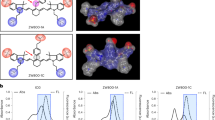
Abstract
Purpose
The goal of the study was to investigate a method for the real-time assessment of a target concentration in vivo using a combination of a spectral unmixing technique and a fluorescent probe specific for amyloid beta (Aβ) species, the biomarkers for Alzheimer’s disease (AD).
Procedures
The probe CRANAD-3 has a significant emission wavelength shift upon binding to Aβ species. It was used to differentiate a bound probe from an unbound probe in a phantom, ex vivo in brain slices and whole brain, and in vivo in a transgenic mouse model of AD.
Results
The ex vivo unmixing imaging of AD brain clearly showed differential distribution of the bound and unbound probes between the brain tissue and blood vessels. The in vivo unmixed signals of bound CRANAD-3 reached a plateau with increasing dosage, demonstrating that these signals correspond to Aβ content, not probe injected dose.
Conclusion
This study provided evidence that signals processed by the spectral unmixing technique could be used as a real-time reporter of Aβ species loading in vivo and ex vivo.






Similar content being viewed by others
References
Xu H, Rice BW (2009) In-vivo fluorescence imaging with a multivariate curve resolution spectral unmixing technique. J Biomed Opt 14:064011
Mayes P, Dicker D, Liu Y, El-Deiry W (2008) Noninvasive vascular imaging in fluorescent tumors using multispectral unmixing. Biotechniques 45:459–464
Naik S, Piwnica-Worms D (2007) Real-time imaging of beta-catenin dynamics in cells and living mice. Proc Natl Acad Sci USA 104:17465–17470
Zimmermann T (2005) Spectral imaging and linear unmixing in light microscopy. Adv Biochem Eng Biotechnol 95:245–265
Liu J, Lau SK, Varma VA, Kairdolf BA, Nie S (2010) Multiplexed detection and characterization of rare tumor cells in Hodgkin's lymphoma with multicolor quantum dots. Anal Chem 82:6237–6243
Raymond SB, Skoch J, Hills ID, Nesterov EE, Swager TM, Bacskai BJ (2008) Smart optical probes for near-infrared fluorescence imaging of Alzheimer's disease pathology. Eur J Nucl Med Mol Imaging 35:s93–s98
Ran C, Xu X, Raymond SB et al (2009) Design, synthesis, and testing of difluoroboron-derivatized curcumins as near-infrared probes for in vivo detection of amyloid-beta deposits. J Am Chem Soc 131:15257–15261
Nesterov EE, Skoch J, Hyman BT, Klunk WE, Bacskai BJ, Swager TM (2005) In vivo optical imaging of amyloid aggregates in brain: design of fluorescent markers. Angew Chem Int Ed Engl 44:5452–5456
Mizusawa K, Ishida Y, Takaoka Y, Miyagawa M, Tsukiji S, Hamachi I (2010) Disassembly-driven turn-on fluorescent nanoprobes for selective protein detection. J Am Chem Soc 132:7291–7293
Dickinson ME, Bearman G, Tille S, Lansford R, Fraser SE (2001) Multi-spectral imaging and linear unmixing add a whole new dimension to laser scanning fluorescence microscopy. Biotechniques 31:1272, 1274–1276, 1278
Ran C, Zhao W, Moir R, Moore A (2011) Non-conjugated small molecule FRET for differentiating monomers from higher molecular weight amyloid beta species. PLoS One 6:e19362
Selkoe D (2008) Soluble oligomers of the amyloid beta-protein impair synaptic plasticity and behavior. Behav Brain Res 192:106–113
Haass C, Selkoe DJ (2007) Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer's amyloid beta-peptide. Nat Rev Mol Cell Biol 8:101–112
Greenspan P, Fowler SD (1985) Spectrofluorometric studies of the lipid probe, Nile red. J Lipid Res 26:781–789
Jankowsky JL, Fadale DJ, Anderson J et al (2004) Mutant presenilins specifically elevate the levels of the 42 residue beta-amyloid peptide in vivo: evidence for augmentation of a 42-specific gamma secretase. Hum Mol Genet 13:159–170
Reiserer RS, Harrison FE, Syverud DC, McDonald MP (2007) Impaired spatial learning in the APPSwe + PSEN1DeltaE9 bigenic mouse model of Alzheimer's disease. Genes Brain Behav 6:54–65
Hintersteiner M, Enz A, Frey P et al (2005) In vivo detection of amyloid-beta deposits by near-infrared imaging using an oxazine-derivative probe. Nat Biotechnol 23:577–583
Gurskaya NG, Verkhusha VV, Shcheglov AS et al (2006) Engineering of a monomeric green-to-red photoactivatable fluorescent protein induced by blue light. Nat Biotechnol 24:461–465
Lukyanov KA, Chudakov DM, Lukyanov S, Verkhusha VV (2005) Innovation: photoactivatable fluorescent proteins. Nat Rev Mol Cell Biol 6:885–891
Johnson I (1998) Fluorescent probes for living cell. Histochem J 30:123–140
Acknowledgments
This work was supported in part by K25AG036760 award to C.R. We would like to thank Marytheresa Ifediba for proofreading this manuscript.
Conflict of interest
The authors declare they have no conflict of interests pertinent to this study.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
SI Fig. 1
Raw images of phantom imaging with CRANAD-3 and Aβ 42 species in PBS buffer. The raw images were obtained with 535-nm excitation and with 14 emission filters ranging from 580 to 840 nm. (PDF 220 kb)
SI Fig. 2
Representative raw image sequence of in vivo imaging of an APP/PS1 mouse with CRANAD-3 (Ex = 570 nm, Em = 620–840 nm with 12 emission filters). (PDF 316 kb)
SI Fig. 3
Fluorescent microscopic images of ex vivo brain slice of APP/PS1 mouse. a Image of Aβ plaque distribution in whole brain slice with ×2 lens. b Image of Aβ plaque in the cortex area (white box in a) with ×10 lens. (PDF 1751 kb)
Rights and permissions
About this article
Cite this article
Ran, C., Moore, A. Spectral Unmixing Imaging of Wavelength-Responsive Fluorescent Probes: An Application for the Real-Time Report of Amyloid Beta Species in Alzheimer’s Disease. Mol Imaging Biol 14, 293–300 (2012). https://doi.org/10.1007/s11307-011-0501-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-011-0501-7