Abstract
Background
Ischemic stroke is one of the leading causes of death and adult disability. The incidence of ischemic stroke continues to rise in young adults. This study aimed to provide a comprehensive evaluation of metabolic changes and explore possible mechanisms in young ischemic stroke patients without common risk factors.
Methods
This study investigated serum metabolomics in 50 young patients with newly suffered ischemic stroke and 50 age-, sex-, and body mass index–matched healthy controls. Liquid chromatography coupled with a Waters Xevo TQ-S mass spectrometer with an electrospray ionization (ESI) source was used to analyze amino acid or bile acid, and free fatty acid or lipid was analyzed by liquid chromatography coupled with a Qtrap5500 mass spectrometer with an ESI source. The metabolomic data were analyzed by performing a multivariate statistical analysis.
Results
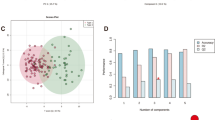
A total of 197 metabolites, including amino acids, bile acids, free fatty acids, and lipids, were identified in all participants. Multivariate models showed significant differences in serum metabolomic patterns between young patients with ischemic stroke and healthy controls. The stroke patients had increased L-methionine, homocysteine, glutamine, uric acid, GCDCA, and PE (18:0/20:4, 16:0/22:5), and decreased levels of L-citrulline, taurine, PC (16:2/22:6, 16:2/20:5, 15:0/18:2), and SM (d18:1/23:0, d20:0/19:1, d18:1/22:0, d16:0/26:1, d16:0/18:0, d16:0/22:1, d18:1/19:1, d16:0/17:1, d16:1/24:1, d18:1/19:0). Based on the identified metabolites, the metabolic pathways of arginine biosynthesis, glycerophospholipid metabolism, and taurine and hypotaurine metabolism were significantly enriched in the young patients with ischemic stroke.
Conclusions
Serum metabolomic patterns were significantly different between young patients with ischemic stroke and healthy controls. Our study is beneficial in providing a further view into the pathophysiology of young patients with ischemic stroke.



Similar content being viewed by others
Abbreviations
- BMI:
-
Body weight index
- TC:
-
Total cholesterol
- HDL-C:
-
High-density lipoprotein cholesterol
- LDL-C:
-
Low-density lipoprotein cholesterol
- TG:
-
Triglycerides
- UA:
-
Uric acid
- ESI:
-
Electrospray ionization
- CV:
-
Coefficients of variation
- QC:
-
Quality control
- VIP:
-
Variable influences on projection
- PCA:
-
Principal component analysis
- OPLS-DA:
-
Orthogonal partial least-squares discriminant analysis
- GCDCA:
-
Glycochenodeoxycholic acid
- PE:
-
Phosphatidylethanolamine
- PC:
-
Phosphatidylcholine
- SM:
-
Sphingomyelin
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- NO:
-
Nitric oxide
References
Alewijnse, A. E., & Peters, S. L. (2008). Sphingolipid signalling in the cardiovascular system: Good, bad or both? European Journal of Pharmacology, 585, 292–302.
Allerton, T. D., Proctor, D. N., Stephens, J. M., Dugas, T. R., Spielmann, G., & Irving, B. A. (2018). l-Citrulline supplementation: Impact on cardiometabolic health. Nutrients, 10(7), 921.
Chong, J., Wishart, D. S., & Xia, J. (2019). Using MetaboAnalyst 4.0 for comprehensive and integrative metabolomics data analysis. Current Protocols in Bioinformatics, 68, e86.
Chung, S. Y., Moriyama, T., Uezu, E., Uezu, K., Hirata, R., Yohena, N., et al. (1995). Administration of phosphatidylcholine increases brain acetylcholine concentration and improves memory in mice with dementia. Journal of Nutrition, 125, 1484–1489.
Crespo Pimentel, B., Willeit, J., Toll, T., Kiechl, S., Pinho, E. M. T., Canhao, P., et al. (2019). Etiologic evaluation of ischemic stroke in young adults: A comparative study between two European Centers. Journal of Stroke and Cerebrovascular Diseases, 28, 1261–1266.
Ekker, M. S., Boot, E. M., Singhal, A. B., Tan, K. S., Debette, S., Tuladhar, A. M., & de Leeuw, F. E. (2018). Epidemiology, aetiology, and management of ischaemic stroke in young adults. Lancet Neurology, 17, 790–801.
Ekker, M. S., Verhoeven, J. I., Vaartjes, I., van Nieuwenhuizen, K. M., Klijn, C. J. M., & de Leeuw, F. E. (2019). Stroke incidence in young adults according to age, subtype, sex, and time trends. Neurology, 92, e2444–e2454.
Garry, P. S., Ezra, M., Rowland, M. J., Westbrook, J., & Pattinson, K. T. (2015). The role of the nitric oxide pathway in brain injury and its treatment–from bench to bedside. Experimental Neurology, 263, 235–243.
Holmes, M. V., Millwood, I. Y., Kartsonaki, C., Hill, M. R., Bennett, D. A., Boxall, R., et al. (2018). Lipids, lipoproteins, and metabolites and risk of myocardial infarction and stroke. Journal of the American College of Cardiology, 71, 620–632.
Jakaria, M., Azam, S., Haque, M. E., Jo, S. H., Uddin, M. S., Kim, I. S., & Choi, D. K. (2019). Taurine and its analogs in neurological disorders: Focus on therapeutic potential and molecular mechanisms. Redox Biol, 24, 101223.
Jove, M., Mauri-Capdevila, G., Suarez, I., Cambray, S., Sanahuja, J., Quilez, A., et al. (2015). Metabolomics predicts stroke recurrence after transient ischemic attack. Neurology, 84, 36–45.
Ke, C., Pan, C. W., Zhang, Y., Zhu, X., & Zhang, Y. (2019). Metabolomics facilitates the discovery of metabolic biomarkers and pathways for ischemic stroke: A systematic review. Metabolomics, 15, 152.
Kim, Y. S., Kim, E. K., Hwang, J. W., Kim, J. S., Shin, W. B., Dong, X., et al. (2017). Neuroprotective effect of taurine-rich Cuttlefish (Sepia officinalis) extract against hydrogen peroxide-induced oxidative stress in SH-SY5Y cells. Advances in Experimental Medicine and Biology, 975(Pt 1), 243–254.
Li, J., Muraki, I., Imano, H., Cui, R., Yamagishi, K., Umesawa, M., et al. (2020). Serum uric acid and risk of stroke and its types: The Circulatory Risk in Communities Study (CIRCS). Hypertension Research, 43(4), 313–321.
Lind, L., Salihovic, S., Ganna, A., Sundstrom, J., Broeckling, C. D., Magnusson, P. K., et al. (2020). A multi-cohort metabolomics analysis discloses sphingomyelin (32:1) levels to be inversely related to incident ischemic stroke. Journal of Stroke and Cerebrovascular Diseases, 29, 104476.
Liu, J., Fu, J., Jia, Y., Yang, N., Li, J., & Wang, G. (2020a). Serum metabolomic patterns in patients with autoimmune thyroid disease. Endocrine Practise, 26, 82–96.
Liu, M., Zhou, K., Li, H., Dong, X., Tan, G., Chai, Y., et al. (2015). Potential of serum metabolites for diagnosing post-stroke cognitive impairment. Molecular BioSystems, 11, 3287–3296.
Liu, P., Li, R., Antonov, A. A., Wang, L., Li, W., Hua, Y., et al. (2017). Discovery of metabolite biomarkers for acute ischemic stroke progression. Journal of Proteome Research, 16, 773–779.
Liu, Y., Croft, K. D., Hodgson, J. M., Mori, T., & Ward, N. C. (2020b). Mechanisms of the protective effects of nitrate and nitrite in cardiovascular and metabolic diseases. Nitric Oxide, 96, 35–43.
Luo, L., Aubrecht, J., Li, D., Warner, R. L., Johnson, K. J., Kenny, J., & Colangelo, J. L. (2018). Assessment of serum bile acid profiles as biomarkers of liver injury and liver disease in humans. PLoS ONE, 13, e0193824.
Luo, L. L., Li, Y. F., Shan, H. M., Wang, L. P., Yuan, F., Ma, Y. Y., et al. (2019). L-glutamine protects mouse brain from ischemic injury via up-regulating heat shock protein 70. CNS Neuroscience & Therapeutics, 25, 1030–1041.
Maaijwee, N. A., Rutten-Jacobs, L. C., Schaapsmeerders, P., van Dijk, E. J., & de Leeuw, F. E. (2014). Ischaemic stroke in young adults: Risk factors and long-term consequences. Nat Rev Neurol, 10, 315–325.
Marderstein, E. L., Bucher, B., Guo, Z., Feng, X., Reid, K., & Geller, D. A. (2003). Protection of rat hepatocytes from apoptosis by inhibition of c-Jun N-terminal kinase. Surgery, 134, 280–284.
McCarty, J. L., Leung, L. Y., Peterson, R. B., Sitton, C. W., Sarraj, A., Riascos, R. F., & Brinjikji, W. (2019). Ischemic infarction in young adults: A review for radiologists. Radiographics, 39, 1629–1648.
Mortality, G.B.D. and Causes of Death, C. (2016). Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet, 388, 1459–1544.
Niazi, F., Aslam, A., Khattak, S., & Waheed, S. (2019). Frequency of homocysteinemia in young ischemic stroke patients and its relationship with the early outcome of a stroke. Cureus, 11, e5625.
Niebergall, L. J., Jacobs, R. L., Chaba, T., & Vance, D. E. (2011). Phosphatidylcholine protects against steatosis in mice but not non-alcoholic steatohepatitis. Biochimica et Biophysica Acta, 1811, 1177–1185.
Pexa, A., Boeger, R. H., Henle, T., Schwedhelm, E., & Deussen, A. (2008). Effects of moderate hyperhomocysteinaemia induced by 4 weeks methionine-enriched diet on metabolite profile and mesenteric artery function in rats. British Journal of Nutrition, 99, 993–999.
Ramonet, D., Rodriguez, M. J., Fredriksson, K., Bernal, F., & Mahy, N. (2004). In vivo neuroprotective adaptation of the glutamate/glutamine cycle to neuronal death. Hippocampus, 14, 586–594.
Ramstedt, B., & Slotte, J. P. (2002). Membrane properties of sphingomyelins. FEBS Letters, 531, 33–37.
Siriratnam, P., Godfrey, A., O’Connor, E., Pearce, D., Hu, C. C., Low, A., et al. (2019). Prevalence and risk factors of ischemic stroke in the young; a regional Australian perspective. Internal Medicine Journal, 50(6), 698–704.
Sun, D., Tiedt, S., Yu, B., Jian, X., Gottesman, R. F., Mosley, T. H., et al. (2019). A prospective study of serum metabolites and risk of ischemic stroke. Neurology, 92, e1890–e1898.
Sun, H., Zhao, J., Zhong, D., & Li, G. (2017). Potential serum biomarkers and metabonomic profiling of serum in ischemic stroke patients using UPLC/Q-TOF MS/MS. PLoS ONE, 12, e0189009.
Taranukhin, A. G., Taranukhina, E. Y., Saransaari, P., Podkletnova, I. M., Pelto-Huikko, M., & Oja, S. S. (2010). Neuroprotection by taurine in ethanol-induced apoptosis in the developing cerebellum. Journal of Biomedical Science, 17(Suppl 1), S12.
Vance, J. E., & Tasseva, G. (2013). Formation and function of phosphatidylserine and phosphatidylethanolamine in mammalian cells. Biochimica et Biophysica Acta, 1831, 543–554.
Wang, D., Kong, J., Wu, J., Wang, X., & Lai, M. (2017). GC-MS-based metabolomics identifies an amino acid signature of acute ischemic stroke. Neuroscience Letters, 642, 7–13.
Watanabe, M., Houten, S. M., Mataki, C., Christoffolete, M. A., Kim, B. W., Sato, H., et al. (2006). Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature, 439, 484–489.
Watanabe, M., Houten, S. M., Wang, L., Moschetta, A., Mangelsdorf, D. J., Heyman, R. A., et al. (2004). Bile acids lower triglyceride levels via a pathway involving FXR, SHP, and SREBP-1c. Journal of Clinical Investigation, 113, 1408–1418.
Yang, Z., Kusumanchi, P., Ross, R. A., Heathers, L., Chandler, K., Oshodi, A., et al. (2019). Serum metabolomic profiling identifies key metabolic signatures associated with pathogenesis of alcoholic liver disease in humans. Hepatology Commununications, 3, 542–557.
Zabot, G. P., Carvalhal, G. F., Marroni, N. P., Hartmann, R. M., da Silva, V. D., & Fillmann, H. S. (2014). Glutamine prevents oxidative stress in a model of mesenteric ischemia and reperfusion. World Journal of Gastroenterology, 20, 11406–11414.
Acknowledgements
This work was supported by grants from the Beijing Talents foundation (2018-12) and Capital Funds for Health Improvement and Research (No. 2018-4-2034) to J.L.; and the Chinese National Natural Science Foundation (No. 81770792) and Key Projects of Science and Technology Planning of Beijing Municipal Education Commission (KZ201810025038) to G.W.
Author information
Authors and Affiliations
Contributions
GW conceived and designed the experiments; JL, JLY, and JWZ performed the experiments; JL, JLY, LZ and QW analyzed and interpreted the patient data; JL and JLY wrote the manuscript; all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
The study protocol was approved by the Ethics Committee of Beijing Chaoyang Hospital Affiliated with Capital Medical University (application number: 2016-K103).
Informed consent
Informed consent was obtained from all individual participants involved in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, J., Yuan, J., Zhao, J. et al. Serum metabolomic patterns in young patients with ischemic stroke: a case study. Metabolomics 17, 24 (2021). https://doi.org/10.1007/s11306-021-01774-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-021-01774-7