts were clearly visible with high contrast after injection of 18F-Al-NOTA-MAL-FSH1 via microPET. At 30, 60 and 120 min postinjection, the tumor uptakes were 2.98 ± 0.29 % injected dose (ID)/g, 2.53 ± 0.20 %ID/g and 1.36 ± 0.12 %ID/g, respectively. Dynamic PET scanning showed that tumor uptake reached a plateau by about 6 min. Heart peaked earlier and then cleared quickly. Biodistribution studies confirmed that the normal organs except kidney uptakes were all below 1 %ID/g at 1 h p.i. The tumor-to-blood and tumor-to-muscle ratio at 10 min, 0.5, 1, and 2 h after injection were 1.64 ± 0.36, 2.97 ± 0.40, 9.31 ± 1.06, and 13.59 ± 2.33 and 7.05 ± 1.10, 10.10 ± 1.48, 16.17 ± 3.29, and 30.88 ± 4.67, respectively. The tracer was excreted mainly through the renal system, as evidenced by high levels of radioactivity in the kidneys. FSHR-binding specificity was also demonstrated by reduced tumor uptake of 18F-Al-NOTA-MAL-FSH1 after coinjection with an excess of unlabeled FSH1 peptide.
Conclusion
NOTA-MAL-FSH1 could be labeled rapidly and efficiently with 18F using one step method. Favorable preclinical data suggest that 18F-Al-NOTA-MAL-FSH1 may be a suitable radiotracer for the non-invasive visualization of FSHR positive tumor in vivo.






Similar content being viewed by others
References
Robinson LJ, Tourkova I, Wang Y et al (2010) FSH-receptor isoforms and FSH-dependent gene transcription in human monocytes and osteoclasts. Biochem Biophys Res Commun 394:12–17
Simoni M, Gromoll J, Nieschlag E (1997) The follicle-stimulating hormone receptor: biochemistry, molecular biology, physiology, and pathophysiology. Endocr Rev 18:739–773
Gartrell BA, Tsao CK, Galsky MD et al (2013) The follicle-stimulating hormone receptor: a novel target in genitourinary malignancies. Urol Oncol: Semin Orig Inv 31:1403–1407
Radu A, Pichon C, Camparo P et al (2010) Expression of follicle-stimulating hormone receptor in tumor blood vessels. N Engl J Med 363:1621–1630
Siraj A, Desestret V, Antoine M et al (2013) Expression of follicle-stimulating hormone receptor by the vascular endothelium in tumor metastases. BMC Cancer 13:246
Siraj MA, Pichon C, Radu A et al (2012) Endothelial follicle stimulating hormone receptor in primary kidney cancer correlates with subsequent response to sunitinib. J Cell Mol Med 16:2010–2016
Grasso P, Santa-Coloma TA, Reichert LE Jr (1991) Synthetic peptides corresponding to human follicle stimulating hormone (hFSH)-h-(1-15) and hFSH-h-(51-65) induce uptake of 45Ca++ by liposomes: evidence for calcium-conducting transmembrane channel formation. Endocrinology 128:2745–2751
Agris PF, Guenther RH, Sierzputowska-Gracz H et al (1992) Solution structure of a synthetic peptide corresponding to a receptor binding region of FSH (hFSH-β33-53). J Protein Chem 11:495–507
Zhang XY, Chen J, Zheng YF et al (2009) Follicle-stimulating hormone peptide can facilitate paclitaxel nanoparticles to target ovarian carcinoma in vivo. Cancer Res 69:6506–6514
Zaheer A, Cho SY, Pomper MG (2009) New agents and techniques for imaging prostate cancer. J Nucl Med 50:1387–1390
Hong H, Zhang Y, Sun JT et al (2010) Positron emission tomography imaging of prostate cancer. Amino Acids 39:11–27
Fani M, Maecke HR (2012) Radiopharmaceutical development of radiolabelled peptides. Eur J Nucl Med Mol Imaging 39:S11–S30
Laverman P, D'Souza CA, Eek A et al (2012) Optimized labeling of NOTA-conjugated octreotide with F-18. Tumour Biol 33:427–434
Liu S, Liu H, Jiang H et al (2011) One-step radiosynthesis of 18F-AlF-NOTA-RGD2 for tumor angiogenesis PET imaging. Eur J Nucl Med Mol Imaging 38:1732–1741
Dijkgraaf I, Franssen GM, McBride WJ et al (2012) PET of tumors expressing gastrin-releasing peptide receptor with an 18F-labeled bombesin analog. J Nucl Med 53:947–952
Wan W, Guo N, Pan D et al (2013) First experience of 18F-alfatide in lung cancer patients using a new lyophilized kit for rapid radiofluorination. J Nucl Med 54:691–698
Ait-Mohand S, Fournier P, Dumulon-Perreault V et al (2011) Evaluation of 64Cu-labeled bifunctional chelate-bombesin conjugates. Bioconjug Chem 22:1729–1735
Yang M, Gao H, Zhou Y et al (2011) 18F-labeled bombesin receptor agonist and antagonist: a comparative study in prostate cancer imaging. Theranostics 1:220–229
Ambrosini V, Fani M, Fanti S et al (2011) Radiopeptide imaging and therapy in Europe. J Nucl Med 52:42S–55S
Graham MM, Menda Y (2011) Radiopeptide imaging and therapy in the United States. J Nucl Med 52:56S–63S
Koopmans KP, Glaudemans AW (2012) Rationale for the use of radiolabelled peptides in diagnosis and therapy. Eur J Nucl Med Mol Imaging 39:S4–S10
McBride WJ, D'Souza CA, Karacay H et al (2012) New lyophilized kit for rapid radiofluorination of peptides. Bioconjug Chem 23:538–547
Lang L, Li W, Guo N et al (2011) Comparison study of [18F]FAl-NOTA-PRGD2, [18F]FPPRGD2, and [68Ga]Ga-NOTA-PRGD2 for PET imaging of U87MG tumors in mice. Bioconjug Chem 22:2415–2422
McBride WJ, Sharkey RM, Karacay H et al (2009) A novel method of 18F radiolabeling for PET. J Nucl Med 50:991–998
Kiesewetter DO, Guo N, Guo J et al (2012) Evaluation of an [18F]AlF-NOTA analog of exendin-4 for imaging of GLP-1 receptor in insulinoma. Theranostics 2:999–1009
Mariani S, Salvatori L, Basciani S et al (2006) Expression and cellular localization of follicle-stimulating hormone receptor in normal human prostate, benign prostatic hyperplasia and prostate cancer. J Urol 175:2072–2077
Parry JJ, Kelly TS, Andrews R et al (2007) In vitro and in vivo evaluation of 64Cu-labeled DOTA-linker-bombesin(7-14) analogues containing different amino acid linker moieties. Bioconjug Chem 18:1110–1117
Acknowledgments
This work was partially supported by National Natural Science Foundation (81171399 and 81101077), CSC Foundation (2011832173), National Significant New Drugs Creation Program (2012ZX09505-001-001), Jiangsu Province Science and Technology Foundation (BE2012622, BK2011166 and BL2012031), Health Ministry of Jiangsu Province Fund (RC2011095 and H201028), public service platform for science and technology infrastructure construction project of Jiangsu Province (BM2012066), and University of Wisconsin-Madison Department of Medical Physics and Department of Radiology (Radiology R&D Award 1105-002).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Donghui Pan equally contributes to the work.
Rights and permissions
About this article
Cite this article
Xu, Y., Pan, D., Zhu, C. et al. Pilot Study of a Novel 18F-labeled FSHR Probe for Tumor Imaging. Mol Imaging Biol 16, 578–585 (2014). https://doi.org/10.1007/s11307-013-0712-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-013-0712-1