Abstract
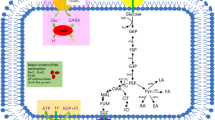
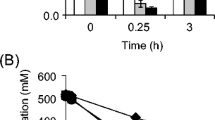
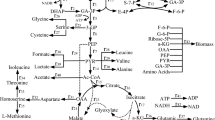
The accumulation of acetate in Escherichia coli inhibits cell growth and desired protein synthesis, and cell density and protein expression are increased by reduction of acetate excretion. Dissolved oxygen (DO) is an important parameter for acetate synthesis, and the accumulation of acetate is inversely correlated to DO level. In this study, the effect of DO levels on glutamate dehydrogenase (GDH) expression was investigated, and then different DO control strategies were tested for effects on GDH expression. DO control strategy IV (50% 0–9 h, 30% 9–18 h) provided the highest cell density (15.43 g/L) and GDH concentration (3.42 g/L), values 1.59- and 1.99-times higher than those achieved at 10% DO. The accumulation of acetate was 2.24 g/L with DO control strategy IV, a decrease of 40.74% relative to that achieved for growth at 10% DO. Additionally, under DO control strategy IV, there was lower expression of PoxB, a key enzyme for acetate synthesis, at both the transcriptional and translational level. At the same time, higher transcription and protein expression levels were observed for a glyoxylate shunt gene (aceA), an acetate uptake gene (acs), gluconeogensis and anaplerotic pathways genes (pckA, ppsA, ppc, and sfcA), and a TCA cycle gene (gltA). The flux of acetate with DO strategy IV was 8.4%, a decrease of 62.33% compared with the flux at 10% DO. This decrease represents both lower flux for acetate synthesis and increased flux of reused acetate.






Similar content being viewed by others
References
Baez A, Shiloach J (2013) Escherichia coli avoids high dissolved oxygen stress by activation of SoxRS and manganese superoxide dismutase. Microb Cell Fact 12:23
Bemal V, Castano-Cerezo S, Canovas M (2016) Acetate metabolism regulation in Escherichia coli: carbon overflow, pathogenicity, and beyond. Appl Microbiol Biotechnol 100(21):8985–9001
Castaño-Cerezo S, Pastor JM, Renilla S, Bemal V, Iborra JL, Canovas M (2009) An insight into the role of phosphotransacetylase (pta) and the acetate/acetyl-CoA node in Escherichia coli. Microb Cell Fact 8:54
Chen L, Zeng AP (2017) Rational design and metabolic analysis of Escherichia coli for effective production of l-tryptophan at high concentration. Appl Microbiol Biotechnol 101(2):559–568
Cheng LK, Tian FR, Fu Q, Li SG, Fu SJ, Yang XY, Qu GG, Miao LZ, Li F, Shen ZQ (2015) Application of dissolved oxygen stage control and dissolved oxygen feedback feeding to improve production of porcine circovirus type 2 capsid protein by Escherichia coli. J Chem Pharm Res 7(11):447–453
De Mets F, Van Melderen L, Gotlesman S (2019) Regulation of acetate netabolism and coordination with the TCA cycle via a processed small RNA. Proc Natl Aced Sci USA 116(3):1043–1052
Dong X, Chen X, Qian Y, Wang Y, Wang L, Qiao W, Liu L (2017) Metabolic engineering of Escherichia coli W3110 to produce L-malate. Biotechnol Bioeng 114(3):656–664
Eiteman MA, Altman E (2006) Overcoming acetate in Escherichia coli recombinant protein fermentations. Trends Biotechnol 24(11):530–536
Enjalbert B, Milland P, Dinclaux M, Portais JC, Letisse F (2017) Acetate fluxes in Escherichia coli are determined by the thermodynamic control of the Pta-AckA pathway. Sci Rep 7:42135
Kamata K, Toya Y, Shimizu H (2019) Effect of precise control flux ratio between the glycolytic pathways on mevalonate production in Escherichia coli. Biotechnol Bioeng 116(5):1180–1188
Kumar R, Shimizu K (2010) Metabolic regulation of Escherichia coli and its gdhA, glnL, gltB, D mutants under different carbon and nitrogen limitations in the continuous culture. Microb Cell Fact 9:8
Kutz R, Okwumabua O (2008) Differentiation of highly virulent strains of Streptococcus suis serotype 2 according to glutamate dehydrogenase electrophoretic and sequence type. J Clin Microbiol 46(10):3201–3207
Lin CW, Cheng LK, Wang J, Zhang SL, Fu Q, Li SG, Li F, Han WY, Shen ZQ (2016) Optimization of culture conditions to improve the expression level of beta1-epstion toxin of Clostridium perfringens type B in Escherichia coli. Biotechnol Buotechnoll Equip 30(2):324–331
Lin PP, Jaeger AJ, Wu TY, Xu SC, Lee AS, Gao P, Chen PW, Liao JC (2018) Construction and evolution of an Escherichia coli strain relying on nonoxidative glycolysis for sugar catabolism. PNAS 115(14):3538–3546
Lun ZR, Wang QP, Chen XG, Li AX, Zhu XQ (2007) Streptococcus suis: an emerging zoonotic pathogen. Lancet Infect Dis 7(3):201–209
Mortazavi A, Williams BA, Mccue K, Schaeffer L, Wold B (2008) Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5(7):621–628
Negrete N, Shiloach J (2017) Improving E. coli growth performance by manipulating small RNA expression. Microb Cell Fact 16(1):198
Parimi NS, Durie IA, Wu X, Niyas AM, Eiterman MA (2017) Eliminating acetate formation improves citramalate production by metabolically engineered Escherichia coli. Microb Cell Fact 16(1):114
Phue JN, Shiloach J (2005) Impact of dissolved oxygen concentration on acetate accumulation and physiology of E. coli BL21, evaluating transcription levels of key genes at different dissolved oxygen conditions. Metab Eng 7:353–363
Piao XY, Wang L, Lin BX, Chen H, Liu WF, Tao Y (2019) Metabolic engineering of Escherichia coli for production of L-aspartate ana its derivative β-alanine with high stoichiometric yield. Metab Eng 54:244–254
Pinhal S, Ropers D, Geiselmann J, de Jong H (2019) Acetate metabolism and the inhibition of bacterial growth by acetate. J Bacteriol 201(13):e00147-e219
Rajaraman E, Agrawal A, Crigler J, Seipelt-Thiemann R, Altman E, Eiteman MA (2016) Transcriptional analysis and adaptive evolution of Escherichia coli strains growing on acetate. Appl Microbiol Biotechnol 100(17):7777–7785
Rui B, Shen T, Zhou H, Liu JP, Chen JS, Pan XS, Liu HY, Wu JH, Zheng HR, Shi YY (2010) A systematic investigation of Escherichia coli central carbon metabolism in response to superoxide stress. BMC Syst Biol 4:122
Song HS, Seo HM, Jeon JM, Moon YM, Hong JG, Bhatia SK, Ahn J, Lee H, Kim W, Park YC, Choi KY, Kim YG, Yang YH (2018) Enhancd isobutanol production from acetate by combinatorial overexpression of acetyl-CoA synthetase and anaplerotic enzymes in engineered Escherichia coli. Biotechnol Bioeng 115(8):1971–1978
Tan ZG, Chen J, Zhang XL (2016) Systematic engineering of pentose phosphate pathway improves Escherichia coli succinate production. Biotechnol Biofuels 9:262
Veeravalli K, Schindler T, Dong E, Yamada M, Hamilton R, Laird MW (2018) Strain engineering to reduce acetate accumulation during microaerobic growth in Escheriichia coli. Biotechnol Prog 34(2):303–314
Wang J, Cheng LK, Wang J, Liu Q, Shen T, Chen N (2013) Genetic engineering of Escherichia coli to enhance production of L-tryptophan. Appl Microbiol Biotechnol 97:7587–7596
Wang Q, Xu JH, Sun ZJ, Luan LQ, Li Y, Wang JS, Liang QF, Qi QS (2019) Engineering an in vivo EP-bifido pathway in Escherichia coli for high-yield acetyl-CoA generation with low CO2 emission. Metab Eng 51:79–87
Wang J, Shang QM, Zhao CG, Zhang SS, Lin CW, Shen ZQ, Cheng LK (2020) Improvement of Streptococcus suis glutamate dehydrogenase expression in Escherichia coli through genetic modification of acetate synthesis pathway. Lett Appl Microbiol 70(2):64–70
Yang F, Yang L, Chang Z, Chang L, Yang B (2018) Regulation of virulence and motility cy acetate in enteropathogenic Escherichia coli. Int J Med Microbiol 308(7):840–847
Zhang CL, Li YJ, Ma J, Liu Y, He JL, Li YZ, Zhu FZ, Meng J, Zhan JJ, Li ZX, Zhao L, Ma Q, Fan XG, Xu QY, Xie XX, Chen N (2018) High production of 4-hydroxyisoleucine in Corynebacterium glutamicum by multistep metabolic engineering. Metab Eng 49:287–298
Zhao CG, Cheng LK, Xu QY, Wang J, Shen ZQ, Chen N (2016) Improvement of the production of L-tryptophan in Escherichia coli by application of a dissolved oxygen stage control strategy. Ann Microbiol 66(2):843–854
Zhao M, Huang DX, Zhang XJ, Koffas MAG, Zhou JW, Deng Y (2018) Metabolic engineering of Escherichia coli for producing adipic acid through the reverse adipate degradation pathway. Metab Eng 47:254–262
Zhao CG, Fang HT, Wang J, Zhang SS, Zhao XB, Li ZL, Lin CW, Shen ZQ, Cheng LK (2020) Application of fermentation process control to increase L-tryptophan production in Escherichia coli. Biotechnol Prog 36(2):e2944
Zheng S, Kwon I (2013) Controlling enzyme inhibition using an expression set of genetically encoded amino acids. Biotechnol Bioeng 110(9):2361–2370
Acknowledgements
This study was funded by the Major Science and Technology Innovation Project of Shandong (2019JZZY020606), the Development of Science and Technology Plan Program of Binzhou (2015ZC0107), the Technological Innovation Project of Shandong (201741916018), the Key Specialized Research and Development Breakthrough program in Henan Province (202102110101, 182102110347), Young Talent Lifting Project in Henan Province (2020HYTP041,2021HYTP038) , the National Natural Science Foundation of China (31702263) and the Youth Backbone Teacher Project of Colleges and Universities of Henan Province (2020GGJS163).
Author information
Authors and Affiliations
Contributions
LC, XX and ZS designed the research. LC and XX performed the experiments. LC, CZ, ZS, and CL acquired and analyzed the data. XZ, JW, JW, and LW contributed to writing of the manuscript. Finally, all authors have reviewed and approved the final submitted manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cheng, L., Zhao, C., Yang, X. et al. Application of a dissolved oxygen control strategy to increase the expression of Streptococcus suis glutamate dehydrogenase in Escherichia coli. World J Microbiol Biotechnol 37, 60 (2021). https://doi.org/10.1007/s11274-021-03025-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-021-03025-2