Abstract
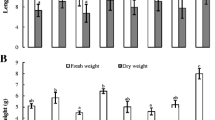
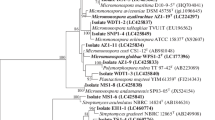
Twenty-seven endophytic actinomycete strains were isolated from five spontaneous plants well adapted to the poor sandy soil and arid climatic conditions of the Algerian Sahara. Morphological and chemotaxonomical analysis indicated that twenty-two isolates belonged to the Streptomyces genus and the remaining five were non-Streptomyces. All endophytic strains were screened for their ability to produce indole-3-acetic acid (IAA) in vitro on a chemically defined medium. Eighteen strains were able to produce IAA and the maximum production occurred with the Streptomyces sp. PT2 strain. The IAA produced was further extracted, partially purified and confirmed by thin layer chromatography (TLC) analysis. The 16S rDNA sequence analysis and phylogenetic studies indicated that strain PT2 was closely related to Streptomyces enissocaecilis NRRL B 16365T, Streptomyces rochei NBRC 12908T and Streptomyces plicatus NBRC 13071T, with 99.52 % similarity. The production of IAA was affected by cultural conditions such as temperature, pH, incubation period and l-tryptophan concentration. The highest level of IAA production (127 μg/ml) was obtained by cultivating the Streptomyces sp. PT2 strain in yeast extract-tryptone broth supplemented with 5 mg l-tryptophan/ml at pH 7 and incubated on a rotary shaker (200 rpm) at 30 °C for 5 days. Twenty-four-hour treatment of tomato cv. Marmande seeds with the supernatant culture of Streptomyces sp. PT2 that contained the crude IAA showed the maximum effect in promoting seed germination and root elongation.






Similar content being viewed by others
References
Ahmad F, Ahmad I, Khan MS (2005) Indole acetic acid production by the indigenous isolates of azotobacter and fluorescent Pseudomonas in the presence and absence of tryptophan. Turk J Biol 29:29–34
Aldesuquy HS, Mansour FA, Abo-Hamed SA (1998) Effect of the culture filtrates of Streptomyces on growth and productivity of wheat plants. Folia Microbiol 43:465–470
Becker B, Lechevalier MP, Gordon RE, Lechevalier HA (1964) Rapid differentiation between Nocardia and Streptomyces by paper chromatography of whole cell hydrolysates. J Appl Microbiol 12:421–423
Benson DR, Silvester WB (1993) Biology of Frankia strains actinomycetes symbionts of actinorhizal plant. Microbiol Rev 57:293–319
Brown ME (1972) Plant growth substances produced by microorganisms of soil and rhizosphere. J Appl Bacteriol 35:443–451
Chun J, Bae KS, Moon EY, Jung SO, Lee HK, Kim SJ (2000) Nocardiopsis kunsanensis sp. nov, a moderately halophilic actinomycete isolated from a saltern. Int J Syst Evol Microbiol 50:1909–1913
Coa L, Qiu Z, You J, Tan H, Zhou S (2004) Isolation and characterization of endophytic Streptomyces from surface-sterilized tomato (Lycopersicun esculentum) roots. Lett Appl Microbiol 39:425–430
Coa L, Qiu Z, You J, Tan H, Zhou S (2005) Isolation and characterization of endophytic streptomycetes antagonists of fusarium wilt pathogen from surface-sterilized banana roots. FEMS Microbiol Lett 247:147–152
Datta C, Basu PS (2000) Indole acetic acid production by a Rhizobium species from root nodules of a leguminous shrub, Cajanus cajan. Microbiol Res 155:123–127
El-Tarabily KA (2008) Promotion of tomato (Lycopersicon esculentum Mill) plant growth by rhizosphere competent 1-aminocyclopropane-1-carboxylic acid deaminase-producing streptomycete actinomycetes. Plant Soil 308:161–174
El-Tarabily KA, Nasser AH, Hardy GESJ, Sivaithamparam K (2008) Plant growth promotion and biological control of Pythium aphanidermatum, a pathogen of cucumber, by endophytic actinomycetes. J App Microbiol 106:13–26
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Goodfellow M, Simpson KE (1987) Ecology of streptomycetes. Frontiers. Appl Microbiol 2:97–125
Hayakawa M, Nonomura H (1987) Humic acid-vitamin agar, a new medium for the selective isolation of soil actinomycetes. J Ferment Technol 65:501–509
Ilic SB, Konstantinovic SS, Todorovic ZB, Lazic ML, Veljkovic VB, Jokovic N, Radovanovic BC (2007) Characterization and antimicrobial activity of the bioactive metabolites in streptomycete isolates. Microbiology 76:421–428
Kaunat H (1969) Bidung von indolderivaten durch rhizospha reenspezifisch bakterien und aktinomyzeten. Zentralblatt fuer Bakeriologie Abteilung II 123:501–515
Khamna S, Yokota A, Peberdy JF, Lumyong S (2010) Indole-3-acetic acid production by Streptomyces sp. isolated from some Thai medicinal plant rhizosphere soils. EurAsia J BioSci 4:23–32
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Kukavica B, Mitrović A, Mojović M, Veljović-jovanović S (2007) Effect of indole-3-acetic acid on pea root growth, peroxidase profiles and hydroxyl radical formation. Arch Biol Sci 59:319–326
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) CLUSTALW and CLUSTALX version 2. Bioinformatics 23:2947–2948
Liu D, Coloe S, Baird R, Pedersen J (2000) Rapid mini-preparation of fungal DNA for PCR. J Clin Microbiol 38:471
Merckx R, Dijkra A, Hartog AD, Veen JAV (1987) Production of root-derived material and associated microbial growth in soil at different nutrient levels. Biol Fert Soils 5:126–132
Qin S, Li J, Chen H, Zhao G, Zhu W, Jiang C, Xu L, Li WJ (2009) Isolation, diversity, and antimicrobial activity of rare actinobacteria from medicinal plants of tropical rain forests in Xishuangbanna, China. Appl Environ Microbiol 75:6176–6186
Ruanpanun P, Tangchitsomkid N, Hyde KD, Lumyong S (2010) Actinomycetes and fungi isolated from plant-parasitic nematode infested soils: screening of the effective biocontrol potential, indole-3-acetic acid and siderophore production. World J Microbiol Biotechnol 26:1569–1578
Sadeghi A, Karimi J, Abaszadeh D, Javid MG, Dalvand Y, Askari H (2012) Plant growth promoting activity of an auxin and siderophore producing isolate of Streptomyces under saline soil conditions. World J Microbiol Biotechnol 28:1503–1509
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sapak Z, Meon S, Zam A (2008) Effect of endophytic bacteria on growth and suppression of Ganoderma infection in oil palm. Int J Agric Biol 10:127–132
Shi Y, Lou K, Li C (2009) Promotion of plant growth by phytohormone-producing endophytic microbes of sugar beet. Biol Fertil Soils 45:645–653
Shirling EB, Gottlieb D (1966) Methods for characterization of Streptomyces species. Int J Syst Bacteriol 16:313–340
Shirokikh IG, Zenova GM, Merzaeva OV, Lapygina EV, Batalova GA, Lysak LV (2007) Actinomycetes in the prokaryotic complex of the rhizosphere of oats in a soddy-podzolic soil. Eurasian Soil Sci 40:158–162
Solans M, Vobis G, Cassa′n F, Luna V, Wall LG (2011) Production of phytohormones by root-associated saprophytic actinomycetes isolated from the actinorhizal plant Ochetophila trinervis. World J Microbiol Biotechnol 27:2195–2202
Suzuki S, Yamamoto K, Okuda T, Nishio M, Nakanishi N, Komatsubara S (2000) Selective isolation and distribution of Actinomadura rugatobispora strains in soil. Actinomycetologica 14:27–33
Taechowisan T, Peberdy JF, Lumyong S (2003) Isolation of endophytic actinomycetes from selected plants and their antifungal activity. World J Microbiol Biotechnol 19:381–385
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum Parsimony methods. Mol Biol Evol 28:2731–2739
Tan HM, Coa LX, He ZF, Su JG, Lin B, Zhou SN (2006) Isolation of endophytic actinomycetes from selected plants and their antifungal activity. World J Microbiol Biotechnol 22:1275–1280
Ting ASY, Meon S, Kadir J, Radu S, Singh G (2008) Endophytic microorganisms as potential growth promoters of banana. Biocontrol 53:541–553
Tokala RK, Strap JL, Jung CM, Crawford DL, Salove MH, Deobald LA, Bailey JF, Morra MJ (2002) Novel plant–microbe rhizosphere interaction involving Streptomyces lydicus WYEC108 and the pea plant (Pisum sativum). App Environ Microbiol 68:2161–2171
Vikram P (2011) Production of indole acetic acid by Azotobacter sp. Recent Res Sci Technol 3:14–16
Wilson D (1995) Endophyte—the evolution of a term and clarification of its use and definition. Oikos 73:274–276
Xie H, Pasternak JJ, Glick BR (1996) Isolation and characterization of mutants of the plant growth-promoting rhizobacterium Pseudomonas putida GR-122 that overproduce indole-3-acetic acid. Curr Microbiol 32:67–71
Yurekli F, Geckil H, Topcuoglu F (2003) The synthesis of indole-3-acetic acid by the industrially important white-rot fungus Lentinus sajor-caju under different culture conditions. Mycol Res 107:305–309
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goudjal, Y., Toumatia, O., Sabaou, N. et al. Endophytic actinomycetes from spontaneous plants of Algerian Sahara: indole-3-acetic acid production and tomato plants growth promoting activity. World J Microbiol Biotechnol 29, 1821–1829 (2013). https://doi.org/10.1007/s11274-013-1344-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-013-1344-y