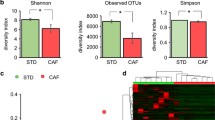
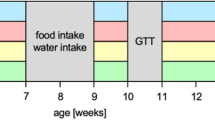
Abstract
Obesity is a major health concern that poses significant risks for many other diseases, including diabetes, cardiovascular disease, and cancer. Prevalence of these diseases varies by biological sex. This study utilizes a mouse (C57BL/6J) model of obesity to analyze liver and fecal metabolic profiles at various time points of dietary exposure: 5, 9, and 12 months in control or high fat diet (HFD)-exposed mice. Our study discovered that the female HFD group has a more discernable perturbation and set of significant changes in metabolic profiles than the male HFD group. In the female mice, HFD fecal metabolites including pyruvate, aspartate, and glutamate were lower than control diet-exposed mice after both 9th and 12th month exposure time points, while lactate and alanine were significantly downregulated only at the 12th month. Perturbations of liver metabolic profiles were observed in both male and female HFD groups, compared to controls at the 12th month. Overall, the female HFD group showed higher lactate and glutathione levels compared to controls, while the male HFD group showed higher levels of glutamine and taurine compared to controls. These metabolite-based findings in both fecal and liver samples for a diet-induced effect of obesity may help guide future pioneering discoveries relating to the analysis and prevention of obesity in people, especially for females.







Similar content being viewed by others
Data availability
The raw data supporting the conclusions of this article will be made available by the authors without undue reservation.
References
Adult Obesity Causes & Consequences | Overweight & Obesity | CDC. (2021). https://www.cdc.gov/obesity/adult/causes.html.
Agellon, L. B. (2002). Chapter 16 Metabolism and function of bile acids. In New Comprehensive Biochemistry (Vol. 36, pp. 433–448). Elsevier. https://doi.org/10.1016/S0167-7306(02)36018-6.
Allam-Ndoul, B., Guénard, F., Garneau, V., Barbier, O., Pérusse, L., & Vohl, M. C. (2015). Associations between branched chain amino acid levels, obesity and cardiometabolic Complications. Integrative Obesity and Diabetes, 1(6), 157–162. https://doi.org/10.15761/IOD.1000134.
Aron-Wisnewsky, J., Warmbrunn, M. V., Nieuwdorp, M., & Clément, K. (2021). Metabolism and metabolic disorders and the Microbiome: The intestinal microbiota Associated with obesity, lipid metabolism, and Metabolic Health—Pathophysiology and therapeutic strategies. Gastroenterology, 160(2), 573–599. https://doi.org/10.1053/J.GASTRO.2020.10.057.
Blachier, F., Boutry, C., Bos, C., & Tomé, D. (2009). Metabolism and functions of l-glutamate in the epithelial cells of the small and large intestines. The American Journal of Clinical Nutrition, 90(3), 814S–821S. https://doi.org/10.3945/AJCN.2009.27462S.
Broadfield, L. A., Duarte, J. A. G., Schmieder, R., Broekaert, D., Veys, K., Planque, M., Vriens, K., Karasawa, Y., Napolitano, F., Fujita, S., Fujii, M., Eto, M., Holvoet, B., Vangoitsenhoven, R., Fernandez-Garcia, J., Van Elsen, J., Dehairs, J., Zeng, J., Dooley, J., & Fendt, S. M. (2021). Fat induces glucose metabolism in nontransformed liver cells and promotes liver tumorigenesis. Cancer Research, 81(8), 1988–2001. https://doi.org/10.1158/0008-5472.CAN-20-1954/673641/AM/FAT-INDUCES-GLUCOSE-METABOLISM-IN-NON-TRANSFORMED.
Burrin, D. G., & Reeds, P. J. (1997). Alternative fuels in the gastrointestinal tract. Current Opinion in Gastroenterology, 13(2), 165–170. https://doi.org/10.1097/00001574-199703000-00015.
Chiang, J. Y. L. (2013). Bile acid metabolism and signaling. Comprehensive Physiology, 3(3), 1191. https://doi.org/10.1002/CPHY.C120023.
Cuevas-Sierra, A., Ramos-Lopez, O., Riezu-Boj, J. I., Milagro, F. I., & Martinez, J. A. (2019). Diet, Gut Microbiota, and obesity: Links with Host Genetics and epigenetics and potential applications. Advances in Nutrition, 10(suppl_1), S17–S30. https://doi.org/10.1093/ADVANCES/NMY078.
Druart, C., Bindels, L. B., Schmaltz, R., Neyrinck, A. M., Cani, P. D., Walter, J., Ramer-Tait, A. E., & Delzenne, N. M. (2015). Ability of the gut microbiota to produce PUFA-derived bacterial metabolites: Proof of concept in germ-free versus conventionalized mice. Molecular Nutrition and Food Research, 59(8), 1603–1613. https://doi.org/10.1002/mnfr.201500014.
Ferslew, B. C., Xie, G., Johnston, C. K., Su, M., Stewart, P. W., Jia, W., Brouwer, K. L. R., & Barritt, S. (2015). Altered bile acid metabolome in patients with nonalcoholic steatohepatitis. Digestive Diseases and Sciences, 60(11), 3318–3328. https://doi.org/10.1007/s10620-015-3776-8
Ghosh, S., Sulistyoningrum, D. C., Glier, M. B., Verchere, C. B., & Devlin, A. M. (2011). Altered glutathione homeostasis in heart augments cardiac lipotoxicity associated with diet-induced obesity in mice. The Journal of Biological Chemistry, 286(49), 42483–42493. https://doi.org/10.1074/jbc.M111.304592.
Gómez-Zorita, S., Aguirre, L., Milton-Laskibar, I., Fernández-Quintela, A., Trepiana, J., Kajarabille, N., Mosqueda-Solís, A., González, M., & Portillo, M. P. (2019). Relationship between changes in Microbiota and Liver Steatosis Induced by High-Fat Feeding—A review of Rodent models. Nutrients 2019, 11(9), 2156. https://doi.org/10.3390/NU11092156. 11.
Gratton, J., Phetcharaburanin, J., Mullish, B. H., Williams, H. R. T., Thursz, M., Nicholson, J. K., Holmes, E., Marchesi, J. R., & Li, J. V. (2016). Optimized Sample Handling Strategy for metabolic profiling of human feces. Analytical Chemistry, 88(9), 4661–4668. https://doi.org/10.1021/acs.analchem.5b04159.
Green, C. J., & Hodson, L. (2014). The influence of Dietary Fat on Liver Fat Accumulation. Nutrients, 6, 5018–5033. https://doi.org/10.3390/nu6115018.
Hajer, G. R., Van Haeften, T. W., & Visseren, F. L. J. (2008). Adipose tissue dysfunction in obesity, Diabetes, and vascular Diseases. European Heart Journal (Vol, 29, 2959–2971. https://doi.org/10.1093/eurheartj/ehn387. Oxford Academic.
Hales, C. M., Carroll, M. D., Fryar, C. D., & Ogden, C. L. (2015). Prevalence of Obesity Among Adults and Youth: United States, 2015–2016 Key findings Data from the National Health and Nutrition Examination Survey. https://www.cdc.gov/nchs/data/databriefs/db288_table.pdf#1.
Heianza, Y., & Qi, L. (2017). Gene-diet interaction and precision nutrition in obesity. International Journal of Molecular Sciences (Vol, 18(4), https://doi.org/10.3390/ijms18040787. MDPI AG.
Hori, S., Abe, T., Lee, D. G., Fukiya, S., Yokota, A., Aso, N., Shirouchi, B., Sato, M., & Ishizuka, S. (2020). Association between 12α-hydroxylated bile acids and hepatic steatosis in rats fed a high-fat diet. Journal of Nutritional Biochemistry, 83. https://doi.org/10.1016/j.jnutbio.2020.108412.
Hubbard, K., Shome, A., Sun, B, Pontré, B., McGregor, A., & Mountjoy, K. G. (2019). Chronic high-fat diet exacerbates sexually dimorphic Pomctm1/tm1 mouse obesity. Endocrinology, 160(5), 1081–1096.
Ivanisevic, J., & Thomas, A. (2018). Metabolomics as a tool to understand pathophysiological processes. Methods in Molecular Biology, 1730, 3–28. https://doi.org/10.1007/978-1-4939-7592-1_1/COVER/.
Jacobs, S. A. H., Gart, E., Vreeken, D., Franx, B. A. A., Wekking, L., Verweij, V. G. M., Worms, N., Schoemaker, M. H., Gross, G., Morrison, M. C., Kleemann, R., Arnoldussen, I. A. C., & Kiliaan, A. J. (2019). Sex-specific differences in Fat Storage, Development of non-alcoholic fatty Liver Disease and Brain structure in Juvenile HFD-Induced obese Ldlr-/-.Leiden Mice. Nutrients 2019, 11(8), 1861. https://doi.org/10.3390/NU11081861. 11.
Jéquier, E. (2002). Pathways to obesity. International Journal of Obesity, 26, S12–S17. https://doi.org/10.1038/sj.ijo.0802123.
Katz, P. P., Yazdany, J., Trupin, L., Schmajuk, G., Margaretten, M., Barton, J., Criswell, L. A., & Yelin, E. H. (2013). Sex differences in assessment of obesity in rheumatoid arthritis. Arthritis Care & Research, 65(1), 62–70. https://doi.org/10.1002/ACR.21810.
Kimura, I., Inoue, D., Hirano, K., & Tsujimoto, G. (2014). The SCFA receptor GPR43 and energy metabolism. Frontiers in Endocrinology, 5(JUN), 85. https://doi.org/10.3389/fendo.2014.00085.
Kimura, I., Ozawa, K., Inoue, D., Imamura, T., Kimura, K., Maeda, T., Terasawa, K., Kashihara, D., Hirano, K., Tani, T., Takahashi, T., Miyauchi, S., Shioi, G., Inoue, H., & Tsujimoto, G. (2013). The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nature Communications, 4(1), 1829. https://doi.org/10.1038/ncomms2852.
Le, L., Jiang, B., Wan, W., Zhai, W., Xu, L., Hu, K., & Xiao, P. (2016). Metabolomics reveals the protective of Dihydromyricetin on glucose homeostasis by enhancing insulin sensitivity. Scientific Reports, 6(1), 1–11. https://doi.org/10.1038/srep36184.
Lin, H., An, Y., Tang, H., & Wang, Y. (2019a). Alterations of bile acids and gut microbiota in obesity Induced by High Fat Diet in Rat Model. Journal of Agricultural and Food Chemistry, 67(13), 3624–3632. https://doi.org/10.1021/acs.jafc.9b00249.
Lin, H., An, Y., Tang, H., & Wang, Y. (2019b). Alterations of bile acids and gut microbiota in obesity Induced by High Fat Diet in Rat Model. Journal of Agricultural and Food Chemistry, 67(13), 3624–3632. https://doi.org/10.1021/ACS.JAFC.9B00249/ASSET/IMAGES/LARGE/JF-2019-00249E_0006.JPEG.
Liu, R., Hong, J., Xu, X., Feng, Q., Zhang, D., Gu, Y., Shi, J., Zhao, S., Liu, W., Wang, X., Xia, H., Liu, Z., Cui, B., Liang, P., Xi, L., Jin, J., Ying, X., Wang, X., Zhao, X., & Wang, W. (2017). Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nature Medicine, 23(7), 859–868. https://doi.org/10.1038/NM.4358.
Lombardi, C., & Dicks, L. M. T. (2022). Gut Bacteria and Neurotransmitters. Microorganisms 2022, Vol. 10, Page 1838, 10(9), 1838. https://doi.org/10.3390/MICROORGANISMS10091838.
Lynch, C. J., & Adams, S. H. (2014). Branched-chain amino acids in metabolic signalling and insulin resistance. Nature Reviews Endocrinology, 10(12), 723–736. https://doi.org/10.1038/nrendo.2014.171.
Mardinoglu, A., Shoaie, S., Bergentall, M., Ghaffari, P., Zhang, C., Larsson, E., Bäckhed, F., & Nielsen, J. (2015). The gut microbiota modulates host amino acid and glutathione metabolism in mice. Molecular Systems Biology, 11(10), 834. https://doi.org/10.15252/MSB.20156487.
Mauvais-Jarvis, F. (2015). Sex differences in metabolic homeostasis, Diabetes, and obesity. Biology of Sex Differences, 6(1), 1–9. https://doi.org/10.1186/s13293-015-0033-y.
Mayengbam, S., Mickiewicz, B., Trottier, S. K., Mu, C., Wright, D. C., Reimer, R. A., Vogel, H. J., & Shearer, J. (2019). Distinct gut microbiota and serum metabolites in response to weight loss Induced by either dairy or Exercise in a rodent model of obesity. Journal of Proteome Research, 18(11), 3867–3875. https://doi.org/10.1021/acs.jproteome.9b00304.
Mazzoli, R., & Pessione, E. (2016). The Neuro-endocrinological role of Microbial Glutamate and GABA Signaling. Frontiers in Microbiology, 7(NOV), 1934. https://doi.org/10.3389/fmicb.2016.01934.
Meiliana, A., Dewi, N. M., & Wijaya, A. (2021). Metabolomics: An Emerging Tool for Precision Medicine. The Indonesian Biomedical Journal, 13(1), 1–18. https://doi.org/10.18585/INABJ.V13I1.1309.
Murakami, Y., Tanabe, S., & Suzuki, T. (2016). High-fat Diet-induced intestinal hyperpermeability is Associated with increased bile acids in the large intestine of mice. Journal of Food Science, 81(1), H216–H222. https://doi.org/10.1111/1750-3841.13166.
Newgard, C. B. (2017). Metabolomics and metabolic Diseases: Where do we stand? Cell Metabolism, 25(1), 43–56. https://doi.org/10.1016/J.CMET.2016.09.018.
Newsholme, P., Lima, M. M. R., Procopio, J., Pithon-Curi, T. C., Doi, S. Q., Bazotte, R. B., & Curi, R. (2003). Glutamine and glutamate as vital metabolites. Brazilian Journal of Medical and Biological Research, 36(2), 153–163. https://doi.org/10.1590/S0100-879X2003000200002.
Nicholson, J. K., Lindon, J. C., & Holmes, E. (1999). Metabonomics: Understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica (Vol. 29, pp. 1181–1189). Taylor and Francis Ltd. 11https://doi.org/10.1080/004982599238047.
Palomo-Buitrago, M. E., Sabater-Masdeu, M., Moreno-Navarrete, J. M., Caballano-Infantes, E., Arnoriaga-Rodríguez, M., Coll, C., Ramió, L., Palomino-Schätzlein, M., Gutiérrez-Carcedo, P., Pérez-Brocal, V., Simó, R., Moya, A., Ricart, W., Herance, J. R., & Fernández-Real, J. M. (2019). Glutamate interactions with obesity, insulin resistance, cognition and gut microbiota composition. Acta Diabetologica, 56(5), 569–579. https://doi.org/10.1007/s00592-019-01313-w.
Pataky, Z., Bobbioni-Harsch, E., & Golay, A. (2010). Obesity: A complex growing challenge. In Experimental and Clinical Endocrinology and Diabetes (Vol. 118, Issue 7, pp. 427–433). MVS Medizinverlage Stuttgart. https://doi.org/10.1055/s-0029-1233448.
Piché, M. E., Tchernof, A., & Després, J. P. (2020). Obesity phenotypes, Diabetes, and Cardiovascular Diseases. Circulation Research, 1477–1500. https://doi.org/10.1161/CIRCRESAHA.120.316101.
Rakhra, V., Galappaththy, L., Bulchandani, S., & Cabandugama, P. K. (2020). Obesity and the Western diet: how We Got here. Missouri Medicine, 117:6(November/December), 536–538.
Ramasamy, A., Laliberté, F., Aktavoukian, S. A., Lejeune, D., DerSarkissian, M., Cavanaugh, C., Smolarz, B. G., Ganguly, R., & Duh, M. S. (2019). Direct and indirect cost of obesity among the privately insured in the United States: A focus on the impact by type of industry. Journal of Occupational and Environmental Medicine, 61(11), 877–886. https://doi.org/10.1097/JOM.0000000000001693.
Rodríguez-Carmona, Y., Meijer, J. L., Zhou, Y., Jansen, E. C., Perng, W., Banker, M., Song, P. X. K., Téllez-Rojo, M. M., Cantoral, A., & Peterson, K. E. (2022). Metabolomics reveals sex-specific pathways associated with changes in adiposity and muscle mass in a cohort of Mexican adolescents. Pediatric Obesity, 17(6), e12887. https://doi.org/10.1111/IJPO.12887.
Ruiz, B., Felipe, A., Casado, J., & Pastor-Anglada, M. (1991). Amino acid uptake by liver of genetically obese Zucker rats. Biochemical Journal, 280(2), 367–372. https://doi.org/10.1042/BJ2800367.
Soontornniyomkij, V., Kesby, J. P., Soontornniyomkij, B., Kim, J. J., Kisseleva, T., Achim, C. L., Semenova, S., & Jeste, D. V. (2016). Age and High-Fat Diet effects on glutamine synthetase immunoreactivity in Liver and Hippocampus and Recognition Memory in mice. Current Aging Science, 9(4), 301–309. https://doi.org/10.2174/1874609809666160413113311.
Szymańska, E., Bouwman, J., Strassburg, K., Vervoort, J., Kangas, A. J., Soininen, P., Ala-Korpela, M., Westerhuis, J., Van Duynhoven, J. P. M., Mela, D. J., MacDonald, I. A., Vreeken, R. J., Smilde, A. K., & Jacobs, D. M. (2012). Gender-dependent associations of metabolite profiles and body fat distribution in a healthy population with central obesity: Towards metabolomics diagnostics. OMICS a Journal of Integrative Biology, 16(12), 652–667. https://doi.org/10.1089/OMI.2012.0062/ASSET/IMAGES/LARGE/FIGURE4.JPEG.
Varani, J., McClintock, S. D., Knibbs, R. N., Harber, I., Zeidan, D., Jawad-Makki, M. A. H., & Aslam, M. N. (2022). Liver protein expression in NASH mice on a high-Fat Diet: Response to multi-mineral intervention. Frontiers in Nutrition, 0, 862. https://doi.org/10.3389/FNUT.2022.859292.
Wang, B., Maldonado-Devincci, A. M., & Jiang, L. (2020). Evaluating line-broadening factors on a reference spectrum as a bucketing method for NMR based metabolomics. Analytical Biochemistry, 606, 113872. https://doi.org/10.1016/j.ab.2020.113872.
White, P. J., McGarrah, R. W., Herman, M. A., Bain, J. R., Shah, S. H., & Newgard, C. B. (2021). Insulin action, type 2 diabetes, and branched-chain amino acids: A two-way street. In Molecular Metabolism (Vol. 52). Elsevier GmbH. https://doi.org/10.1016/j.molmet.2021.101261.
Windmueller, H. G., & Spaeth, A. E. (1975). Intestinal metabolism of glutamine and glutamate from the lumen as compared to glutamine from blood. Archives of Biochemistry and Biophysics, 171(2), 662–672. https://doi.org/10.1016/0003-9861(75)90078-8.
Won, E. Y., Yoon, M. K., Kim, S. W., Jung, Y., Bae, H. W., Lee, D., Park, S. G., Lee, C. H., Hwang, G. S., & Chi, S. W. (2013). Gender-specific metabolomic profiling of obesity in Leptin-Deficient ob/ob mice by 1H NMR spectroscopy. Plos One, 8(10), e75998. https://doi.org/10.1371/journal.pone.0075998.
Wu, H., Southam, A. D., Hines, A., & Viant, M. R. (2008). High-throughput tissue extraction protocol for NMR- and MS-based metabolomics. Analytical Biochemistry, 372(2), 204–212. https://doi.org/10.1016/j.ab.2007.10.002.
Xie, B., Waters, M. J., & Schirra, H. J. (2012). Investigating potential mechanisms of obesity by metabolomics. Journal of Biomedicine & Biotechnology, 2012, 805683. https://doi.org/10.1155/2012/805683.
Xie, G., Ma, X., Zhao, A., Wang, C., Zhang, Y., Nieman, D., Nicholson, J. K., Jia, W., Bao, Y., & Jia, W. (2014). The metabolite profiles of the obese Population are gender-dependent. Journal of Proteome Research, 13(9), 4062–4073. https://doi.org/10.1021/pr500434s.
Yang, H., Wang, X., Xiong, X., & Yin, Y. (2016). Energy metabolism in intestinal epithelial cells during maturation along the crypt-villus axis. Scientific Reports, 6. https://doi.org/10.1038/SREP31917.
Yang, Q., Vijayakumar, A., & Kahn, B. B. (2018). Metabolites as regulators of insulin sensitivity and metabolism. Nature Reviews Molecular Cell Biology 2018 19:10, 19(10), 654–672. https://doi.org/10.1038/s41580-018-0044-8.
Yang, X., Dong, B., An, L., Zhang, Q., Chen, Y., Wang, H., & Song, Z. (2021). Ginsenoside Rb1 ameliorates Glycemic Disorder in Mice With High Fat Diet-Induced Obesity via Regulating Gut Microbiota and Amino Acid Metabolism. Frontiers in Pharmacology, 12. https://doi.org/10.3389/fphar.2021.756491.
Yoshitsugu, R., Kikuchi, K., Hori, S., Iwaya, H., Hagio, M., Shimizu, H., Hira, T., & Ishizuka, S. (2020). Correlation between 12α-hydroxylated bile acids and insulin secretion during glucose tolerance tests in rats fed a high-fat and high-sucrose diet. Lipids in Health and Disease, 19(1), https://doi.org/10.1186/s12944-020-1193-2.
Yoshitsugu, R., Kikuchi, K., Iwaya, H., Fujii, N., Hori, S., Lee, D. G., & Ishizuka, S. (2019). Alteration of bile acid metabolism by a high-Fat Diet is Associated with plasma transaminase activities and glucose intolerance in rats. Journal of Nutritional Science and Vitaminology, 65(1), 45–51. https://doi.org/10.3177/JNSV.65.45.
Yu, J. C., Jiang, Z. M., & Li, D. M. (1999). Glutamine: A precursor of glutathione and its effect on liver. World Journal of Gastroenterology, 5(2), 143. https://doi.org/10.3748/WJG.V5.I2.143.
Zeng, H., Umar, S., Rust, B., Lazarova, D., & Bordonaro, M. (2019). Secondary bile acids and short chain fatty acids in the Colon: A focus on colonic microbiome, cell proliferation, inflammation, and Cancer. International Journal of Molecular Sciences 2019, 20(5), 1214. https://doi.org/10.3390/IJMS20051214. 20.
Zhang, S., Zeng, X., Ren, M., Mao, X., & Qiao, S. (2017). Novel metabolic and physiological functions of branched chain amino acids: A review. Journal of Animal Science and Biotechnology, 2017 8:1(1), 1–12. https://doi.org/10.1186/S40104-016-0139-Z. 8.
Funding
This study was supported by the 2021 North Carolina Agriculture and Technical State University NCTracs Interdisciplinary Team Award (JH, AMD, BW, SHH), the National Center for Advancing Translational Sciences, National Institutes of Health, through Grant KL2TR002490 (AMD), and the National Science Foundation through Grant 2245530 (BW).
Author information
Authors and Affiliations
Contributions
BW and VJ performed metabolomics experiments and data analysis. BW, VJ, and JH wrote the manuscripts. AO, EC, and AC carried out mice husbandry and physiological experiments. SHH and AMD revised the manuscript. JH, BW, and AMD designed the experiments and provided general project guidance for students. JLG revised the manuscript and supported the graduate student AO to conduct the research.
Corresponding author
Ethics declarations
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. All authors have read the version and approved it to be published.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, B., Jadhav, V., Odelade, A. et al. High fat diet reveals sex-specific fecal and liver metabolic alterations in C57BL/6J obese mice. Metabolomics 19, 97 (2023). https://doi.org/10.1007/s11306-023-02059-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-023-02059-x