Abstract
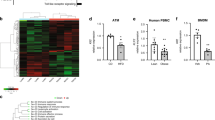
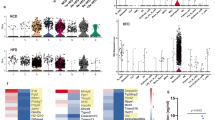
The purinergic receptor P2Y2 binds ATP to control chemotaxis of myeloid cells, and global P2Y2 receptor knockout mice are protected in models of acute inflammation. Chronic inflammation mediated by macrophages and other immune cells in adipose tissue contributes to the development of insulin resistance. Here, we investigate whether mice lacking P2Y2 receptors on myeloid cells are protected against acute and chronic inflammation. Wild-type mice were transplanted with either wild-type or P2Y2 receptor null bone marrow and treated with a sublethal dose of endotoxin as a model of acute inflammation, or fed a high-fat diet to induce obesity and insulin resistance as a model of chronic inflammation. P2Y2−/− chimeric mice were protected against acute inflammation. However, high-fat diet feeding induced comparable inflammation and insulin resistance in both WT and P2Y2−/− chimeric mice. Of note, confocal microscopy revealed significantly fewer crown-like structures, assemblies of macrophages around adipocytes, in P2Y2−/− chimeric mice compared to WT chimeric mice. We conclude that P2Y2 receptors on myeloid cells are important in mediating acute inflammation but are dispensable for the development of whole body insulin resistance in diet-induced obese mice.





Similar content being viewed by others
References
Junger WG (2011) Immune cell regulation by autocrine purinergic signalling. Nat Rev Immunol 11:201–212
Eltzschig HK, Sitkovsky MV, Robson SC (2012) Purinergic signaling during inflammation. N Engl J Med 367:2322–2333
Cekic C, Linden J (2016) Purinergic regulation of the immune system. Nature reviews. Immunology 16:177–192
Inoue Y, Chen Y, Hirsh MI, Yip L, Junger WG (2007) A3 and P2Y2 receptors control the recruitment of neutrophils to the lungs in a mouse model of sepsis. Shock 1
Ayata CK, Ganal SC, Hockenjos B, Willim K, Vieira RP, Grimm M et al (2012) Purinergic P2Y2 receptors promote neutrophil infiltration and hepatocyte death in mice with acute liver injury. Gastroenterology 143:1620–1629.e4
Desai BN, Leitinger N (2014) Purinergic and calcium signaling in macrophage function and plasticity. Front Immunol 5:580
Eun SY, Seo J, Park SW, Lee JH, Chang KC, Kim HJ (2014) LPS potentiates nucleotide-induced inflammatory gene expression in macrophages via the upregulation of P2Y2 receptor. Int Immunopharmacol 18:270–276
Elliott MR, Chekeni FB, Trampont PC, Lazarowski ER, Kadl A, Walk SF et al (2009) Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature 461:282–286
Chekeni FB, Elliott MR, Sandilos JK, Walk SF, Kinchen JM, Lazarowski ER et al (2010) Pannexin 1 channels mediate ‘find-me’ signal release and membrane permeability during apoptosis. Nature 467:863–867
Adamson SE, Leitinger N (2014) The role of pannexin1 in the induction and resolution of inflammation. FEBS Lett 588:1416–1422
Burnstock G, Pelleg A (2014) Cardiac purinergic signalling in health and disease. Purinergic Signal 11:1–46
Burnstock G, Novak I (2013) Purinergic signalling and diabetes. Purinergic Signal 9:307–324
Sun K, Kusminski CM, Scherer PE (2011) Adipose tissue remodeling and obesity. J Clin Investig 121:2094–2101
Weisberg SP, Hunter D, Huber R, Lemieux J, Slaymaker S, Vaddi K et al (2006) CCR2 modulates inflammatory and metabolic effects of high-fat feeding. J Clin Investig 116:115–124
Patsouris D, Li P, Thapar D, Chapman J, Olefsky JM, Neels JG (2008) Ablation of CD11c-positive cells normalizes insulin sensitivity in obese insulin resistant animals. Cell Metab 8:301–309
Stienstra R, van Diepen JA, Tack CJ, Zaki MH, van de Veerdonk FL, Perera D et al (2011) Inflammasome is a central player in the induction of obesity and insulin resistance. Proc Natl Acad Sci U S A 108:15324–15329
Vandanmagsar B, Youm YH, Ravussin A, Galgani JE, Stadler K, Mynatt RL et al (2011) The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat Med 17:179–188
Sun S, Xia S, Ji Y, Kersten S, Qi L (2012) The ATP-P2X7 signaling axis is dispensable for obesity-associated inflammasome activation in adipose tissue. Diabetes 61:1471–1478
Meher AK, Sharma PR, Lira VA, Yamamoto M, Kensler TW, Yan Z et al (2012) Nrf2 deficiency in myeloid cells is not sufficient to protect mice from high-fat diet-induced adipose tissue inflammation and insulin resistance. Free Radic Biol Med 52:1708–1715
Linton MF, Atkinson JB, Fazio S (1995) Prevention of atherosclerosis in apolipoprotein E-deficient mice by bone marrow transplantation. Science (New York, NY) 267:1034–1037
Kadl A, Meher AK, Sharma PR, Lee MY, Doran AC, Johnstone SR et al (2010) Identification of a novel macrophage phenotype that develops in response to atherogenic phospholipids via Nrf2. Circ Res 107:737–746
Adamson SE, Griffiths R, Moravec R, Senthivinayagam S, Montgomery G, Chen W et al (2016) Disabled homolog 2 controls macrophage phenotypic polarization and adipose tissue inflammation. J Clin Invest 126:1311–1322
Ayala JE, Samuel VT, Morton GJ, Obici S, Croniger CM, Shulman GI et al (2010) Standard operating procedures for describing and performing metabolic tests of glucose homeostasis in mice. Dis Model Mech 3:525–534
Shaul ME, Bennett G, Strissel KJ, Greenberg AS, Obin MS (2010) Dynamic, M2-like remodeling phenotypes of CD11c+ adipose tissue macrophages during high-fat diet-induced obesity in mice. Diabetes 59:1171–1181
Wentworth JM, Naselli G, Brown WA, Doyle L, Phipson B, Smyth GK et al (2010) Pro-inflammatory CD11c+CD206+ adipose tissue macrophages are associated with insulin resistance in human obesity. Diabetes 59:1648–1656
Nishida M, Ogushi M, Suda R, Toyotaka M, Saiki S, Kitajima N et al (2011) Heterologous down-regulation of angiotensin type 1 receptors by purinergic P2Y2 receptor stimulation through S-nitrosylation of NF-kappaB. Proc Natl Acad Sci U S A 108:6662–6667
Thomas AC, Mattila JT (2014) “Of mice and men”: arginine metabolism in macrophages. Front Immunol 5:479
Caldwell RB, Toque HA, Narayanan SP, Caldwell RW (2015) Arginase: an old enzyme with new tricks. Trends Pharmacol Sci 36:395–405
Kobayashi Y (2010) The regulatory role of nitric oxide in proinflammatory cytokine expression during the induction and resolution of inflammation. J Leukoc Biol 88:1157–1162
Kanda H (2006) MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J Clin Investig 116:1494–1505
Tanaka M, Ikeda K, Suganami T, Komiya C, Ochi K, Shirakawa I et al (2014) Macrophage-inducible C-type lectin underlies obesity-induced adipose tissue fibrosis. Nat Commun 5:4982
Corriden R, Insel PA (2012) New insights regarding the regulation of chemotaxis by nucleotides, adenosine, and their receptors. Purinergic Signal 8:587–598
Funding
This study was supported by grants from the National Institutes of Health NIH-P01HL120840 (to NL). SEA was supported by an AHA predoctoral fellowship and by an NIH training grant (HL007284).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Samantha E. Adamson declares that she has no conflict of interest.
Garren Montgomery declares that he has no conflict of interest.
Scott A. Seaman declares that he has no conflict of interest.
Shayn M. Peirce-Cottler declares that she has no conflict of interest.
Norbert Leitinger declares that he has no conflict of interest.
Ethical approval
All animal studies were approved by the Animal Care and Use Committee at the University of Virginia.
Electronic supplementary material
Supplementary Figure 1
P2Y2 receptor activation is not required for macrophage expression of chemokines, Cxcl1 and Cxcl2. WT and P2Y2 deficient bone marrow derived macrophages were treated with 100 μm ATP or 200 μm UTP for 6 h and mRNA of chemokines Cxcl1 and Cxcl2 was measured by qRT-PCR. Data is normalized to β2-microglobulin mRNA and expressed as mean +/− s.e.m. (A) (GIF 4 kb)
Rights and permissions
About this article
Cite this article
Adamson, S.E., Montgomery, G., Seaman, S.A. et al. Myeloid P2Y2 receptor promotes acute inflammation but is dispensable for chronic high-fat diet-induced metabolic dysfunction. Purinergic Signalling 14, 19–26 (2018). https://doi.org/10.1007/s11302-017-9589-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11302-017-9589-9